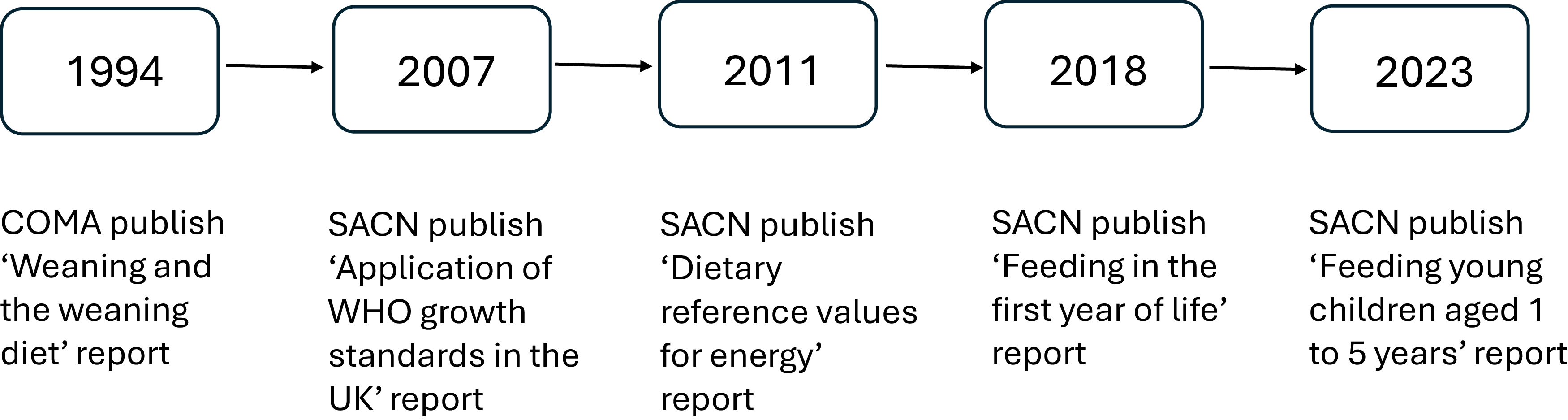
Between 1974 and 1994, the Committee on Medical Aspects of Food and Nutrition Policy (COMA) published a series of reports on infant feeding practices in the UK and made recommendations for infant and young child feeding (Figure 1). The last of these reports, ‘Weaning and the weaning diet,(1)’ was published in 1994 and underpins many current UK government dietary recommendations on feeding young children, including vitamin supplementation.
Timeline of key publications informing young child feeding recommendations in the UK. Abbreviations: COMA (Committee on Medical Aspects of Food Policy); SACN (Scientific Advisory Committee on Nutrition).

Subsequent recommendations made by the Scientific Advisory Committee on Nutrition (SACN) and by international expert committees have had implications for current infant and young child feeding policy. These include the adoption of WHO Growth Standards(2–4) and revisions to energy requirements.(5)
Accordingly, in 2009, SACN requested its Subgroup on Maternal and Child Nutrition (SMCN) to review recent developments in this area. To complement this work, the Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment (COT) was asked by the Department of Health to conduct a review of the risks of toxicity from chemicals in the diets of infants and young children. COT was also asked to examine the evidence relating to the influence of the infant diet on the development of allergic and autoimmune disease, which resulted in a joint SACN-COT statement on the introduction of peanut and hen’s egg into the infant diet.(6)
SACN’s review of the scientific basis of dietary recommendations for infants and young children aged 1 to 5 years was published in 2 reports: ‘Feeding in the first year of life’ (2018),(7) which focused on children aged under 1 year, and ‘Feeding young children aged 1 to 5 years’ (2023).(8)
Methods underpinning the SACN report ‘Feeding young children aged 1 to 5 years’
SACN’s remit is to assess the risks and benefits of nutrients, dietary patterns, food or food components to health by evaluating scientific evidence and to make dietary recommendations for the UK based on its assessment.(9)
SACN’s ‘Framework for the Evaluation of Evidence (10)’ was used as the basis for assessing evidence that informed ‘Feeding young children aged 1 to 5 years.(8)’
The report considered evidence from systematic reviews (SR) and meta-analyses (MA), primarily of randomised controlled trials, non-randomised studies of interventions and prospective cohort studies. The report was also informed by data and analyses of food and drink consumption and nutrient intakes and status in young children living in the UK.
Public Health England conducted a search of key online databases: MEDLINE, Embase, the Cochrane Library, Food Science Technology Abstracts and Scopus to identify relevant English-language articles published between 1990 and 13 November 2018, using an agreed list of search terms focusing on diet and nutrition and health in children aged 1 to 5 years. There was particular interest in the following outcomes: body composition, growth, child obesity, child allergy and autoimmune disease, child and adult bone or skeletal health, child and adult cancers, adult CHD and diabetes. A supplementary online database search focusing on oral health was conducted in October 2019.
SACN invited interested parties to highlight relevant evidence that satisfied the inclusion criteria for the report through a call for evidence from 11 March 2019 to 5 April 2019. The draft report was then made available for public consultation from 20 July 2022 to 20 September 2022, and interested parties were invited to alert SACN to any evidence it may have missed.
The quality of included SR and MA was assessed by the approach outlined in the SACN Framework for the Evaluation of Evidence(9,10) and the AMSTAR 2 tool.(11) Expert judgement, based on pre-specified criteria, was used to grade the strength of the evidence (adequate, moderate, limited, inconsistent or insufficient) for all exposure–outcome relationships of interest.(8) Only exposure–outcome relationships where the evidence base was graded as adequate or moderate were used to inform SACN’s recommendations.
Data from two UK dietary surveys were considered: the 2011‘Diet and Nutrition Survey of Infants and Young Children’ (DNSIYC)(Reference Lennox, Sommerville and Ong12) for children aged 12 to 18 months and the National Diet and Nutrition Survey (NDNS), mainly from survey years 2016 to 2019,(13) for children aged 18 to 60 months. Of the micronutrients examined, the report focussed on Fe, Zn, vitamin A and vitamin D because there was evidence that more than 5% of young children may be at risk of inadequate intakes of these micronutrients. Consideration was also given to vitamin C intakes in young children because it is recommended that all children aged 6 months to 5 years are given vitamin supplements containing vitamins A, C and D.(14) Supplements containing these vitamins are provided through the Healthy Start scheme in England, Wales and Northern Ireland(15) and through the Best Start Foods scheme in Scotland.(16)
Findings of the report were contextualised with the latest available data on the prevalence of children living with overweight and obesity entering primary school (aged 4 to 5 years) from national child measurement programmes in England,(17) Scotland(18) and Wales(19) (no comparable data was available for Northern Ireland); and the prevalence of dental caries in young children in the UK.(20–24)
Systematic review evidence identified in the SACN report ‘Feeding young children aged 1 to 5 years’
Fifty-eight SR (22 with MA) were included in the SACN report(8) (Figure 2). Health outcomes on which the most SR evidence was identified were growth, body composition or weight status, followed by oral health, neurodevelopment or cognitive development, child bone or skeletal health and childhood morbidities (including respiratory diseases). No evidence from SR which met the inclusion criteria was identified on paediatric cancer (such as childhood leukaemia), child allergic and autoimmune disease, adult neurological health and adult bone or skeletal health.
Flow diagram showing the number of publications assessed for eligibility and included in the evidence review(8).

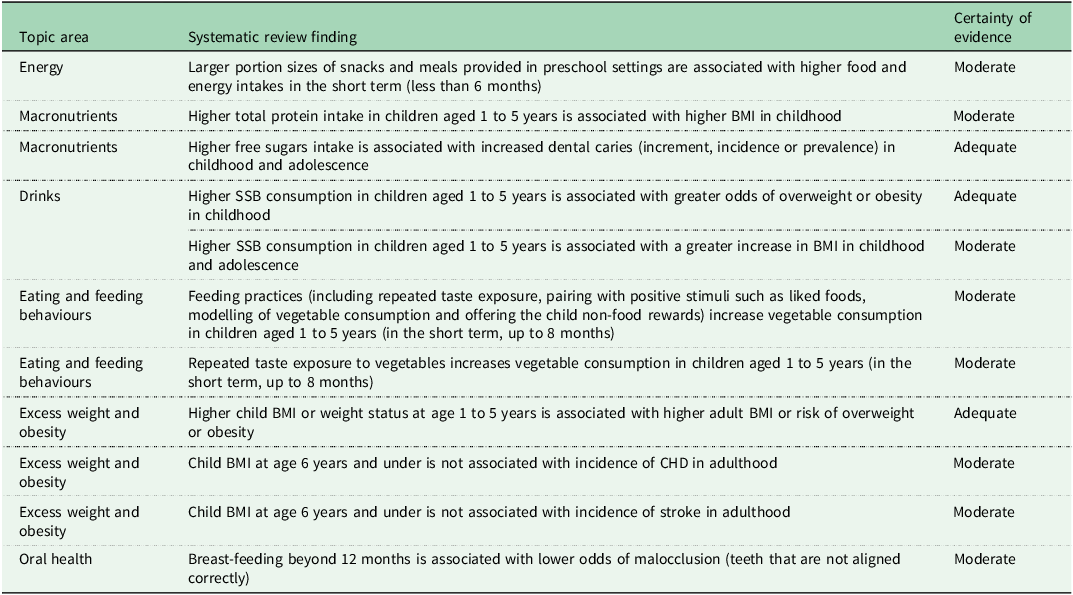
Following grading of the certainty of evidence, there was limited or insufficient SR evidence for the majority of exposure–outcome relationships of interest.(8) Only 11 exposure–outcome relationships had evidence that could be graded at higher levels of certainty (adequate or moderate) (Table 1). The highest certainty evidence (graded adequate) was for the relationship between: higher free sugars intake and increased development of dental caries; higher sugar-sweetened beverage consumption and higher weight status; and the longitudinal tracking of child BMI on adult BMI.
Systematic review evidence in children aged 1 to 5 years graded ‘adequate’ and ‘moderate’(8)

SSB, sugar-sweetened beverage.
Evidence from UK dietary surveys that informed the SACN report
Energy and macronutrients
Data from DNSIYC(Reference Lennox, Sommerville and Ong12) and the NDNS(13) indicated that intakes of energy, free sugars and protein in children aged 1 to 5 years in the UK exceeded dietary recommendations,(25) while dietary fibre intakes were below the recommended level for this age group.
Ninety per cent of children aged 1 to 2 years and seventy per cent of children aged 2 to 3 years had reported total dietary energy intakes (TDEI) above the Estimated Average Requirement(5) for these age groups.
Mean intake of free sugars in children aged 1 to 5 years was double the maximum recommendation of 5% TDEI, which, at the time of the publication of the SACN report,(8) applied from age 2 years.(25,26) More than 80% of children aged 1 to 2 years had intakes above the 5% recommendation, while 97% of children aged 4 to 5 years had intakes above the recommendation.(Reference Lennox, Sommerville and Ong12,13)
Between 70 and 88% of children aged 1.5 years and 5 years had dietary fibre intakes below the dietary reference value of 15 grams per d, which applies from ages 2 years to 5 years.(25)
Mean protein intakes were more than twice the Reference Nutrient Intake (RNI)(27) in children aged 1 to 1.5 years and children aged 4 to 5 years and close to three times the RNI in children aged 1.5 to 4 years.(Reference Lennox, Sommerville and Ong12,13)
The dietary reference value for saturated fat is that the population average intake should be no more than 10% TDEI.(28) Although this recommendation does not apply before age 2 years, and at the time of publication of the report only applied in full from age 5 years,(25,29) more than 90% of children aged 1 to 5 years had saturated intakes above the dietary reference value.
Micronutrients
Data from the NDNS(13) indicated that 70 per cent of children aged 1.5 to 4 years had salt intakes above the target average salt intake for that age group.(30)
Although NDNS data(13) indicated that mean intakes of Fe, Zn, vitamin A and vitamin C were above the RNI for these micronutrients(27) in almost all age groups, between 8 and 11% of children aged 1.5 to 4 years had intakes below the Lower Reference Nutrient Intake (LRNI) for Fe, Zn and vitamin A(27) and 20% of children aged 4 to 5 years had intakes below the LRNI for Zn.(27) Secondary analysis of the NDNS data(13) undertaken for the SACN report(8) indicated that inadequate intakes of Fe, Zn, vitamin A and vitamin D may be more prevalent among children from lower socio-economic status households and certain ethnic groups. SACN concluded in its report(8) that, although the data should be interpreted with caution given that misreporting of energy intake in self-reported dietary methods is a well-documented source of bias,(Reference Poslusna, Ruprich and de Vries31) the extent to which energy underreporting affects the assessment of micronutrient intakes is unclear.
Current UK recommendations are that children aged 1 to 5 years should be given a daily supplement containing vitamins A, D and C.(14) NDNS data(13) indicated that only 25% of children aged 1.5 to 3 years were given a vitamin D supplement (data were not available for the other age groups), while children with vitamin A intakes below the LRNI did not obtain any vitamin A from dietary supplements.(8) In contrast, none of the participants at any age between 1 and 5 years had vitamin C intakes below the LRNI, regardless of whether they were given a supplement containing vitamin C.(8,13)
Foods
There are currently no UK government recommendations on portion sizes for vegetables and fruit for young children. Data from DNSIYC(Reference Lennox, Sommerville and Ong12) and NDNS(13) indicated that children aged 1 to 5 years consumed more fruit than vegetables, that there was a significant reduction in vegetable consumption over a 9-year period (NDNS data collection years 2008 to 2017(32)) and that consumption of vegetables and fruit decreased with increasing deprivation as assessed by the Index of Multiple Deprivation for England.(8,33)
Children aged 1 to 1.5 years obtained approximately 16% TDEI from foods that are energy dense and high in saturated fat, salt or free sugars,(8,Reference Lennox, Sommerville and Ong12) while children aged 1.5 to 5 years obtained between a quarter and nearly a third of TDEI from these foods.(8,13)
Sixty-five per cent of children aged 1 to 1.5 years consumed commercially manufactured foods and drinks marketed specifically for infants and young children and obtained approximately 20% of free sugars intakes from these products.(Reference Lennox, Sommerville and Ong12,34) However, these figures from the 2011 DNSIYC(Reference Lennox, Sommerville and Ong12) are likely underestimates given evidence of substantial growth in the market for these products in recent years.(34)
Drinks
Data from DNSIYC(Reference Lennox, Sommerville and Ong12) and NDNS(13) indicated that formula milks (mainly follow-on formula and milks marketed for children aged over 1 year) were consumed by 36% of children aged 1 to 1.5 years contrary to the current UK recommendation that formula milks are not needed beyond age 1 year.(35) Children who consumed formula milks obtained 50% of free sugars intakes from these products.(13)
Substitution analysis of DNSIYC data(Reference Lennox, Sommerville and Ong12) undertaken for the SACN report(8) indicated that replacing whole cows’ milk with semi-skimmed cows’ milk for children aged 1 to 1.5 years would be unlikely to have a detrimental effect on nutrient intakes at the population level. By contrast, replacing whole milk with skimmed or 1% milk may result in a greater risk of inadequate intakes of vitamin A in young children.(8)
Twenty-six per cent of children aged 1 to 1.5 years(Reference Lennox, Sommerville and Ong12) and 40% of children aged 1.5 to 5 years(13) consumed fruit juice (100% fruit juice and fruit-based non-dairy smoothies). Fruit juice contributed 11% to free sugars intakes in children aged 1.5 to 4 years.(13)
Twenty-six per cent of children aged 1 to 1.5 years(Reference Lennox, Sommerville and Ong12) and over 20% of children aged 1.5 to 5 years(13) consumed sugar-sweetened beverages (including squashes, cordials, carbonated energy drinks). These drinks provided approximately 3 to 4% to free sugars intakes in children aged 1 to 5 years.(Reference Lennox, Sommerville and Ong12,13)
Nutritional and toxicological aspects associated with the consumption of plant-based drinks by the UK population, including children aged 1 to 5 years, were considered in a separate benefit-risk assessment jointly conducted by SACN and COT. Findings and recommendations from this assessment were published in 2025.(36)
Limitations of the evidence base that informed the SACN report
SACN identified a range of limitations in the evidence base that informed its report ‘Feeding young children aged 1 to 5 years.(8)’
There was either no or insufficient SR evidence for a number of dietary exposures (including saturated fat and dietary fibre) and health outcomes which were included in the scope and literature search for the report.(8)
Many of the SR identified for the SACN report(8) had a broad search strategy that included population groups outside the age range of interest for this report (children aged 1 to 5 years) and it was difficult to determine whether their search strategy for the target population was comprehensive.(8)
The evidence base on most topic areas was mainly observational, short term and highly heterogeneous (exposures, dietary assessment methods, outcome measures, populations, settings and study designs).(8) The heterogeneous evidence base prevented the pooling of results by MA or other methods of quantitative synthesis. The lack of quantitative synthesis meant that the risk of publication bias was seldom formally assessed by SR authors.(8)
The SR evidence identified on micronutrients was drawn almost exclusively from supplementation and food fortification trials designed for populations in low-income, lower-middle or upper-middle income countries(37) and therefore may not be generalisable to children living in the UK.(8) The impact of dietary or nutritional exposures on nutritional status or health outcomes among different ethnic groups was seldom considered.(8)
There were also several limitations of the evidence from dietary surveys. DNSIYC(Reference Lennox, Sommerville and Ong12) was conducted in 2011. Dietary patterns may have changed significantly in the period since the data were collected.
While the NDNS(13) is designed to be representative of the UK population, the sample of children that provided blood samples for status measures was much smaller. Children aged 1.5 to 5 years who gave a blood sample were also more likely to come from higher socio-economic status households.(8)
Conclusions of the SACN report
SACN’s report ‘Feeding young children aged 1 to 5 years (8)’ considered the scientific basis of current UK recommendations for feeding young children aged 1 to 5 years. It showed that the current diet of young children in the UK does not meet current dietary recommendations for several nutrients. This is of concern given the high prevalence of children living with overweight and obesity in the UK particularly in lower socio-economic groups and in some ethnic groups and the high prevalence of dental caries in young children in the UK.
Recommendations from the SACN report
The SACN report(8) makes several recommendations that are suitable for children aged 1 to 5 years who are able to consume a varied diet and are growing appropriately for their age. These include:
-
• Between 1 and 2 years of age, children’s diets should continue to be gradually diversified in relation to foods, dietary flavours and textures. A flexible approach is recommended to the timing and extent of dietary diversification, taking into account the variability between young children in developmental attainment and the need to satisfy their individual nutritional requirements.
-
• Current UK dietary recommendations as depicted in the Eatwell Guide(38) should apply from around age 2 years, with some exceptions including:
-
○ UK dietary recommendations on average intake of free sugars (that free sugars intake should not exceed 5% of TDEI) should apply from age 1 year;
-
○ milk or water, in addition to breast milk, should constitute the majority of drinks given to children aged 1 to 5 years;
-
○ pasteurised whole and semi-skimmed cows’ milk can be given as a main drink from age 1 year, as can goats’ and sheep’s milks;
-
○ children aged 1 to 5 years should not be given sugar-sweetened beverages.
-
-
• Foods (including snacks) that are energy dense and high in saturated fat, salt or free sugars should be limited in children aged 1 to 5 years in line with current UK dietary recommendations.
-
• Commercially manufactured foods and drinks marketed specifically for infants and young children are not needed to meet nutritional requirements.
-
• Children aged 1 to 5 years should be presented with unfamiliar vegetables on multiple occasions (as many as 8 to 10 times or more for each vegetable) to help develop and support their regular consumption.
The SACN report(8) also endorses a number of existing SACN and COMA recommendations including on salt,(30) introduction of allergenic foods,(6) vitamins A and D.(14) In addition, it recommends several actions for the UK government to support children aged 1 to 5 years to eat a healthy diet.
The report concludes with a range of research recommendations derived from gaps in the evidence identified during its development.
Acknowledgements
The SACN report was developed by SACN and its Subgroup on Maternal and Child Nutrition (SMCN). A list of SACN and SMCN members is available here: Scientific Advisory Committee on Nutrition (SACN) - GOV.UK (www.gov.uk).
SACN is an independent committee of the Office for Health Improvement and Disparities (OHID) within the Department of Health and Social Care (DHSC).
Author contributions
E. H. prepared the first draft of the manuscript. R. E. and A. C. commented on the first draft of the manuscript. All authors commented on subsequent drafts and approved the final manuscript.
Financial support
Work on the report commenced while the authors were employed by Public Health England. The report was funded by Public Health England and DHSC.
Competing interests
EH, RE and AC are employed by DHSC. They are members of the Association for Nutrition. AC is also a member of the Nutrition Society Advisory Council. Declared interests of SACN and SMCN members are available here Scientific Advisory Committee on Nutrition (SACN) - GOV.UK (www.gov.uk).