Introduction
Malaria remains one of the world’s leading infectious diseases, causing approximately 263 million cases and 597 000 deaths annually (World Malaria Report, 2024). It is transmitted to humans through the bite of Anopheles mosquitoes. The World Health Organization recently identified climate change as one of the greatest threats to human health. Climatic factors play a crucial role in the occurrence and distribution of vector-borne diseases, including malaria (Caminade et al., Reference Caminade, McIntyre and Jones2019; Nazemosadat et al., Reference Nazemosadat, Shafiei, Ghaedamini, Najjari, Nazemosadat-Arsanjani and Hatam2022). Although the relationship between climate and malaria is not yet fully understood, factors such as temperature, precipitation and humidity are known to create optimal conditions for mosquito reproduction and parasite transmission. Rising temperatures and changing precipitation patterns may expand malaria transmission to higher altitudes and latitudes or, conversely, reduce transmission where conditions become too hot or dry (Van Lieshout et al., Reference Van Lieshout, Kovats, Livermore and Martens2004).
Malaria transmission exhibits seasonal patterns in tropical and subtropical regions, typically following precipitation cycles. Precipitation provides breeding habitats for mosquitoes, while temperature affects mosquito development and the parasite’s extrinsic incubation period. Because transmission depends on these vector biological processes, climatic effects often appear with a delay, creating lag relationships between climatic conditions and malaria incidence (Kim et al., Reference Kim, Park and Cheong2012; Caminade et al., Reference Caminade, Kovats, Rocklov, Tompkins, Morse, Colón-González, Stenlund, Martens and Lloyd2014; Zinszer et al., Reference Zinszer, Kigozi, Charland, Dorsey, Brewer, Brownstein, Kamya and Buckeridge2015). Understanding these seasonal patterns and lag relationships is essential for improving prediction models and the optimal timing of interventions.
Vector control remains a cornerstone of malaria prevention. The World Health Organization (WHO, 2019) recommends long-lasting insecticidal nets and indoor residual spraying (IRS) as primary malaria vector control measures, with larviciding applied where appropriate. Understanding the effects of vector control interventions and climatic conditions provides a basis for designing effective, evidence-based prevention strategies. Although several studies in Iran have examined the relationship between climate and malaria (Babaie et al., Reference Babaie, Barati, Azizi, Ephtekhari and Sadat2018; Mohammadkhani et al., Reference Mohammadkhani, Khanjani, Bakhtiari, Tabatabai and Sheikhzadeh2019; Nili et al., Reference Nili, Khanjani, Ghaderi and Mohammadkhani2023), these analyses have mainly focused on ecological correlations or short-term data and have not considered IRS operational coverage or delayed effects. Therefore, the evidence is limited, and it is still unclear what direct contribution climate change and IRS implementation have at the sub-provincial level.
In Iran, malaria remains a climate-sensitive public health issue, particularly in the southeastern provinces of Sistan and Baluchestan and Hormozgan, which account for the majority of cases nationwide. Transmission in these areas is related to environmental conditions and the presence of malaria vector Anopheles species (Norouzinezhad et al., Reference Norouzinezhad, Ghaffari, Raeisi, Norouzinejad and Kaveh2017; Hanafi-Bojd et al., Reference Hanafi-Bojd, Sedaghat, Vatandoost, Azari-Hamidian and Pakdad2018).
This study aimed to examine the association between climatic variability and malaria incidence in southeast Iran using longitudinal count-based regression models that account for lagged effects. In a separate analytical framework, the relationship between IRS coverage indicators and malaria incidence over time in the same region was also assessed. We hypothesized that elevated precipitation and temperature, after accounting for biologically relevant time lags, would be positively associated with subsequent increases in malaria incidence. In contrast, higher IRS coverage was expected to result in a reduction in malaria incidence over the ensuing periods. Moreover, seasonal patterns of malaria transmission were anticipated across the province.
Materials and methods
Study area
Sistan and Baluchestan Province covers an area of 180 726 square kilometres and is the second largest province in Iran. In terms of international borders, the province shares borders with Afghanistan to the north and Pakistan to the east. The southern border of the province is the coast of the Sea of Oman (Mohammadkhani et al., Reference Mohammadkhani, Khanjani, Bakhtiari, Tabatabai and Sheikhzadeh2019). During the study period (2001–2023), the province underwent multiple administrative boundary changes, increasing the number of counties to 26. To ensure temporal comparability in long-term analyses, spatial harmonization was performed. For analyses assessing temporal trends and the association between climatic factors and malaria incidence, data were aggregated to the eight original base counties defined at the beginning of the study period: Iranshahr, Khash, Zabol, Zahedan, Chabahar, Sarbaz, Saravan and Nikshahr. Counties established during the study period were reassigned to their respective original counties to maintain consistent spatial units over time (Figure 1).
Study area map.

In contrast, analyses evaluating the impact of IRS and other vector control interventions were conducted at the 26-county level, as intervention coverage data were recorded and implemented according to the updated administrative structure. IRS coverage data were available for the period 2013–2023, which did not fully overlap with the period of climatic data collection; therefore, intervention-effect analyses were restricted to the timeframe with available IRS records. Using the finer spatial resolution allowed for a more accurate representation of IRS coverage and retained local variability in intervention intensity.
Study design
This is a retrospective descriptive–analytical study examining the relationship between climatic factors, seasonal patterns and vector control interventions on malaria incidence in Sistan and Baluchestan Province.
Data collection and management
Meteorological data: monthly average data on temperature (°C), relative humidity (%) and precipitation (mm) were obtained from the National Meteorological Organization (affiliated with the Iranian Ministry of Roads and Urban Development) and active synoptic stations in the study cities for the period 2001–2023.
Malaria incidence rate: Monthly malaria case data for 2001–2023 were obtained from the Communicable Disease Management of the Ministry of Health and Medical Education and the Sistan and Baluchestan University of Medical Sciences. Monthly incidence rates per 100 000 population were calculated using county-level population denominators (see formula 1).
Population data: County-level population denominators were harmonized to the original administrative boundaries to ensure temporal comparability across the 23-year study period. For years without available population data, values were interpolated using annual growth rates derived from census data, allowing accurate estimation of malaria incidence and enabling the inclusion of population as an offset in regression analyses. Population mobility and cross-border migration were not explicitly accounted for; however, harmonized and interpolated denominators minimize their impact on incidence calculations.
Vector control interventions: Data on IRS and bed net distribution were obtained from the Center for Communicable Disease Management of the Ministry of Health. IRS records, including household, structural and population coverage, were available for selected counties from 2013 to 2023. Bed net distribution data were generally available from 2009 to 2022. Analyses of vector control interventions were restricted to counties and years with available data, using the finer 26-county spatial resolution to preserve local variability in intervention intensity.
 \begin{align*}{\text{Formula }}1: & {\text{ Incidence Rate }} \nonumber\\
& = \frac{{{{Number\,\,of\,\,new\,\,cases\,\,of\,\,disease}}}}{{{{Total\,\,person}} - {{time\,\,at\,\,risk}}}} \times 100000\end{align*}
\begin{align*}{\text{Formula }}1: & {\text{ Incidence Rate }} \nonumber\\
& = \frac{{{{Number\,\,of\,\,new\,\,cases\,\,of\,\,disease}}}}{{{{Total\,\,person}} - {{time\,\,at\,\,risk}}}} \times 100000\end{align*}Statistical analysis
For the eight base counties, malaria incidence was analysed using count-based regression models. Negative binomial (NB) regression was applied to Chabahar, Nikshahr, Zahedan, Saravan, Sarbaz and Iranshahr due to overdispersion in the counts. In Zabol and Khash, where a high proportion of zero counts made the NB model unstable, a Zero-Inflated Poisson (ZIP) model was used to account for both structural zeros and non-zero counts. These models were used to investigate the associations between malaria incidence and climatic factors (temperature, relative humidity and precipitation), considering lagged exposures of 1–3 months to account for delayed effects of climate on transmission.
The relationship between Seasonal patterns and malaria incidence was assessed using a linear mixed-effects model (LMM) with log-transformed incidence rates as the dependent variable. Season (winter, spring, summer and autumn) and year (centred) were included as fixed effects, and county as a random intercept. Winter was the reference. Fold-change estimates relative to winter were obtained by exponentiating model coefficients, with 95% confidence intervals. Seasonal differences are reported at the mean year, controlling for long-term temporal trends.
Due to differences in spatial and temporal resolution between climatic and IRS datasets, integrated modelling was not feasible. We retrospectively analysed the impact of vector control interventions on malaria incidence in 13 counties of Sistan and Baluchestan province, Iran, from 2013 to 2023. IRS was conducted biannually in March and September. For each county, 6-month cumulative IRS coverage was calculated for household, structural and population indicators. High collinearity among these variables (VIF > 20 for household and population coverage) led us to select population coverage for modelling, as it best reflects the proportion of the population protected while avoiding multicollinearity. Population coverage was scaled in 10% increments to facilitate the interpretation of incidence rate ratios (IRRs). Monthly malaria incidence per 100 000 population was modelled using a NB generalized linear model with logarithmic link, including county as a fixed effect. In this model, Chabahar County was selected as the reference county due to its maximum data coverage, and other counties were compared to it. Spearman correlation was applied to assess monotonic associations between climatic factors, vector control interventions and malaria incidence. This method was chosen due to non-normal distributions and potential extreme values, providing robust and interpretable correlations.
For bed net distribution, county-level data were limited in both spatial and temporal coverage. Formal regression models (Poisson or NB) were inappropriate due to the small sample size, which could yield unstable estimates. Instead, Spearman correlation was employed and results visualized using heatmaps to descriptively evaluate the relationship between the number of distributed bed nets and malaria incidence, minimizing potential bias or overfitting. Statistical analyses were performed using Python (Python Software Foundation) in the Jupyter Notebook environment, with data management and manipulation carried out using the Pandas library.
Results
Monthly average data on temperature, relative humidity and precipitation from 2001 to 2023 were analysed to assess their association with malaria incidence in eight counties of Sistan and Baluchestan Province (Supplementary Table 1). In addition to climatic analyses, seasonal patterns and the impact of vector control interventions, including IRS and bed net distribution, were examined.
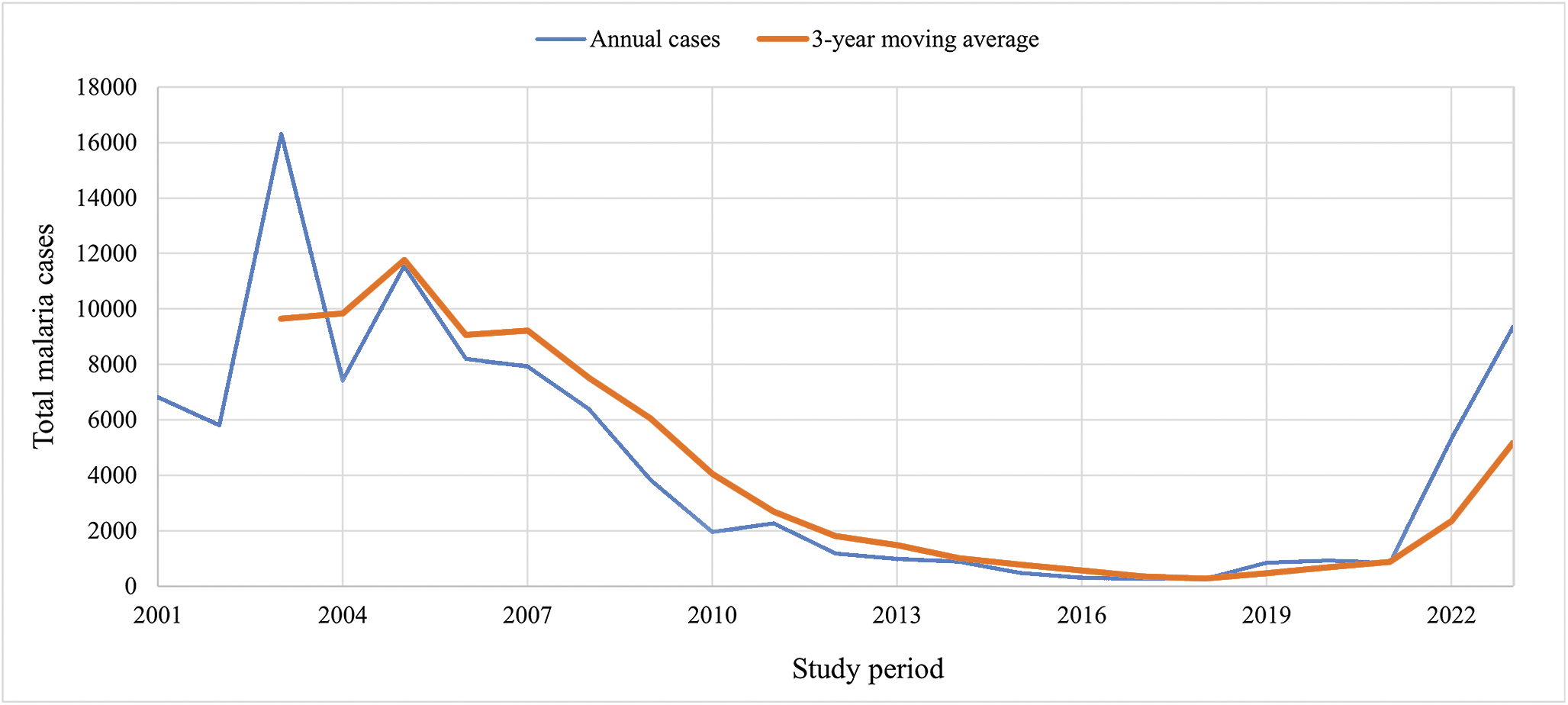
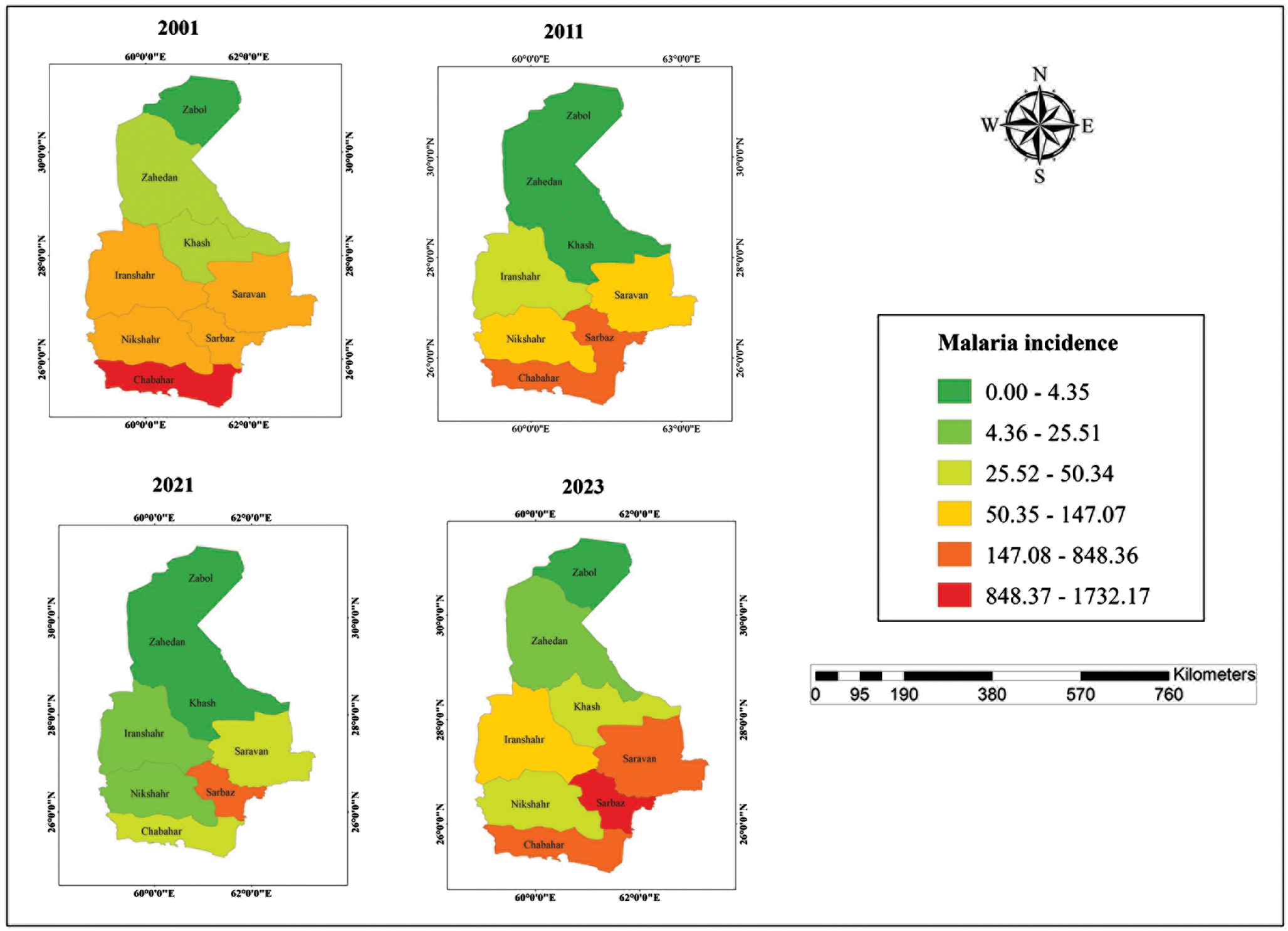
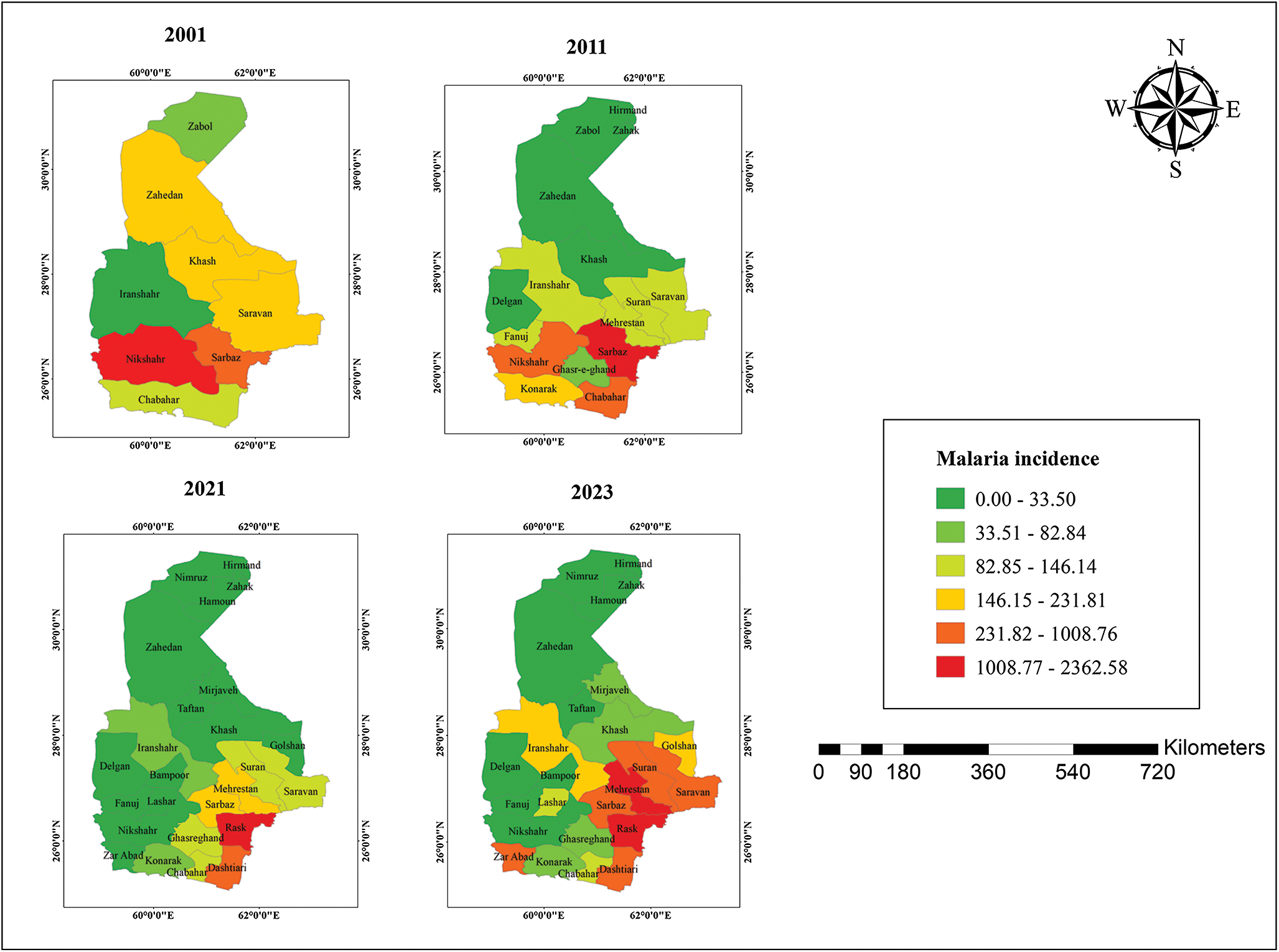
Between 2001 and 2023, malaria incidence exhibited an overall declining trend, and during the elimination phase, local transmission reached zero for three consecutive years. However, a resurgence was observed in 2022 and 2023, with 5314 and 9366 reported cases, respectively (Figure 2). These data were obtained from the Center for Communicable Disease Control, Ministry of Health and Medical Education, Iran, and the Sistan and Baluchestan Universities of Medical Sciences. A clear north–south spatial gradient in malaria incidence was observed (Figure 3). Southern counties consistently exhibited higher incidence rates, whereas northern counties such as Zabol reported substantially lower transmission. At finer spatial resolution (26 counties), this heterogeneity became more pronounced, revealing localized hotspots within the southern districts (Figure 4).
Trend of total malaria cases in Sistan and Baluchestan Province, Iran, 2001–2023.

Incidence of malaria (per 100 000 population) in various Counties of Sistan and Baluchestan Province, Iran.

Malaria incidence (per 100 000 population) in various cities of Sistan and Baluchestan Province, Iran.

The relationship between malaria incidence and climatic factors
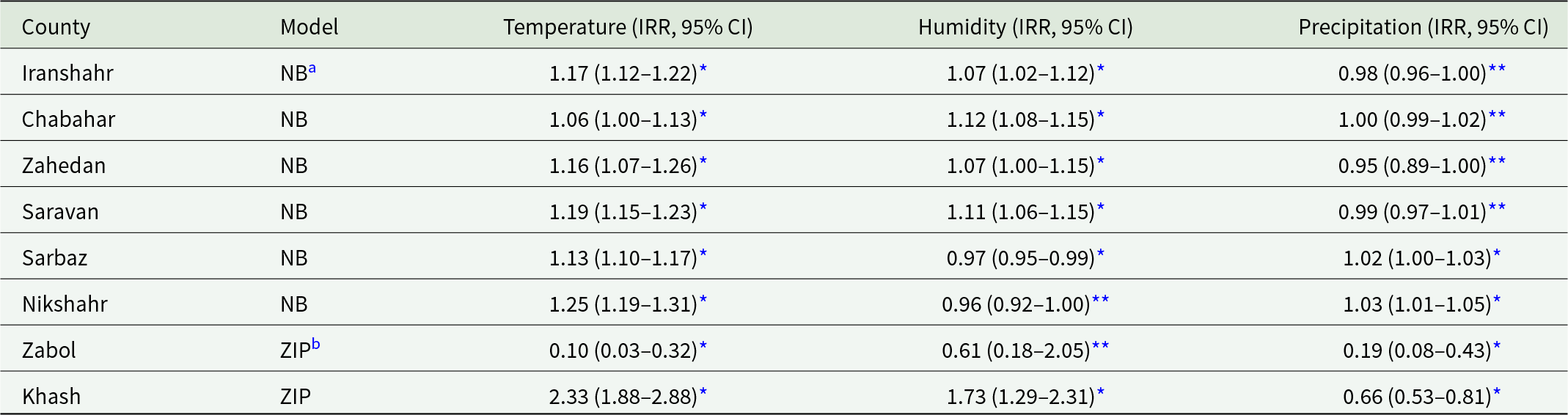
Across all eight counties, temperature showed a consistent positive association with malaria incidence across both modelling approaches. Increases in lagged mean temperature were associated with higher incidence in all counties, with statistically significant effects throughout. In contrast, associations for relative humidity and precipitation were more heterogeneous. Relative humidity demonstrated significant positive associations in several counties but was non-significant in others. Precipitation effects were inconsistent, with positive associations in some counties and negative or non-significant associations in others. The results of the regression analyses examining the associations between climatic factors and malaria incidence across the 8 study counties are summarized in Table 1.
Association between climatic factors and malaria incidence across eight counties using NB and ZIP models

a NB = Negative binomial regression (lag 1–3 month mean values).
b ZIP = Zero-Inflated Poisson regression (count component).
* Significant (P < 0.001).
** Non-significant (P > 0.05).
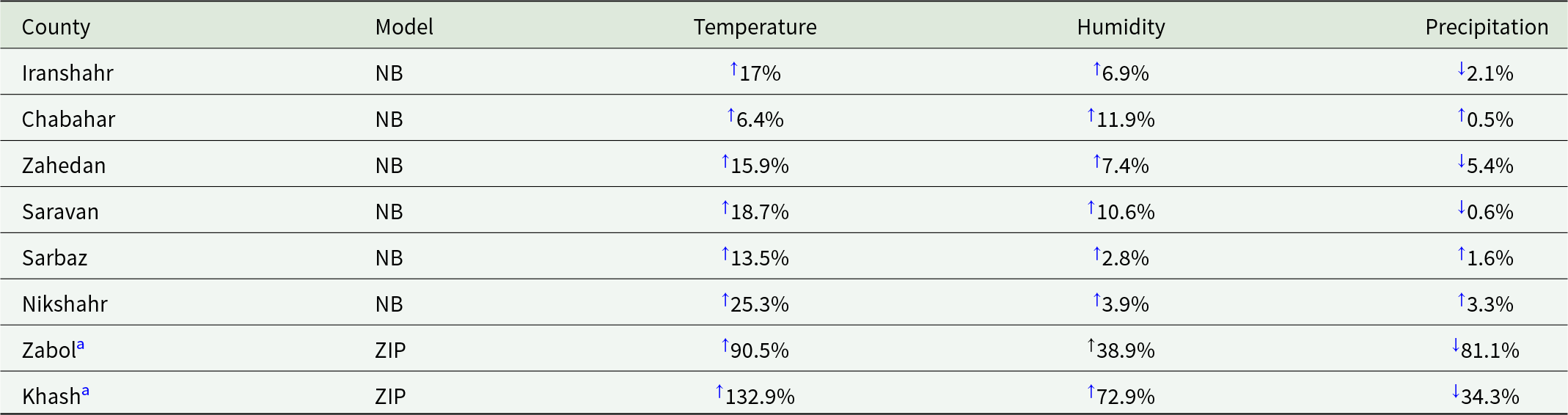
The corresponding percentage change in malaria incidence per one-unit increase in each climatic variable is presented in Table 2, highlighting the spatial variation in effect sizes across counties. (In Zabol and Khash, due to a high proportion of zero counts, resulting in larger and sometimes negative percentage changes for temperature, humidity and precipitation, reflecting the impact of many months with zero cases.) Forest plots summarizing county-specific associations between climatic factors and malaria incidence are presented in (Supplementary Figure 1), providing a visual representation of IRRs and 95% CIs across counties.
Percentage change in malaria incidence per one-unit increase in climatic factors (NB and ZIP models)

↑ Increase.
↓ Decrease.
a Zabol and Khash used standardized climatic variables (1 SD increase).
Heat map analysis of climatic factors and malaria incidence (Supplementary Figure 2) suggested a generally positive association between temperature and malaria incidence across the studied counties, with relatively stronger correlations observed in Chabahar (correlation coefficient = 0.50). Relative humidity showed positive correlations in several counties, including Iranshahr, Chabahar, Khash, Zahedan and Nikshahr, again with comparatively higher values in Chabahar (0.54). In contrast, precipitation demonstrated weak and less consistent correlations across counties. Generally, these results indicate that temperature and relative humidity are the climatic factors most consistently associated with malaria incidence across the studied counties, whereas precipitation generally shows limited influence.
The relationship between malaria incidence and seasonal patterns
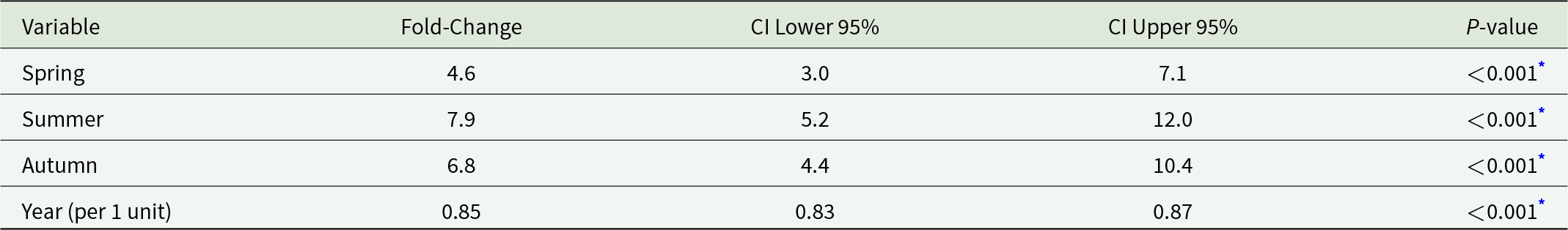
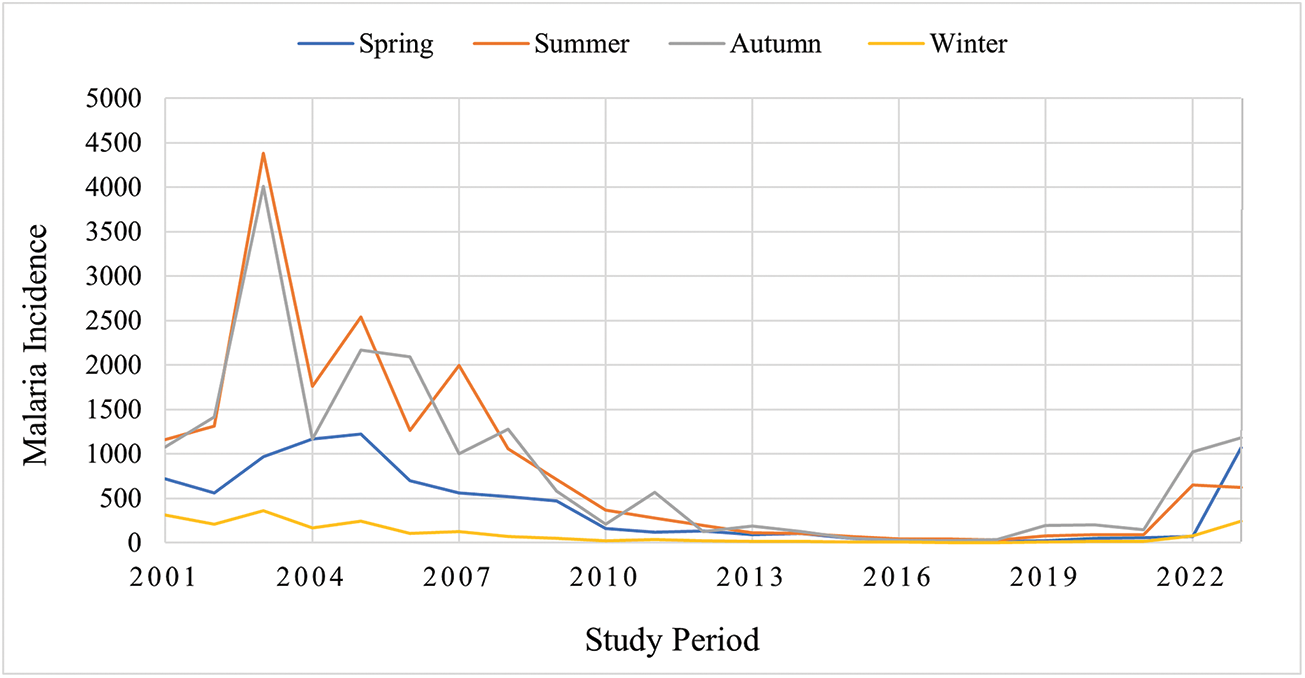
Malaria incidence also exhibited seasonal patterns. Based on exponentiated coefficients from a mixed-effects model, incidence was approximately 4.6-fold higher in spring (95% CI: 3.0–7.1), 7.9-fold higher in summer (95% CI: 5.2–12.0), and 6.8-fold higher in autumn (95% CI: 4.4–10.4), relative to winter (Table 3). A declining temporal trend was also observed, with an average decrease of 15% per year (95% CI: 13–17%). These findings indicate that malaria transmission peaks during the warmer and transitional seasons – particularly summer and autumn – while remaining lowest in winter. The results account for inter-county variation and highlight the influence of seasonality on malaria incidence.
Fold-change estimates for seasonal patterns and malaria incidence

* Significant (P < 0.001).
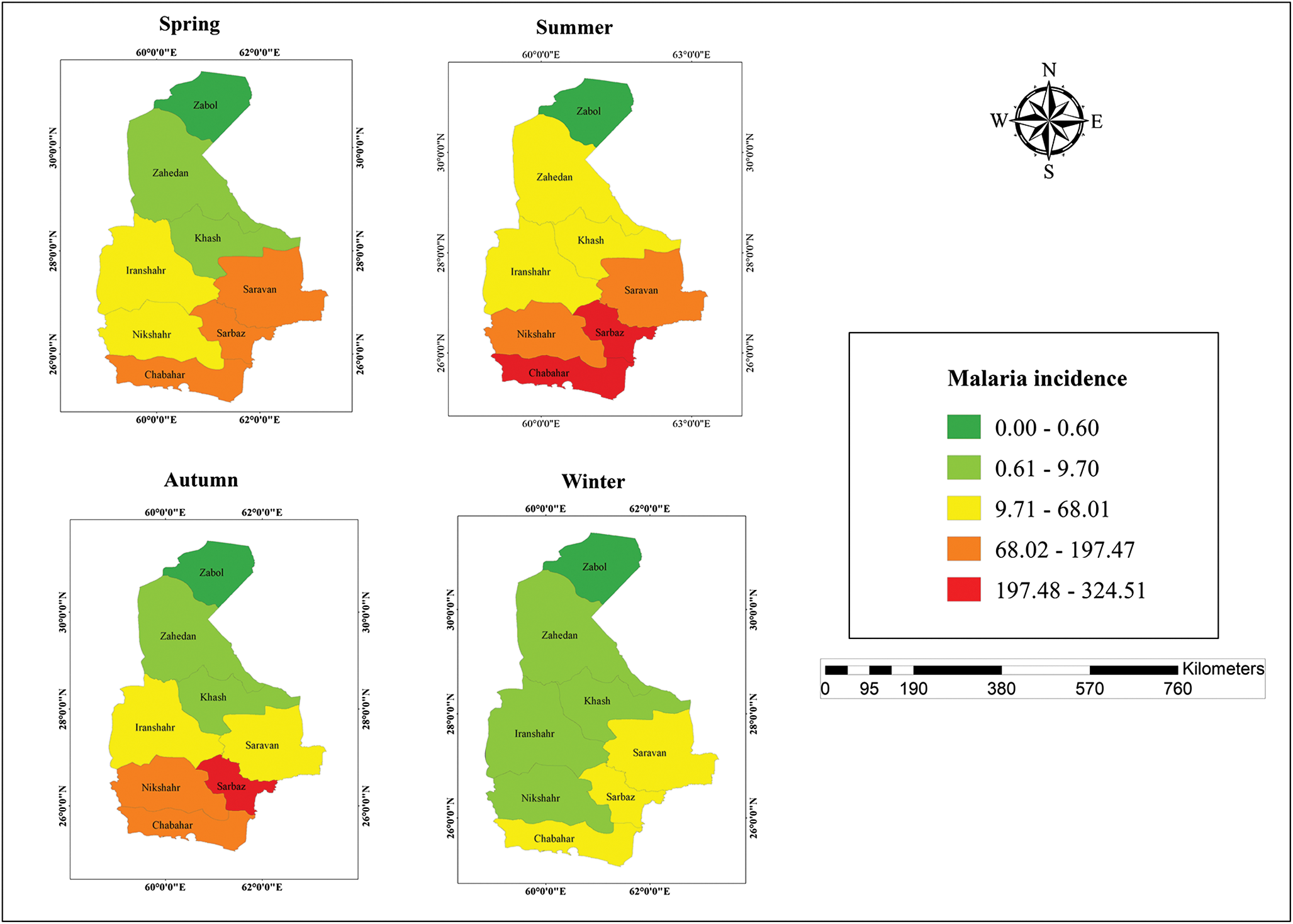
Seasonal incidence maps illustrate the spatial distribution of malaria cases across spring, summer, autumn and winter over the 23-year study period (Figure 5), highlighting the heterogeneous seasonal burden across counties. The seasonal trend plot further demonstrates that malaria incidence peaks during summer and autumn and reaches its lowest values in winter (Figure 6), consistent with the mixed-effects model estimates.
Map of seasonal malaria incidence (per 100 000 population) in counties of Sistan and Baluchestan Province, Iran, 2001–2023.

Map of seasonal Malaria incidence in eight counties of Sistan and Baluchestan Province, Iran, 2001–2023.

Relationship between vector control interventions and malaria incidence
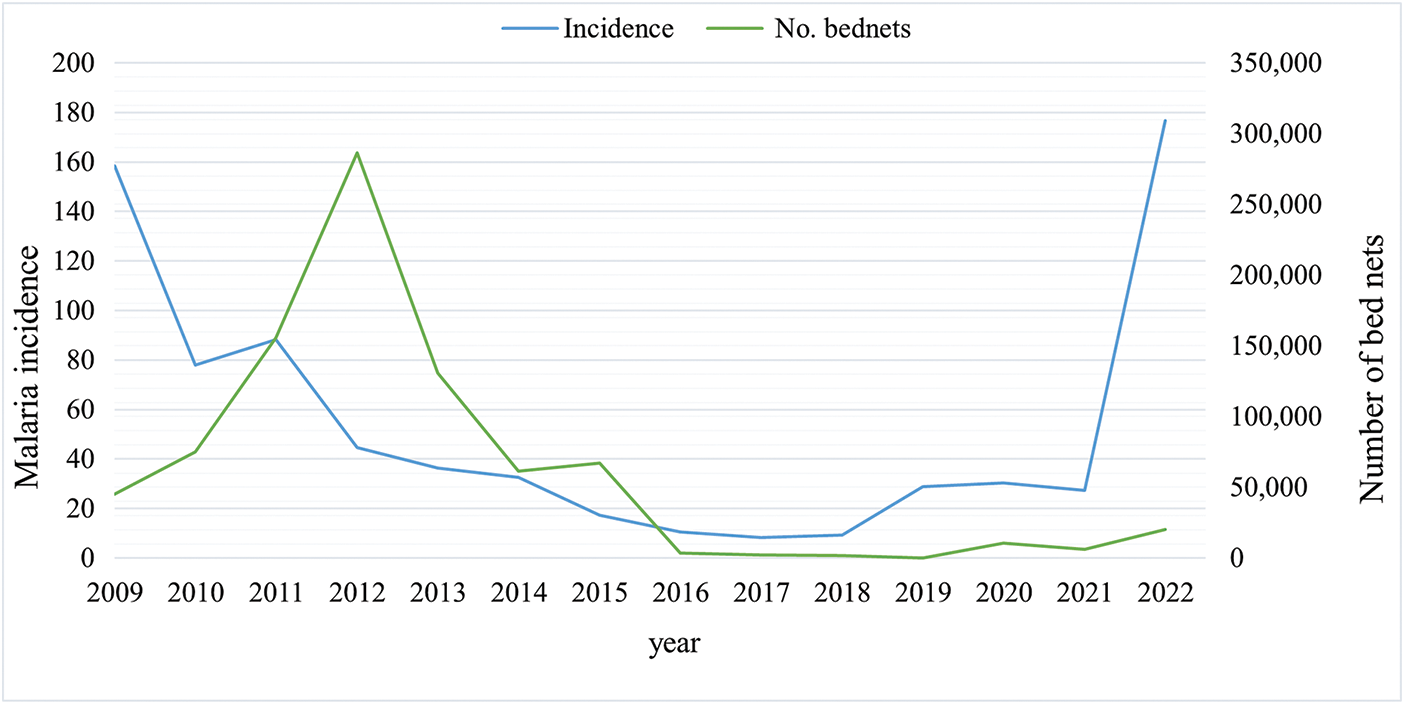
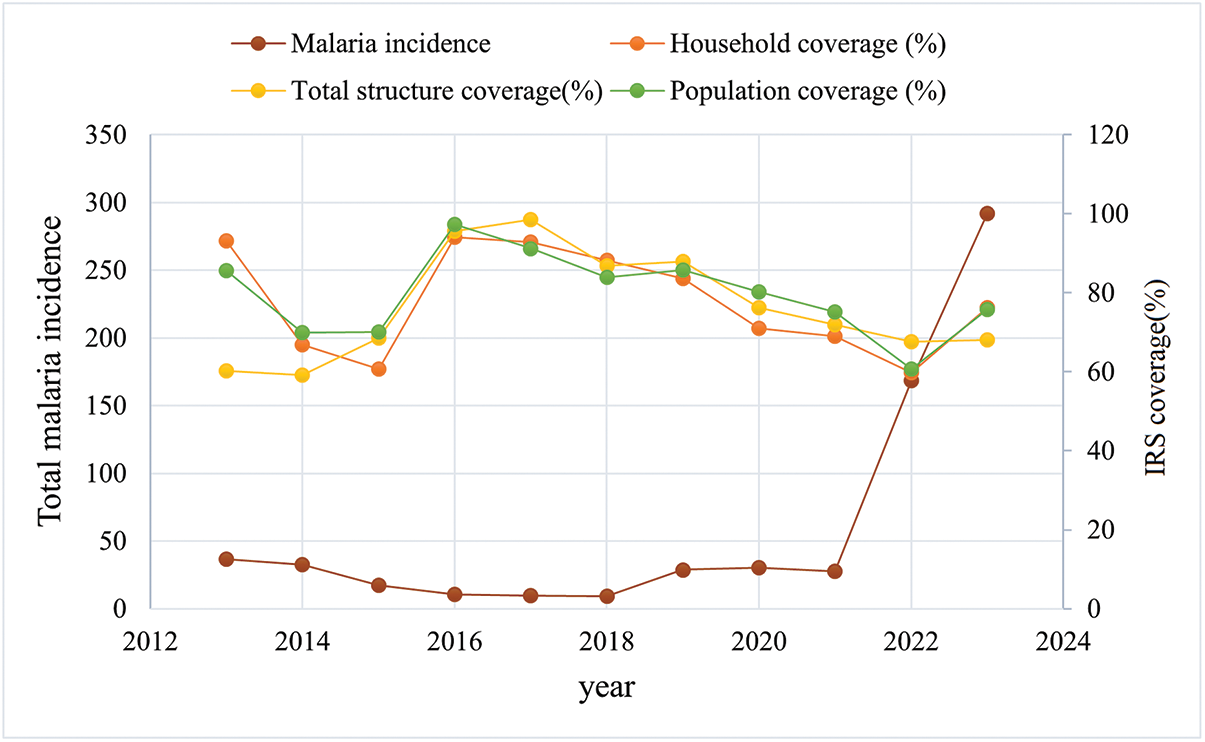
This study examined the relationship between IRS and bed net distribution and malaria incidence. According to the analyses, generally, higher coverage of these vector control interventions corresponded with a declining trend in malaria incidence (Figures 7 and 8).
Number of Bed nets distributed and malaria incidence in Sistan and Baluchestan Province, Iran, 2009–2022.

Number of houses sprayed and number of people protected by IRS, against malaria incidence in Sistan and Baluchestan Province, Iran, 2012–2023.

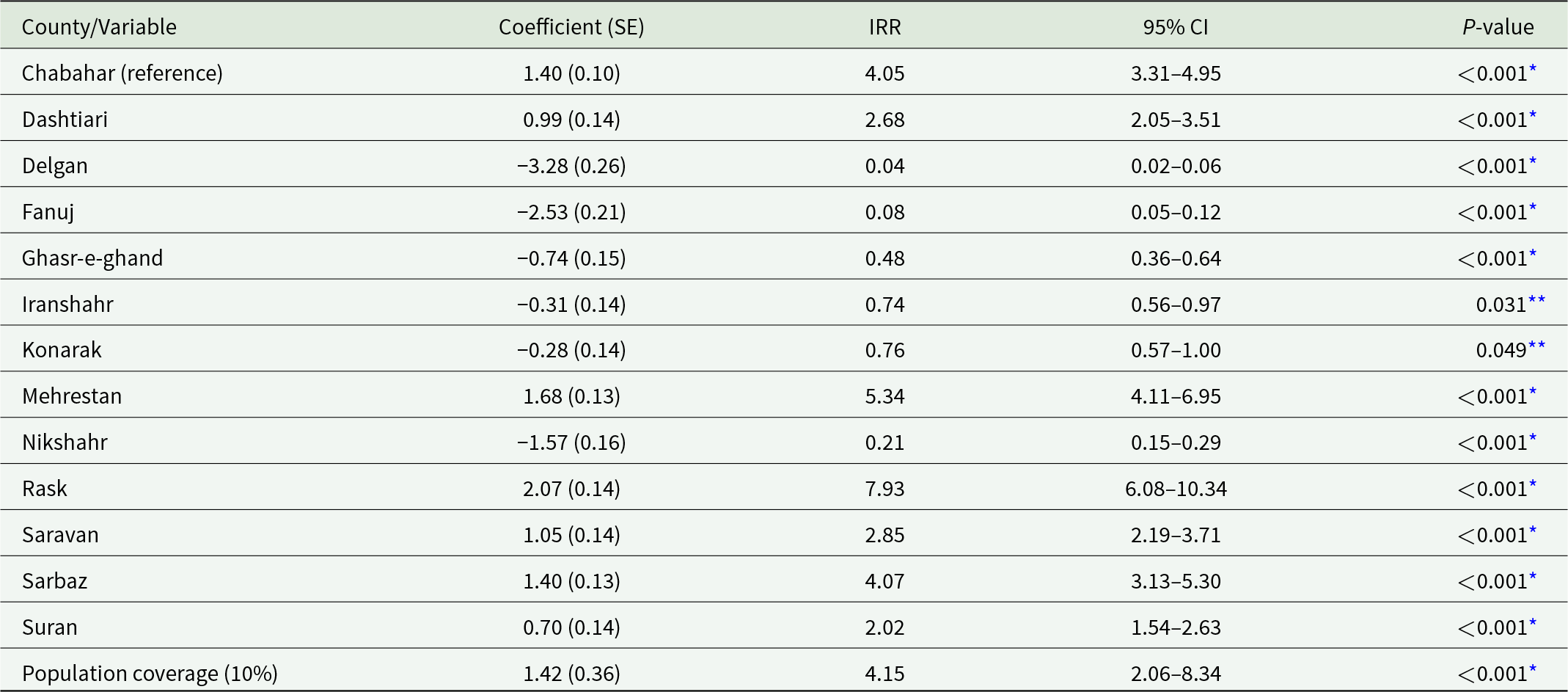
Although reductions in malaria incidence were observed in some cities, the overall model indicated that a 10% increase in cumulative IRS population coverage over the previous 6 months was associated with a 4.15-fold increase in incidence (IRR = 4.15, 95% CI: 2.06–8.34, P < 0.001; Table 4). County-specific effects, with Chabahar as the reference, reflected local differences in endemicity and intervention targeting. Some counties, such as Delgan and Fanuj, exhibited reductions in incidence (IRR = 0.04 and 0.08, respectively; P < 0.001; Table 4), whereas others, including Mehrestan and Rask, showed higher incidence relative to Chabahar, likely reflecting residual transmission and timing of interventions. Counties without statistically significant differences remain epidemiologically important due to their high malaria burden.
Impact of cumulative IRS population coverage on malaria incidence in Sistan and Baluchestan, Iran (2013–2023) – negative binomial GLM

* Significant (P < 0.001).
** Non-significant (P > 0.05).
The relationship between bed net distribution and malaria incidence was heterogeneous. Positive correlations were observed in several counties, while others showed negative or non-significant associations. Heat map analyses summarizing correlations between IRS coverage, bed net distribution and malaria incidence are presented in the Supplementary file (Supplementary Figures 3 and 4). These findings indicate that, although vector control measures reduce malaria incidence, the magnitude and direction of their effects vary across counties, emphasizing the importance of local context, intervention timing and sustained coverage for achieving optimal impact.
Discussion
Malaria incidence in eight counties of Sistan and Baluchestan Province declined overall from 2001 to 2023, reaching zero during the elimination phase for three consecutive years. However, a resurgence in 2022–2023, particularly in southern counties, highlights the vulnerability of elimination programs to multiple challenges. Cross-border movements from endemic neighbours, extreme weather events (droughts followed by flooding), COVID-19–related disruptions, limited funding, insecticide and parasite resistance, and competing public health priorities, such as dengue control, likely contributed to renewed transmission (Alizadeh and Sedaghat, Reference Alizadeh and Sedaghat2024; Khammarnia et al., Reference Khammarnia, Peyvand, Barfar, Setoodehzadeh, Kargar and Rokni2025). The observed north–south gradient and localized hotspots emphasize the need for sustained surveillance, targeted interventions and cross-border collaboration to prevent further setbacks.
Malaria transmission in Sistan and Baluchestan Province is associated with climatic factors, indicating its sensitivity to variations in temperature, humidity and other weather conditions (Mohammadkhani et al., Reference Mohammadkhani, Khanjani, Bakhtiari, Tabatabai and Sheikhzadeh2019). Our analysis of eight counties from 2001 to 2023 showed that temperature and relative humidity were the most consistent climatic determinants of malaria incidence, whereas precipitation effects were generally heterogeneous.
In counties with significant associations, higher temperatures within the optimal range for mosquito development accelerated larval growth, biting frequency and parasite development, consistent with findings from Africa, Asia and southern Iran (Eikenberry and Gumel, Reference Eikenberry and Gumel2018; Abbasi et al., Reference Abbasi, Foroushani, Jafari-Koshki, Pakdad, Vatandoost and Hanafi-Bojd2020; Liu et al., Reference Liu, Wang, Deng, Yan, Qin, Du, Liu and Liu2024). Relative humidity was also related to transmission, with positive associations observed in most counties, although Saravan exhibited a negative correlation, likely reflecting the local dominance of Anopheles culicifacies adapted to semi-arid conditions (Salahi-Moghadam et al., Reference Salahi-Moghadam, Khoshdel, Barati and Sedaghat2014; Sahu et al., Reference Sahu, Gunasekaran, Krishnamoorthy, Vanamail, Mathivanan, Manonmani and Jambulingam2017). In contrast, Zabol showed no significant climatic associations, possibly due to very low incidence, drought and strong seasonal winds limiting mosquito populations, highlighting the need for further research in regions with limited transmission.
Seasonal patterns further modulated malaria dynamics, with the highest overall incidence observed in summer and the lowest in winter. Malaria incidence reached its peak during summer in Chabahar and Saravan, whereas in Khash, Iranshahr, Zahedan, Nikshahr and Sarbaz, the highest incidence was observed in autumn. Winter consistently exhibited the lowest transmission across all counties, emphasizing the inhibitory effect of colder temperatures on vector activity. These findings are consistent with previous studies demonstrating marked seasonal increases in malaria transmission during warmer months or following rainy periods (Nigussie et al., Reference Nigussie, Zewotir and Muluneh2023; Mariën et al., Reference Mariën, Mukomena, Tevuzula, Leirs and Huyse2024; Khazaee-Pool et al., Reference Khazaee-Pool, Moosazadeh, Asadi-Aliabadi, Yazdani and Ponnet2025). Such seasonal variability underscores the importance of accounting for both temporal and ecological contexts in malaria surveillance and intervention planning.
IRS coverage was inversely associated with malaria incidence in several high-burden counties. Although this pattern aligns with the expected protective role of IRS, the ecological nature of the study precludes causal inference. In elimination settings, spraying activities are frequently intensified in response to rising case counts; thus, IRS coverage may partly reflect programmatic responsiveness rather than independent intervention effects, introducing the possibility of reverse causality. Interpreting the impact of the IRS on malaria reduction is complex.
Populations of Anopheles culicifacies and Anopheles stephensi in southeastern Iran exhibit resistance to multiple classes of insecticides, which may reduce the operational effectiveness of IRS and weaken the observed association between coverage and incidence (Gorouhi et al., Reference Gorouhi, Vatandoost, Oshaghi, Raeisi, Enayati, Mirhendi, Hanafi-Bojd, Abai, Salim-Abadi and Rafi2016; Vatandoost et al., Reference Vatandoost, Hanafi-Bojd, Raeisi, Abai and Nikpour2017; Abbasi et al., Reference Abbasi, Hanafi-Bojd, Yaghoobi-Ershadi, Vatandoost, Oshaghi, Hazratian, Sedaghat, Fekri, Safari, Mojahedi and Salari2019). Additionally, the timing of spraying relative to vector activity, such as the Growing Degree Days–based threshold for the seasonal peak of An. stephensi plays a critical role in intervention success (Abbasi et al., Reference Abbasi, Oshaghi, Sedaghat, Hazratian, Rahimi Foroushani, Jafari-Koshki, Yaghoobi-Ershadi, Abai, Vatandoost, Fekri Jaski, Bozorg Omid and Hanafi-Bojd2023). Accordingly, IRS coverage and bed net distribution in this study should be interpreted as ecological and programmatic indicators rather than definitive measures of intervention effectiveness. The observed relationships must be understood within a complex transmission system involving climate variability, vector ecology, insecticide resistance and operational factors, emphasizing the need to consider these interacting components when interpreting malaria dynamics.
This study has several limitations. First, temporal and spatial misalignment existed between datasets: climate and malaria incidence were available monthly over 2001–2023, whereas IRS data were only available for 2013–2023, were annual, incomplete and limited to selected counties. IRS was conducted only in March and September, and detailed information on coverage quality was not consistently available, limiting dose–response assessment. Population denominators were harmonized with county boundaries and missing values interpolated using census growth rates; however, population movement and cross-border migration were not explicitly included. Second, the ecological design precludes individual-level inference, and unmeasured confounders such as vector density and microclimate variation may have influenced observed patterns. Third, analytical challenges, including overdispersion, excess zeros, autocorrelation and multiple testing across counties, may affect model stability. Finally, due to incomplete bed net data, only Spearman correlation with heat map visualization was applied, and formal regression modelling was not feasible.
Conclusions
This 23-year analysis demonstrates associations between malaria transmission and temperature as well as seasonal patterns in Sistan and Baluchestan Province. Higher incidence was consistently observed during warmer seasons, indicating the importance of climatic variability in shaping transmission dynamics. The positive association between IRS coverage and malaria incidence likely reflects increased spraying activities in response to rising case numbers rather than a direct causal effect, as causal relationships cannot be established in an observational study. Overall, these findings support the value of incorporating climatic information into malaria surveillance and planning. Future research should focus on improving predictive models, examining delayed (lagged) effects more closely, and considering factors such as population movement and cross-border mobility to better understand transmission patterns.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0031182026102066.
Acknowledgements
We would like to thank the Center for Communicable Disease Management of the Ministry of Health, and the Health Vice-Departments of Zahedan, Iranshahr, and Chabahar Universities of Medical Sciences for their cooperation in providing malaria data. Grammarly’s artificial intelligence tools were used to edit the article, and then the article was reviewed by all authors.
Author contributions
A.D. collected data, performed analyses, wrote initial draft of article. A.R.F. has provided statistical consulting to conduct analyses. M.R. has provided advice and guidance on climate data. J.N. has collaborated in collecting malaria data in the study area. F.Y. has collaborated in collecting weather and climate data. F.N. and A.R. have made the necessary coordination at the level of the National Center for Disease Control to provide malaria data. A.A.H.B., as the project manager and presenter of the research idea, supervised the various stages of the work. All authors contributed to writing the article and editing the initial draft.
Financial support
This study was financially supported by the Deputy of Research, Tehran University of Medical Sciences, project Number 40111260001.
Competing interest
The authors declare that they have no conflicts of interest.
Ethical standards
This project has been approved by the Ethics Committee of the Vice Chancellor for Research of Tehran University of Medical Sciences with code IR.TUMS.SPH.REC.1403.076.