The Fetal Gonads
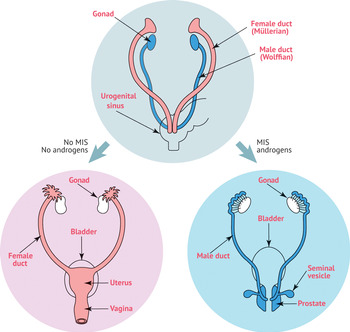
The female and male reproductive tracts originate from the same embryonic/fetal tissue. The gonads and internal and external genitalia begin as bipotential tissues. The indifferent gonad consists of a medulla and cortex. Human female and male embryos develop in the same way for the first 6 weeks, regardless of genetic sex (46,XX or 46,XY karyotype) (Figure 1.1). The one way to tell the difference between 46,XX and 46,XY embryos during this time period is by looking for a Barr body (“inactive” one X chromosome) or a Y chromosome. The medulla of the XY embryo will develop into the testes and the cortex will regress. In the XX embryo, the ovary will originate from the cortex and the medulla will decline. A complete 46,XX chromosomal complement is necessary for normal ovarian development. The second X chromosome contains elements essential for ovarian development.
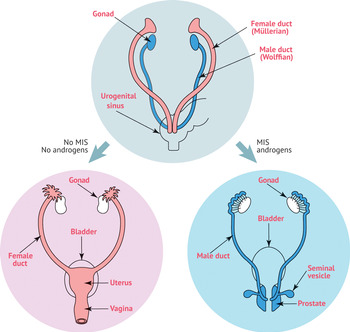
Figure 1.1 Human male and female embryos develop similarly for the first 6 weeks, regardless of genetic sex. MIS, Müllerian-inhibiting substance.
The Fetal Ovary
The development of the human ovary during fetal life can be divided into five stages:
1. Indifferent gonad stage
2. Stage of differentiation
3. Period of oogonia formation (mitosis and migration)
4. Period of oocyte formation (meiosis and differentiation)
5. Stage of follicle formation (follicle assembly)
The gonads begin with development from the mesothelial layer of the peritoneum. The ovary differentiates into a central part – the medulla, which is covered by a surface layer, called the germinal epithelium. At approximately 4–5 weeks of gestation, the paired gonads structurally form the gonadal ridges [Reference Eddy, Clark, Gong and Fenderson1]. The immature ova originate from 50 to 80 germ cells of the dorsal endoderm of the yolk sac (Figure 1.2). These progenital or primordial germ cells (PGCs) multiply by mitosis, to yield approximately 30 000 at migration. By the time they reach the gonadal ridge (between 5 and 6 weeks of gestation), they are called oogonia (diploid stem cells of the ovary with underdeveloped endoplasmic reticulum and differentiated nucleus) [Reference Konishi, Fujii, Okamura, Parmley and Mori2–Reference Francavilla, Cordeschi and Properzi3]. The factors that initiate and direct the migration of the germ cells are not known. Migrating PGCs still express core pluripotency genes such as SOX2, OCT4, and NANOG that are characteristic for early embryonic stem cells [Reference Ulloa-Montoya, Kidder and Pauwelyn4]. After migration, PGCs begin to express Mvh [Reference Eguizabal, Shovlin, Durcova-Hills, Surani and McLaren5], which marks the end of their migration and the beginning of sexual dimorphic development in the undifferentiated gonadal ridge and thus their development into primary oogonia. DAX1 is a gene typically expressed in both testicular and ovarian tissues (a short arm of the X chromosome). DAX1 downregulates the effectiveness of the male sex reversal Y gene (SRY) or its downstream elements, resulting in an ovary.

Figure 1.2 Human embryonic primordial germ cell (PGC) migration.
At approximately week 6–7 of development, in the absence of anti-Müllerian hormone (AMH) (from Sertoli cells), the Müllerian ducts develop into the female internal genitalia. The development of female internal and external structures is gonad independent.
The source of the gonadal somatic cells is still uncertain. Besides germ cells, the earliest recognizable gonad contains somatic cells derived from at least three different tissues: coelomic epithelium, mesenchyme, and mesonephric tissue. Ultrastructural studies have even suggested that both the coelomic epithelial and underlying mesonephric cells offer the somatic cells that are intended to become follicular cells. The germ cells are first identifiable at the end of the third week after fertilization and can be found in the primitive endoderm at the caudal end in the dorsal wall of the adjacent yolk sac. At 6–8 weeks, the first signs of ovarian differentiation are reflected by the rapid mitotic multiplication of germ cells, reaching 6–7 million oogonia by weeks 16–20 (Figure 1.3). Oogonia go into prophase of the first meiotic division and form clusters (oogonia surrounded by a single layer of flattened follicular cells) called primordial follicles.

Figure 1.3 Female gestational development of oocytes. GV, germinal vesicle.
Around gestation week 10, the primary oocytes, which are arrested at the diplotene stage in prophase of the first meiotic division, cluster together in germ cell nests, in a structure known as ovarian cords, which can be found either in the developing ovary medulla or in the cortex. Around gestation week 20, the germ cell nests of the medulla of the ovary break down. While the exact mechanisms involved in germ cell nest breakdown are unknown, it is associated with a wave of oocyte apoptosis, ultimately resulting in the establishment of primordial follicles. Oogonia become fully surrounded by a layer of coelomic epithelial cells (pre-granulosa cells, derived from both the peritoneum and mesonephros), and form the rudiments of the ovarian follicles. Interstitial theca cells originate from two sources: coelomic epithelium and mesonephros.
A pituitary follicle-stimulating hormone peak can be observed at 20–23 weeks, and circulating levels peak at 28 weeks. The ovary begins to express gonadotropin receptors in the second half of pregnancy. The loss of oocytes during fetal life cannot be solely explained by the decline in gonadotropins. During meiosis progression and follicle formation, approximately 70% of germ cells are eliminated [Reference Baker6]. The reason underlying constitutive germ cell death remains poorly understood, but has been suggested to ensure the elimination of germ cells exhibiting defective nuclear or mitochondrial genomes. The constitutive elimination of germ cells during ovarian differentiation may, thus, be a critical process, which could, intriguingly, favor reproductive success. Tight control of the balance between germ cell survival and death is, however, critical in preventing excessive germ cell death leading to premature ovarian failure.
The Fetal Testicle
Human embryos, as mentioned, develop similarly for the first 6 weeks, regardless of genetic sex. The testes-determining factor, a product of a single gene located on the Y chromosome, within a region of SRY, is the factor that determines whether the indifferent gonad will become a testis. This gene is expressed in the Sertoli cells [Reference Li, Zheng and Lau7] and its expression results in a cascade of events leading to the development of the seminiferous tubules. As the seminiferous tubules form, the PGCs enter the gonad and associate with the tubules. Through the rete testis, the seminiferous tubules become connected with outgrowths from the mesonephros, which form the efferent ducts of the testis. In contrast to the female, male PGCs do not start meiotic division and do not differentiate to spermatozoa before puberty.
At approximately the seventh week of development, the embryo Sertoli cells (following up SRY expression) secrete AMH, also named Müllerian-inhibiting substance, to suppress the development of the Müllerian ducts, leading to their degeneration and stimulating the differentiation of Leydig cells (secreting testosterone a week later, at week 8) from mesenchymal cells. The prostate, seminal vesicles, and bulbourethral glands develop at 10–13 weeks. Although AMH suppresses the development of the Müllerian ducts, mutations in the AMH receptor gene results in the presence of the uterus, Fallopian tubes, and the upper vagina in 46,XY men with normal external virilization.
Androgen secretion increases in conjunction with increasing Leydig cell numbers and Leydig cell hypertrophy, elicited by human chorionic gonadotropin, until a peak is reached at 15–18 weeks of embryo development. At this time, Leydig cell regression begins, and, at birth, only a few Leydig cells are present.
In the presence of testosterone, secreted by the Leydig cells, and functional androgen receptors, encoded by a gene located on the long proximal arm of the X chromosome (locus Xq11-Xq12), the Wolffian ducts develop into the epididymides, vasa deferentia, and seminal vesicles [Reference Hannema and Hughes8]. In the process of masculine differentiation, the development of these Wolffian duct structures is dependent on testosterone as the intracellular mediator, whereas development of the urogenital sinus (scrotum) and urogenital tubercle (penis) into the male external genitalia, urethra, and prostate requires the conversion of testosterone to dihydrotestosterone (DHT). In this manner, the development of the internal and external genitalia in the male is dependent on both testicular testosterone and DHT.
Although the initial testosterone production and sexual differentiation are in response to the fetal levels of adrenocorticotropic hormone (ACTH) and human chorionic gonadotropin (hCG), further testosterone production and masculine differentiation are maintained by the fetal pituitary gonadotropins. If the testes fail to secrete testosterone, or the androgen receptors do not function properly, the Wolffian ducts degenerate.
Rare cases of phenotypic, infertile males with a 46,XX karyotype have been reported; the male differentiation is due to a translocation (meiotic recombination) of a Y chromosome fragment containing SRY to an autosome or an X chromosome in 75% to 90% of sporadic cases. In XY gonads, SRY induces autosomal Sox9, a gene closely related to SRY structurally, and involved in testes differentiation, and tips differentiation toward testis development. Duplication of SOX9 transcription factor may be responsible for some familial cases of XX sex reversal. In XX gonads lacking SRY, DAX1 represses Sox9 and promotes ovary development. Most male genes (Dhh, Sox9, Cbln1) are activated in XY gonads. Many female genes (Irx3, Wnt4, Msx1) are repressed in XY gonads.