Introduction
Tapeworms belonging to the genus Spirometra Faust, Campbell and Kellogg, 1929 (Cestoda: Diphyllobothriidae) are distributed worldwide. The larval plerocercoid (also known as a sparganum) parasitizes tissues of various vertebrates, including humans, and causes 2 types of sparganosis: non-proliferative and proliferative sparganosis. The former is caused by infection by the plerocercoids of (mainly) Spirometra erinaceieuropaei and Spirometra mansoni (e.g. Liu et al., Reference Liu, Li, Wang, Zhao and Zhu2015; Kuchta et al., Reference Kuchta, Phillips and Scholz2024; Yamasaki et al., Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b). These plerocercoids do not multiply in the second intermediate hosts. This contrasts with proliferative sparganosis, caused by plerocercoids of Sparganum proliferum which multiply asexually and metastasise in the second intermediate hosts (Scholz et al., Reference Scholz, Kuchta and Brabec2019; Kuchta et al., Reference Kuchta, Kołodziej-Sobocińska, Brabec, Młocicki, Salamatin and Scholz2021).
The adult worm dwells in the small intestines of carnivores, mainly felines and canines, causing spirometrosis. In rare cases, the plerocercoid can also develop into adult worm in humans (Scholz et al., Reference Scholz, Kuchta and Brabec2019; Kuchta et al., Reference Kuchta, Kołodziej-Sobocińska, Brabec, Młocicki, Salamatin and Scholz2021).
Humans become infected with the parasite by drinking untreated water contaminated by copepods (first intermediate hosts) that harbour the procercoid stage, by consuming raw or undercooked flesh of the second intermediate and paratenic hosts such as frogs, snakes and wild boars, or by using frog meat as poultices on skin wounds (Wiwanitkit, Reference Wiwanitkit2005; Anantaphruti et al., Reference Anantaphruti, Nawa and Vanvanitchai2011; Liu et al., Reference Liu, Li, Wang, Zhao and Zhu2015; Chai et al., Reference Chai, Jung, Ryu, Kim, Hong, Htoon, Tin, Na and Sohn2020). Sparganosis is an important food- and water-borne parasitic infection that is often overlooked and is certainly under-reported.
The first sparganosis case reported in Asia was a Chinese patient who died in Fujian province, China: the causative species was described as Ligula mansoni by Cobbold (Reference Cobbold1883). Subsequently, L. mansoni Cobbold, 1883 was proposed as a junior synonym of Dubium erinacei europaei Rudolphi, 1819 (Rudolphi, Reference Rudolphi1819) and placed within the genus Diphyllobothrium Cobbold, 1858 by some authors (see synonymy in Iwata, Reference Iwata1972). The generic name ‘Dubium’ was at Rudolphi’s time a collective term used for larval parasites of uncertain taxonomic placement. The abbreviated specific name ‘erinacei’ was used by Rudolphi later (Rudolphi, Reference Rudolphi1819) and was often used by later workers instead of ‘erinacei europaei’. Rudolphi’s name was paired with another collective generic term for larval cestodes, Sparganum, by Diesing (Reference Diesing1854), as Sparganum erinacei-europaei. A new subgenus, Spirometra, was proposed within Diphyllobothrium by Faust et al. (Reference Faust, Campbell and Kellogg1929). The name Spirometra erinaceieuropaei was accepted by Bray et al. (Reference Bray, Jones, Andersen, Khalli, Jones and Bray1994). This led to the long-standing assumption that S. erinaceieuropaei is the major etiological agent of sparganosis globally. However, recent molecular studies have revealed that the genuine S. erinaceieuropaei is present only in Europe, and that worms previously identified as S. erinaceieuropaei, S. decipiens (Diesing, 1850) and S. ranarum (Gastaldi, 1854) in Asia and/or Oceania in fact belonged to S. mansoni or S. asiana (Kuchta et al., Reference Kuchta, Kołodziej-Sobocińska, Brabec, Młocicki, Salamatin and Scholz2021, Reference Kuchta, Phillips and Scholz2024; Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b). All cases for which these parasite names were used were non-proliferative.
The first case of proliferative sparganosis was reported in a Japanese patient. The etiological agent was described as Plerocercoides prolifer based on the finding of numerous branched and peculiar-shaped plerocercoids (Ijima, Reference Ijima1905). The adult stage of this species was unknown. Under a provision of the International Code of Zoological Nomenclature (Article 42.2.1), larval parasites of unknown species may be assigned a collective-group generic name based on their larval form. Thus P. prolifer was renamed as Sparganum (Getestius) proliferum (Ijima, Reference Ijima1905) Stiles, 1908 (Stiles, Reference Stiles1908). Since then, it has been usual to regard Spa. proliferum as the species causing proliferative sparganosis (e.g. Moulinier et al., Reference Moulinier, Martinez, Torres, Noya, Noya and Reyes1982; Liu et al., Reference Liu, Li, Wang, Zhao and Zhu2015; Kikuchi and Murayama, Reference Kikuchi and Maruyama2020).
This review focuses on Spirometra infections in Southeast and South Asia and Oceania, and overviews the causative species, endemic areas, current situation in humans, diagnostics and future perspectives. We also discuss the identity of Spa. proliferum and whether the organism reported from Asia under this name is different from Spa. proliferum from Venezuela.
Molecular phylogeny and biogeography
Molecular phylogeny
Morphological identification of Spirometra species is difficult due to morphological similarities among species. Features of adults, such as the shape of uterus and the number of uterine coils, vary depending on fixation methods and the age of individual tapeworms, making them inappropriate for distinguishing species (Iwata, Reference Iwata1972). Despite this, S. erinaceieuropaei, S. decipiens and S. ranarum have all been reported from Asia based on adult morphology. Not surprisingly, these morphological identifications and their geographical distributions have been questioned (Scholz et al., Reference Scholz, Kuchta and Brabec2019).
Recent taxonomic revisions of Spirometra species in Asia have been conducted based on analysis of the complete sequences of the mitochondrial cytochrome c oxidase subunit 1 gene (cox1, 1566 bp) (Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021). One hundred and eight haplotypes were identified in a haplotype network based on this gene using 149 sequences of Spirometra species from Asia. These haplotypes fell into 2 groups: a large group consisting of 106 haplotypes and a small group consisting of 2 haplotypes (Thanchomnang et al., Reference Thanchomnang, Sanpool, Yamasaki, Intapan, Maleewong, Petney, Saijuntha and Mehlhorn2021; Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021). The former group included ‘S. erinaceieuropaei’, originally identified as ‘L. mansoni’ from Fujian and Guangdong provinces in China, as well as ‘S. decipiens’ and ‘S. ranarum’. The molecular data indicate that all members of this large haplogroup should be assigned to S. mansoni (Kuchta et al., Reference Kuchta, Kołodziej-Sobocińska, Brabec, Młocicki, Salamatin and Scholz2021, Reference Yamasaki, Nakamura, Intapan, Maleewong, Morishima, Sugiyama, Matsuoka, Kobayashi, Takayama and Kobayashi2014; Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b). The smaller haplogroup included only the species recently described as Spirometra asiana Yamasaki, Sugiyama & Morishima, 2024 and mistakenly reported as S. erinaceieuropaei from South Korea.
These results have also been supported by a phylogenetic analysis. Worms from Asia referred to by various authors as S. erinaceieuropaei, S. decipiens and S. ranarum all belonged to the same monophyletic clade as S. mansoni, and a smaller group consisted of S. asiana (Figure 1 and Supplementary Figure 1). The cox1 sequences of 50 S. asiana specimens from Japan were all identical, and only 8 out of 1566 bases differed from those of specimens from South Korea. This is in contrast with the higher level of genetic diversity seen in S. mansoni (Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021). Spirometra mansoni and S. asiana are phylogenetically closely related, but distinct species, and are neither S. erinaceieuropaei native to Europe, S. theileri from Africa, S. decipiens complex 1, Spirometra sp. 2 and Spirometra sp. 3 from North and South America nor Spa. proliferum from South America (Figure 1 and Supplementary Figure 1).

Figure 1. Maximum likelihood tree inferred from complete cox1 sequences of Spirometra species and related taxa. HKY + G + I was used as the best substitution model. The species name used for each sequence in the tree is the name assigned to it in the relevant GenBank accession number. The Russian species (registered as S. erinaceieuropaei) included in the tree, PP002117, is from Astrakhan at the northern end of the Caspian Sea and therefore technically belongs in Europe, very close to the accepted boundary with Asia. The etiological agents from cases of proliferative sparganosis in mammals are indicated with stars. Spirometra decipiens complex 1, Spirometra sp. 2 and Spa. proliferum from Venezuela have been tentatively assigned to Spirometra sp. 2 (Kuchta et al., Reference Kuchta, Phillips and Scholz2024). Bootstrap values (>90% in 1000 replications) are shown at each node. The scale bar indicates the number of nucleotide substitutions/site. Dibothriocephalus nihonkaiensis was used as an outgroup.
What is Spa. proliferum?
Several questions have remained about Spa. proliferum, (i) what is its taxonomic position?, (ii) is Spa. proliferum actually present in Asia?, (iii) why has the adult form of Spa. proliferum remained unknown despite extensive research in companion animals and wildlife? and (iv) why does Spa. proliferum proliferate asexually in the intermediate hosts?
Regarding question (i), Ijima (Reference Ijima1905) speculated that the plerocercoids of P. prolifer might turn out to be L. mansoni if followed up to the mature stage.
Iwata (Reference Iwata1972) transplanted plerocercoids incised at head or tail, or diagonally, of Diphyllobothrium erinacei (a synonym of S. erinaceieuropaei but in this case actually referring to S. mansoni) into the subcutaneous tissues and abdominal cavities of rabbits, guinea pigs and a monkey and observed that filamentous and branched plerocercoids regenerated that were histologically comparable to those of Spa. proliferum. He also reported that the numbers of plerocercoids recovered from the animals were less than the numbers of transplanted plerocercoids, indicating regeneration but not proliferation in the recipient animals. Based on the histological observations that the irregularly branched plerocercoids obtained in regeneration experiments and in the Japanese striped snake (Elaphe quadrivirgata) were comparable to those of the original specimen reported by Ijima (Reference Ijima1905), Iwata and Inatomi (Reference Iwata and Inatomi1972) stated that ‘Spa. proliferum’ from Japan might be aberrant plerocercoids of S. mansoni that had regenerated.
As shown in Figure 1, Spa. proliferum from Venezuela, along with S. decipiens complex 1 from Chile and Spirometra sp. 2 from the USA, clustered with Spirometra sp. 2 and were all tentatively assigned to Spirometra sp. 2 by Kuchta et al. (Reference Kuchta, Phillips and Scholz2024). Thus, Spa. proliferum from Venezuela is phylogenetically close to Spirometra species from North and South America and distantly related to S. mansoni and S. asiana (Kuchta et al., Reference Kuchta, Kołodziej-Sobocińska, Brabec, Młocicki, Salamatin and Scholz2021, Reference Kuchta, Phillips and Scholz2024; Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b; Fredes et al., Reference Fredes, Mercado, Salas, Sugiyama, Kobayashi and Yamasaki2022). Although nomenclatural changes are likely soon, we will use the name Spa. proliferum in this review when that name was used in the original reports.
The genetic distance (d value) of cox1 between Spa. proliferum from Venezuela and S. decipiens complex 1 from Chile is 0.0821 (Fredes et al., Reference Fredes, Mercado, Salas, Sugiyama, Kobayashi and Yamasaki2022), which is slightly smaller than the d values between S. mansoni and S. asiana (0.0986–0.1350) and roughly equivalent to the d values between Dibothriocephalus nihonkaiensis and Dibothriocephalus latus (0.0852–0.0869) (Yamasaki et al., Reference Yamasaki, Sato, Kuramochi and Mercado2023). Although the d values alone cannot determine whether a species is conspecific, it can serve as a reference.
Regarding question (ii), it has been reported that Spa. proliferum from Venezuela and Japan are conspecific based on mitochondrial and nuclear gene analyses (Kokaze et al., Reference Kokaze, Miyadera, Kita, Machinami, Noya, Alarcón de Noya, Okamoto, Horii and Kojima1997; Miyadera et al., Reference Miyadera, Kokaze, Kuramochi, Kita, Machinami, Noya, Alarcón de Noya, Okamoto and Kojima2001). However, it is important to note that the Japanese isolate analysed by Kokaze et al. (Reference Kokaze, Miyadera, Kita, Machinami, Noya, Alarcón de Noya, Okamoto, Horii and Kojima1997) and Miyadera et al. (Reference Miyadera, Kokaze, Kuramochi, Kita, Machinami, Noya, Alarcón de Noya, Okamoto and Kojima2001) were not Ijima’s specimens, and there is no DNA data available for them. The Japanese specimen they used was reported in 1909 (Usui, Reference Usui1909) and had been stored in formaldehyde for nearly a century. This raises questions about the quality of DNA in their sample. Some of their cloned cox1 sequences from Japan were identical to those form Venezuela (Miyadera et al., Reference Miyadera, Kokaze, Kuramochi, Kita, Machinami, Noya, Alarcón de Noya, Okamoto and Kojima2001), which raises the suspicion of contamination. Only the DNA data from Venezuela seems reliable (Kokaze et al., Reference Kokaze, Miyadera, Kita, Machinami, Noya, Alarcón de Noya, Okamoto, Horii and Kojima1997; Miyadera et al., Reference Miyadera, Kokaze, Kuramochi, Kita, Machinami, Noya, Alarcón de Noya, Okamoto and Kojima2001; Kikuchi et al., Reference Kikuchi, Dayi, Hunt, Ishiwata, Toyoda, Kounosu, Sun, Maeda, Kondo, Alarcón de Noya, Alarcón de Noya, Kojima, Kuramochi and Maruyama2021). Thus, there is no decisive evidence that the Japanese and Venezuelan specimens are conspecific.
Recently, S. mansoni has been reported as a causative species of proliferative sparganosis in Japan (Tokiwa et al., Reference Tokiwa, Fushimi, Chou, Yoshida, Kinoshita, Hikima, Kikuchi and Ozaki2024), the USA (originally reported as S. mansoni/S. ranarum/S. erinaceieuropaei, Semenova et al., Reference Semenova, Walden, Bollenback, Cook and Ossiboff2025) and Australia (originally reported as S. erinaceieuropaei, Young et al., Reference Young, Malik, Brown, Wang, Ash, Korhonen and Gasser2025). All these cases were from dogs or cat and all the reports included molecular data. This clearly indicates that S. mansoni, not Spa. proliferum, causes proliferative sparganosis in Asia and Oceania, including North America (stars in Figure 1, Supplementary Figure 1 and Table 1). In these cases, S. mansoni may have proliferated asexually, but the possibility of multiple (non-proliferative) infections due to simultaneous or repeated exposures to the plerocercoids of S. mansoni needs to be considered.
Regarding question (iii), the purported adult worms of Spa. proliferum have been found in wild felids in Argentina (Arrabal et al., Reference Arrabal, Pérez, Arce and Kamenetzky2020). However, the identification was not reliable because the cox1 sequences (296 bp) used for identifying the species were short, and further analysis using longer sequences is needed (Fredes et al., Reference Fredes, Mercado, Salas, Sugiyama, Kobayashi and Yamasaki2022; Kuchta et al., Reference Kuchta, Phillips and Scholz2024). If S. mansoni is the causative species of proliferative sparganosis in Asia, Oceania and North America, as outlined in the previous paragraph, then the adult stage of this ‘Spa. proliferum’ is already known to parasitize in dogs, cats and wild animals (Nakamura et al., Reference Nakamura, Hara, Matsuoka, Kawabata and Tsuji1990; Yamasaki et al., Reference Yamasaki, Sugiyama, Morishima and Sako2024c).
Regarding question (iv), some genes that are putatively important for asexual proliferation of Spa. proliferum, such as a variety of tolloid-like protein (peptidases) and oncogene-like genes, have been identified in budding plerocercoids using transcriptome analyses (Kikuchi et al., Reference Kikuchi, Dayi, Hunt, Ishiwata, Toyoda, Kounosu, Sun, Maeda, Kondo, Alarcón de Noya, Alarcón de Noya, Kojima, Kuramochi and Maruyama2021).
Geographical distribution
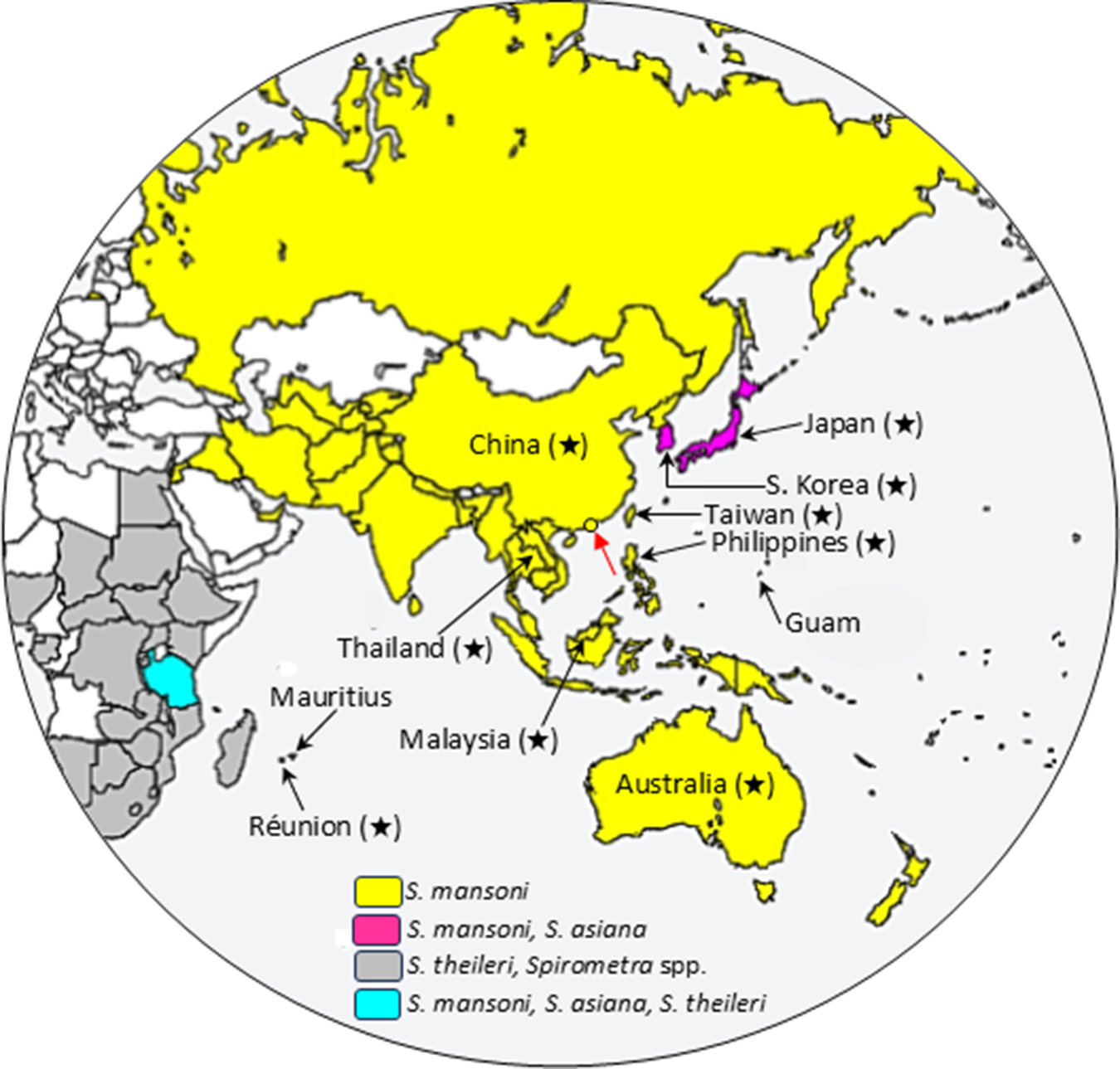
The global distribution of Spirometra species has been reviewed (Kuchta et al., Reference Kuchta, Phillips and Scholz2024). The distribution of Spirometra species in Asia and Oceania, including the eastern part of Africa and Mauritius, is shown in Figure 2, based on many data (Zhu et al., Reference Zhu, Beveridge, Berger, Barton and Gasser2002; Okamoto et al., Reference Okamoto, Iseno, Shibahara, Sato, Wandra, Craig and Ito2007; Yamasaki et al., Reference Yamasaki, Nakaya, Nakao, Sako and Ito2007; Koonmee et al., Reference Koonmee, Intapan, Yamasaki, Sugiyama, Muto, Kuramochi, Kularbkeaw, Kanpittaya, Maleewong and Nawa2011; Boonyasiri et al., Reference Boonyasiri, Cheunsuchon, Srirabheebhat, Yamasaki, Maleewong and Intapan2013, Reference Boonyasiri, Cheunsuchon, Suputtamongkol, Yamasaki, Sanpool, Maleewong and Intapan2014; Hoffdorf et al., Reference Holldorf, Siers, Richmond, Klug and Reed2015; Zhang et al., Reference Zhang, Cui, Liu, Jiang, Wang, Qi, Wu and Wang2015; Badria et al., Reference Badria, Eslahi, Majidiania and Pirestani2017; Le et al., Reference Le, Lqt, Nguyen, Nguyen and Do2017; Sato et al., Reference Sato, Sato, Yoonuan, Pongvongsa, Sanguankiat, Kounnavong, Maipanich, Chigusa, Moji and Waikagul2017; Kuchta et al., Reference Kuchta, Phillips and Scholz2024; Young et al., Reference Young, Malik, Brown, Wang, Ash, Korhonen and Gasser2025). To date, there have been no reports of Spirometra infections from Mongolia.
Although S. mansoni is widely distributed in the Asia–Oceania region, the species has also recently been identified, based on DNA analysis, in Tanzania (originally reported as S. ranarum) (Eom et al., Reference Eom, Park, Lee, Choe, Kang, Bia, Lee, Keyyu, Fyumagwa and Jeon2018), the United States (Sanders et al., Reference Sanders, Sobotyk, Castro, Abdu, Baade, Borst, Dangoudoubiyam, Delcambre, Gruntmeir, Lee, Leutenegger, Lozoya, Murphy, Pulaski, Schaefer, Vatta, Walden, Lejeune and Verocai2025; Semenova et al., Reference Semenova, Walden, Bollenback, Cook and Ossiboff2025), Mexico (Salazar-Grosskelwing et al., Reference Salazar-Grosskelwing, Rodriguez-Vinas, Bolio-González, Romero-Salas, Ramos-Beltrán, Solano-Barquero and Rojas2025), Costa Rica (Alvarado-Hidalgo et al., Reference Alvarado-Hidalgo, Campos-Camacho, Arguedas-Morales, Romero-Vega, Alfaro-Alarcón, Anchia-Ureña, Bass, Berrocal-Ávila, Hagnauer, Olivares, Solano-Barquero, Traube-Rivera, Montenegro-Hidalgo and Rojas2024) and Colombia (Brabec et al., Reference Brabec, Uribe, Chaparro-Gutiérrez and Hermosilla2022). A parasite identified as S. decipiens was reported in Cuba (Morales et al., Reference Morales, Laird-Pérez, Capó, Iglesias, Fonte, Plascencia-Hernández, Calderón, Eom, de Armas and Pérez-Gómez2022). However, the S. decipiens-specific primers used to amplify the target genes were actually based on the sequences of S. mansoni (misidentified as S. decipiens in Korea by Jeon et al., Reference Jeon, Kim, Sohn and Eom2018a), and the amplified PCR products were not sequenced. The Spirometra species from Cuba therefore appears to have been misidentified and is reasonably interpreted as S. mansoni, but further verification is required to confirm the species identity. Thus, S. mansoni is widely distributed in the Northern and Southern Hemispheres. Meanwhile S. asiana appears to be restricted to Japan, South Korea and Tanzania (Figure 2); however, as molecular identification is increasingly performed, it is likely to be found in other regions (Yamasaki et al., Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b).

Figure 2. Distribution of Spirometra spp. in Asia and Oceania, including eastern part of Africa. Countries where Spirometra mansoni is distributed are coloured yellow, countries where S. mansoni and Spirometra asiana co-exist and S. mansoni, S. asiana and Spirometra theileri are distributed are indicated in magenta and turquoise, respectively. Countries where S. theileri and/or Spirometra spp. are distributed are shown in grey. Countries where cases of proliferative sparganosis have been reported are marked with stars after the country name. A red arrow indicates Hong Kong where cases due to S. mansoni have been confirmed.
The identity of putative Spa. proliferum reported from Réunion Island, located east of Madagascar in the Indian Ocean, is uncertain due to the lack of molecular data (Meric et al., Reference Meric, Ilie, Hofman, Rioux-Leclercq, Michot, Haffaf, Nelson, Neafie and Hofman2010).
Morphological and molecular identification
Many researchers have considered S. erinaceieuropaei to be the main causative species of sparganosis. However, the morphology of adults neither of authentic S. erinaceieuropaei nor of S. mansoni, identified based on DNA analysis, was described until very recently (Kuchta et al., Reference Kuchta, Phillips and Scholz2024).
Although the morphological identification of all stages of Spirometra species is very difficult and there are no clear diagnostic criteria for distinguishing the species, morphological differences observed between plerocercoids of S. mansoni and S. asiana are intriguing. The plerocercoids of S. mansoni found in frogs, snakes, wild boars and humans are slender forms, 0.5–1 mm in width (Figure 3A), whereas those of S. asiana found in wild boars (Figure 3B) and humans (Figure 3C,D) are thicker and wider (5–7 mm), giving the impression of being an adult worm at first glance. However, we must point out that the number of plerocercoids identified as S. asiana is still small. It remains to be determined whether the plerocercoids of S. asiana differ in size depending on their hosts (e.g. frogs and snakes) (Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b).

Figure 3. Morphological differences between plerocercoids of Spirometra mansoni and Spirometra asiana. (A) S. mansoni plerocercoid from human (Masuya et al., Reference Masuya, Aizawa, Shirai, Tokumitsu, Sako and Sugano2008); (B) S. asiana from wild boar; (C) and (D) S. asiana from human (Kudo et al., Reference Kudo, Fujioka, Korenaga, Yamasaki, Morishima, Sugiyama, Nakajima and Sano2017). Arrows indicate the scolex. Note remarkable differences between the sizes of plerocercoids (A, B and D are shown at the same scale).
If the surgically excised plerocercoids and adult worms obtained by deworming are available, molecular identification of the causative species is recommended in medical and veterinary institutions. For that, the parasite samples should be fixed in molecular-grade ethanol (>80%) rather than formalin because formalin fixation cross-links and degrades the DNA (Bianchi et al., Reference Bianchi, Navonne and Conti1991), making the tissue solubilization and DNA extraction and PCR amplification more difficult.
Molecular techniques using mitochondrial cox1 sequences have become the most reliable tools for accurately identifying Spirometra species. PCR followed by DNA sequence analysis can be used for histopathological specimens (Yamasaki et al., Reference Yamasaki, Nakaya, Nakao, Sako and Ito2007; Koonmee et al., Reference Koonmee, Intapan, Yamasaki, Sugiyama, Muto, Kuramochi, Kularbkeaw, Kanpittaya, Maleewong and Nawa2011). Related approaches include pyrosequencing (Thanchomnang et al., Reference Thanchomnang, Tantrawatpan, Intapan, Sanpool, Lulitanond, Tourtip, Yamasaki and Maleewong2016), multiplex PCR (Jeon et al., Reference Jeon, Kim, Sohn and Eom2018a), cox1 sequencing (Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b) and restriction fragment length polymorphism (RFLP) (Yamasaki et al., Reference Yamasaki, Morishima and Sugiyama2024a). No molecular method for differentiating S. mansoni, S. asiana and Spa. proliferum using a single-reaction protocol has been established yet. However, S. mansoni (originally reported as S. decipiens) from Asia and Spa. proliferum from South America are distinguishable by pyrosequencing (Thachomnang et al., Reference Thanchomnang, Tantrawatpan, Intapan, Sanpool, Lulitanond, Tourtip, Yamasaki and Maleewong2016).
The easiest molecular method is RFLP. A restriction enzyme Hinc II (or Hpa I) cleavage site exists at position 590 of the cox1 of S. mansoni, whereas this site is absent in S. asiana and other Spirometra species (Yamasaki et al., Reference Yamasaki, Morishima and Sugiyama2024a). Therefore, if only Spirometra species from Asian and Oceanian regions are tested, and if the PCR-amplified cox1 is cleaved with Hinc II, the species can be identified as S. mansoni if it is not cleaved, it is S. asiana. Formalin-fixed specimens and formalin-fixed-paraffin-embedded sections are also available, but they are not recommended for molecular diagnosis due to problems in obtaining good-quality DNA. It is necessary to design primers that can amplify short regions (less than 300 bp) for such cases (Yamasaki et al., Reference Yamasaki, Nakaya, Nakao, Sako and Ito2007; Koonmee et al., Reference Koonmee, Intapan, Yamasaki, Sugiyama, Muto, Kuramochi, Kularbkeaw, Kanpittaya, Maleewong and Nawa2011).
Human sparganosis/spirometrosis cases and serodiagnosis
For human cases, PubMed, Google Scholar and Medical Central Journal (Igaku Chuo Zasshi) Web were searched using the keywords ‘sparganosis’, ‘Spirometra infection’, ‘S. erinaceieuropaei’, ‘S. mansoni’, ‘Spa. proliferum’, ‘proliferative sparganosis’ and ‘spirometrosis’. Cases reported in the most recent reviews from each country have been supplemented with additional information until 2025. Cases of Asians diagnosed outside Asia have also been added.
Non-proliferative sparganosis
In most cases, single plerocercoids are found, but multifocal cases have also been reported. Plerocercoids have been found in various tissues and organs, particularly subcutaneous tissues (chest, abdomen, trunk and thighs), with ocular, visceral and cerebral cases also often reported (Kagei, Reference Kagei2000; Yoshikawa et al., Reference Yoshikawa, Ouji, Nishiofuku, Lshizaka and Nawa2010; Anantaphruti et al., Reference Anantaphruti, Nawa and Vanvanitchai2011; Liu et al., Reference Liu, Li, Wang, Zhao and Zhu2015; Kim et al., Reference Kim, Ahn, Sohn, Nawa and Kong2018; Nawa et al., Reference Nawa, Kokubo-Tanaka and Yamasaki2025). Symptoms vary depending on the parasite’s location: tumour-like mass, cord-like protrusions or creeping eruption in the subcutaneous cases, paralysis and seizures in the cerebral cases and inflammation of the eyelids and conjunctiva in the ocular cases.
More than 2000 human sparganosis cases have been reported worldwide, with most (80%) from China. Numbers of human cases reported in Asia and Oceania are presented in Table 2.
In China, the first case of sparganosis was reported in 1882, with 1359 cases reported between 1949 and 2014 (Liu et al., Reference Liu, Li, Wang, Zhao and Zhu2015). At least 33 additional cases were reported by 2025 (e.g. Yu et al., Reference Yu, Shen, Yuan, Xia, Gao, Jiang and Jiang2016; Du et al., Reference Du, Tao, Ma, Weng, Gong, Lin, Shen, Mo and Cao2018; see Table 2 for others), including a Chinese immigrant diagnosed in Germany (Bennett et al., Reference Bennett, Mok, Gkrania-Klotsas, Tsai, Stanley, Antoun, Coghlan, Harsha, Traini, Ribeiro, Steinbiss, Lucas, Allinson, Price, Santarius, Carmichael, Chiodini, Holroyd, Dean and Berriman2014). In one case, the removed worm was mistakenly reported as an adult, even though it was obviously a plerocercoid (Fu et al., Reference Fu, Ding, Li, Liu, Yang and Feng2019).
In Japan, the first sparganosis case was found in 1881 in Kyoto. Kagei (Reference Kagei2000) reported 546 cases from 1881 to 2000. A further 77 cases (Yoshikawa et al., Reference Yoshikawa, Ouji, Nishiofuku, Lshizaka and Nawa2010) and 32 cases (Nawa et al., Reference Nawa, Kokubo-Tanaka and Yamasaki2025) were added to the total. A further 5 cases that were not included in these reports (Yamasaki et al., Reference Yamasaki, Nakaya, Nakao, Sako and Ito2007; Ikuno et al., Reference Ikuno, Akao and Yamasaki2018) brings the total number to at least 660 by 2025, including a Japanese immigrant diagnosed in Germany (Tappe et al., Reference Tappe, Berger, Haeupler, Muntau, Racz, Harder, Specht, Prazeres da Costa and Poppert2013) and a sparganosis case due to S. asiana (Kudo et al., Reference Kudo, Fujioka, Korenaga, Yamasaki, Morishima, Sugiyama, Nakajima and Sano2017; Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b). The species reported by Tappe et al. as S. erinaceieuropaei is undoubtedly S. mansoni based on the cox1 sequence (Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021).
Kagei (Reference Kagei2000) reported 16 Japanese cases of sparganosis with multiple lesions (2‒6 plerocercoids per patient) and these cases are likely due to simultaneous ingestion of multiple plerocercoids or repeated infections, most probably due to S. mansoni.
In South Korea, 438 cases were reported between 1942 and 2015 (Kim et al., Reference Kim, Ahn, Sohn, Nawa and Kong2018), and at least 18 cases have been added by 2025 (e.g. Jo et al., Reference Jo, Lee, Hong, Kim and Han2016; Hong et al., Reference Hong, Kim, Lee, Kim and Kim2017; see Table 2 for others). Jeon et al. (Reference Jeon, Park, Lee, Choe, Kim, Huh, Sohn, Chai and Eom2015) reported 50 human sparganosis cases, of which 16 were caused by ‘S. decipiens’ and 34 were infected with ‘S. erinaceieuropaei’. However, according to the latest taxonomy, these must be classified as S. mansoni and S. asiana, respectively (Yamasaki et al., Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b). Interestingly, the image of a plerocercoid identified as S. erinaceieuropaei is shown (Jeon and Eom, Reference Jeon, Eom and Liu2018), but the plerocercoid closely resembles that of S. asiana (Kudo et al., Reference Kudo, Fujioka, Korenaga, Yamasaki, Morishima, Sugiyama, Nakajima and Sano2017; Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021). In South Korea, more human cases are due to S. asiana than to S. mansoni, while in Japan, the number of human cases caused by S. asiana is extremely low. The frequency of S. mansoni and S. asiana appears to vary by country and region (Yamasaki et al., Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b).
The situation of sparganosis in North Korea is unknown. Lee et al. (Reference Lee, Nam, Lee, Kang, Choi, Choi and Lee2018) reported a case of a North Korean defector with eosinophilic gastritis due to suspected cerebral sparganosis.
In Thailand, 52 cases reported between 1943 and 2005 were collated (Anantaphruti et al., Reference Anantaphruti, Nawa and Vanvanitchai2011), with at least 23 additional cases to 2025 (e.g. Akkarachinorate et al., Reference Akkarachinorate, Udnan, Chaisiri, Komalamisra, Dekumyoy, Pakdee, Praevanich, Mongkhonmu and Watthanakulpanich2013; Boonyasiri et al., Reference Boonyasiri, Cheunsuchon, Srirabheebhat, Yamasaki, Maleewong and Intapan2013; see Table 2), including a Thai immigrant diagnosed in Switzerland (Muigg et al., Reference Muigg, Ruf, Schwarzkopf, Huang, Denisjuk, Stürmann, Ritzler, Wampfler, Roppert and Neumayr2019).
A case of pulmonary sparganosis with multifocal lesions was reported in Thailand 30 years ago (Kanpittaya et al., Reference Kanpittaya, Boonsawad, Intarapoka, Kularbkaew, Tesana and Tantisirintr1995; Phunmanee et al., Reference Phunmanee, Boonsawat, Indharapoka, Tuntisirin and Kularbkeaw2001) and was considered at the time to be caused by Spa. proliferum, but molecular diagnosis using pathological tissue sections ultimately identified the cause as S. mansoni (Koonmee et al., Reference Koonmee, Intapan, Yamasaki, Sugiyama, Muto, Kuramochi, Kularbkeaw, Kanpittaya, Maleewong and Nawa2011).
In India, 20 cases were reported between 1998 and 2000 (Kaur and Shukla, Reference Kaur and Shukla2023). The number has been updated to 25 (from 1982 to 2023) (Nawa et al., Reference Nawa, Tanaka and Yoshikawa2024). Two additional cases have since been reported (Chougule et al., Reference Chougule, Mohite, Joshi and Agrawal2023; Rathore et al., Reference Rathore, Padmanabha, Mahale, Arora, Goyal, Reddy, Sipani, Pruthi, Lingaraju, Nagarathna, Yasha, Saini, Nashi, Pooja and Mathuranath2024), bringing the total number of cases since 1982 to at least 27.
In other Asian countries and regions, sparganosis has sporadically been reported: 25 cases from Taiwan (Tsai et al., Reference Tsai, Chang, Ho and Wang1993, Reference Tsai, Joseph and Chang2024; Chang et al., Reference Chang, Lin and Yeh1999; Tung et al., Reference Tung, Lin and Chou2005; Chiu et al., Reference Chiu, Chiou, Hsu and Yen2010; Ho et al., Reference Ho, Lin, Yu, Lai, Sheu and Bee2013; Hong et al., Reference Hong, Xie, Zhu, Wan, Xu and Wu2013; Hsu et al., Reference Hsu, Chung, Chen and Chung2020), 12 cases from Hong Kong (Huang and Kirk, Reference Huang and Kirk1962; Ng et al., Reference Thk, Wong, Fund and Leung1989; Ko, Reference Ko1991; Tang et al., Reference Tang, Wong, Lai, Poon, Chan, Wu, Cheung, Poon, Tsang, Tang and Wu2017), 6 cases from the Philippines (Ira-Concepcion, 1935 from Yogore and Tangco, Reference Yogore and Tangco1953; Eduardo, Reference Eduardo1991; Kazanjian and Mattia, Reference Kazanjian and Mattia1994; Tang et al., Reference Tang, Wong, Lai, Poon, Chan, Wu, Cheung, Poon, Tsang, Tang and Wu2017), 5 cases from Sri Lanka (Wijesundera et al., Reference Wijesundera, Ratnatunga, Kumarasinghe and Dissanaike1997; Dissanaike et al., Reference Dissanaike, Anthonis, Sheriffdeen, Ihalamulla and Karunaweera2001; Alibhoy et al., Reference Alibhoy, Fernando and Perera2007), 4 cases from Nepal (Sah et al., Reference Sah, Khatri, Kharel, Kc, Rabaan, Tiwari, Dhama, Malik, Donovan, Rodriguez-Morales, Muigg and Neumayr2020), including a Nepalese immigrant diagnosed in United Kingdom (Blundell et al., Reference Blundell, Deshmukh and McGregor2022), 3 cases from Indonesia (Bonne, Reference Bonne1942; Margono et al., Reference Margono, Sutjahyono, Kurniawan, Nakao, Mulyani, Wandra and Ito2007) and 1 case each of a Myanmar man diagnosed in Thailand (Iampreechakul et al., Reference Iampreechakul, Tirakotai, Lertbutsayanukul, Khunvutthidee, Thammachantha and Siriwimonmas2020), a Bangladesh immigrant diagnosed in Switzerland (Gonzenbach et al., Reference Gonzenbach, Kong, Beck, Buc, Weller and Semmler2013), East Timorese refugee diagnosed in Australia (Munckhof et al., Reference Munckhof, Grayson, Susil, Pullar and Turnidge1994; Tran et al., Reference Tran, Tran and Mehanna2019) and a case of a Melanesian man diagnosed in Papua New Guinea (Ashford et al., Reference Ashford, Murthy and Parsons1978; Owen, Reference Owen2005).
In Australia, there have been 5 case reports from 1905 to1994 and 3 cases were added in 2019. The etiological agents of the last 3 cases were identified as Spirometra mansonoides (Tran et al., Reference Tran, Tran and Mehanna2019), a species described from North America (Mueller, Reference Mueller1935), but no justification for this identification was given. Human sparganosis has occurred in Australian-born people who had never travelled overseas (Hughes and Biggs, Reference Hughes and Biggs2002; Tran et al., Reference Tran, Tran and Mehanna2019; Barton et al., Reference Barton, Fahey, Jenkins and Shamsi2023). However, most Australians cook meat thoroughly and there is a low risk of disease. Nevertheless, sparganosis is likely an under-recognized zoonotic disease in Australia (Tran et al., Reference Tran, Tran and Mehanna2019).
In addition to case reports of actual plerocercoids, serological tests for sparganosis greatly increase estimates of prevalence. In South Korea, seroprevalence was as high as 3.4% between 1993 and 2006, but it had decreased to 1.6% by 2006 (Lee et al., Reference Lee, Hong and Kim2010). In Indonesia, the seroprevalence rates were 2.9%, 6.9% and 2.7% in North Sumatra, Bali and Papua provinces, respectively (Margono et al., Reference Margono, Sutjahyono, Kurniawan, Nakao, Mulyani, Wandra and Ito2007). The situation of human sparganosis in North Korea is unknown, but it was reported that 11 (4.1%) of 270 North Korean refugees were seropositive (Shen et al., Reference Shen, Li, Zheng, Choi, Bae and Hong2007).
Proliferative sparganosis
Proliferative sparganosis is caused by plerocercoids which can invade various organs, including the subcutaneous tissue, bone and spinal cord, and then proliferate at a single site by continuous branching and budding, producing numerous progeny plerocercoids. The disease begins as a tumour-like subcutaneous nodule and has a poor prognosis, usually resulting in death (Ijima, Reference Ijima1905; Stiles, Reference Stiles1908; Moulinier et al., Reference Moulinier, Martinez, Torres, Noya, Noya and Reyes1982; Lo et al., Reference Lo, Chao, Yan, Liu, Chu, Huang, Chang and Liu1987; Nakamura et al., Reference Nakamura, Hara, Matsuoka, Kawabata and Tsuji1990; Kikuchi and Maruyama, Reference Kikuchi and Maruyama2020).
Eighteen cases of proliferative sparganosis were reported worldwide (Kikuchi and Maruyama, Reference Kikuchi and Maruyama2020), with an additional 5 to be added (Marjoribanks and Le Sueur, Reference Marjoribanks and Le Sueur1924; Connor et al., Reference Connor, Sparks, Strano, Neafie and Juvelier1976; Lin et al., Reference Lin, Su, Lu and Yang1978; LaChance et al., Reference LaChance, Clark and Connor1983; Olson et al., Reference Olson, Yoder, Fajardo L-G, Marty, van de Pas, Olivier and Relman2003), bringing the total to 23, 17 of which were in Asia, including 6 in Japan. Other cases reported from Asian countries are listed in Table 2.
Although not all cases yielded abundant plerocercoids, proliferative sparganosis has traditionally been diagnosed based on the peculiar plerocercoid morphology and histology (e.g. Ijima, Reference Ijima1905; Usui, Reference Usui1909; see Table 2 for others).
Ijima (Reference Ijima1905) and Iwata and Inatomi (Reference Iwata and Inatomi1972) stated that ‘Spa. proliferum’ might be aberrant plerocercoids of the closed related to L. mansoni (Ijima, Reference Ijima1905) and D. erinacei (= S. mansoni) (Iwata and Inatomi, Reference Iwata and Inatomi1972). Although these statements seemed to have been largely overlooked, Nakamura et al. (Reference Nakamura, Hara, Matsuoka, Kawabata and Tsuji1990) supported it, stating that Spa. proliferum is not a rare species but an aberrant form of the commonly distributed species, Spirometra erinacei (a synonym of S. erinaceieuropaei, but actually referring to S. mansoni), and speculated that abnormalities in the immune system of the host might play an important role in proliferation.
In proliferative cases reported in Japan, Taiwan, Thailand and Malaysia, the patient’s immune status was not documented (Ijima, Reference Ijima1905; Usui, Reference Usui1909; Inoue, Reference Inoue1911; Yoshida, Reference Yoshida1914; Akanuma, Reference Akanuma1920; Tashiro, Reference Tashiro1921; Marjoribanks and Le Sueur, Reference Marjoribanks and Le Sueur1924; Aoshima et al., Reference Aoshima, Nakata, Matsuoka, Kawabata and Nakamura1989). A Chinese patient diagnosed with proliferative sparganosis had been treated with anticancer drugs (Ho and Huang, Reference Ho and Huand1984), but the immune status of the patient was unknown.
Overseas, disseminated sparganosis cases have been reported in Hodgkin disease and AIDS patients (Connor et al., Reference Connor, Sparks, Strano, Neafie and Juvelier1976; Olson et al., Reference Olson, Yoder, Fajardo L-G, Marty, van de Pas, Olivier and Relman2003; Meric et al., Reference Meric, Ilie, Hofman, Rioux-Leclercq, Michot, Haffaf, Nelson, Neafie and Hofman2010), possibly due to mutated plerocercoids (Connor et al., Reference Connor, Sparks, Strano, Neafie and Juvelier1976).
Schauer et al. (Reference Schauer, Poppert, Technau-Hafsi, Mockenhaupt, Muntau, Häcker, Tappe and Jokob2014) reported a patient who had travelled to Bolivia, Brazil and Paraguay and was diagnosed with Spa. proliferum by DNA analysis. However, since the excised single plerocercoid showed neither proliferation nor branching, and the molecular data for the parasite are also not available in GenBank, the identity is uncertain. The authors speculated that Spa. proliferum might exhibit proliferative characteristics only in immunocompromised individuals.
In addition to proliferative sparganosis in immunocompromised patients, 3 proliferative sparganosis cases have recently been reported in cat and dogs in Japan, the USA and Australia (Tokiwa et al., Reference Tokiwa, Fushimi, Chou, Yoshida, Kinoshita, Hikima, Kikuchi and Ozaki2024; Semenova et al., Reference Semenova, Walden, Bollenback, Cook and Ossiboff2025; Young et al., Reference Young, Malik, Brown, Wang, Ash, Korhonen and Gasser2025). Molecular data indicate that the causative species in these cases are all S. mansoni (Table 1 and Figure 1). Corticosteroids had been administered for 1 year to relieve itching in the cat case (Tokiwa et al., Reference Tokiwa, Fushimi, Chou, Yoshida, Kinoshita, Hikima, Kikuchi and Ozaki2024).
Table 1. Etiological agents caused proliferative sparganosis with molecular data

* Accession numbers with asterisks indicate cox1 sequences used for phylogenetic analysis.
Although the causal relationship between the asexual proliferation of plerocercoids and the immune status is unknown, these findings strongly support Ijima (Reference Ijima1905) and Iwata’s (Reference Iwata1933, Reference Iwata1972) statements that proliferative sparganosis in Asia is likely caused by aberrant plerocercoids of S. mansoni, different from Spa. proliferum from Venezuela.
While the factor(s) causing plerocercoids to become aberrant remain unknown, transcriptome analysis of proliferative plerocercoids of Venezuelan Spa. proliferum has identified some genes involved in proliferation (Kikuchi et al., Reference Kikuchi, Dayi, Hunt, Ishiwata, Toyoda, Kounosu, Sun, Maeda, Kondo, Alarcón de Noya, Alarcón de Noya, Kojima, Kuramochi and Maruyama2021), As mentioned above, some reports suggest that the host’s immune status may be involved in asexual proliferation and aberrant forms, but this remains an issue that requires further investigation.
We consider that the species causing proliferative sparganosis in Asia and Oceania is S. mansoni and/or S. asiana, although there is as-yet no evidence that S. asiana causes proliferative sparganosis.
Spirometrosis
Spirometrosis (infection with adults) in humans rarely occurs. The adult worm usually inhabits the small intestine, producing symptoms that are generally mild, such as abdominal distension and diarrhoea (Kagei, Reference Kagei2000; Yamaura et al., Reference Yamaura, Kikuchi, Itoda, Yasunami, Totsuka, Shimada, Nakamura, Hayashi, Isoda, Kusumoto, Kobayakawa, Kagei and Araki2002; Le et al., Reference Le, Lqt, Nguyen, Nguyen and Do2017). Twenty-two cases have been reported in China since 1932 (Tu et al., Reference Tu, Chiu and Chou1973; Wang et al., Reference Wang, Tang, Yang, Xu, Wang, Jin, Zhu, Huang and Gan2012). In Japan, 16 cases have been reported (Kagei, Reference Kagei2000; Yamaura et al., Reference Yamaura, Kikuchi, Itoda, Yasunami, Totsuka, Shimada, Nakamura, Hayashi, Isoda, Kusumoto, Kobayakawa, Kagei and Araki2002), and 2 and 1 cases have been reported in South Korea (Lee et al., Reference Lee, Chai, Seo and Cho1984) and Vietnam (Le et al., Reference Le, Lqt, Nguyen, Nguyen and Do2017), respectively (Table 2).
Table 2. Human sparganosis/spirometrosis reported in Asian and Oceanian regions

Infections with adult tapeworms reported in tropical regions in Asia (e.g. India and Bangladesh) where Dibothriocephalus species are not known to occur should be re-evaluated and the possibility of spirometrosis is considered (Kuchta et al., Reference Kuchta, Phillips and Scholz2024).
In remote areas where patients with suspected sparganosis and spirometrosis often reside, hospitals and laboratory facilities may be poorly established, and infected individuals may not seek medical attention. There is thus likely to be a substantial under-reporting of sparganosis and spirometrosis. The numbers of the cases shown in Table 2 are probably very underestimated, especially in countries where sparganosis is endemic due to dietary habits of eating flesh of tadpoles, frogs and snakes and the use of frog meat as a traditional poultice in China (Li et al., Reference Li, Song, Li, Lin, Xie, Lin and Zhu2011; Lu et al., Reference Lu, Shi, Lu, Wu, Li, Rao and Yin2014; Liu et al., Reference Liu, Li, Wang, Zhao and Zhu2015; Cui et al., Reference Cui, Wang, Zhang, Lin, Zhang, Wang and Chen2017), South Korea (Liu et al., Reference Liu, Li, Wang, Zhao and Zhu2015), Thailand (Wiwanitkit, Reference Wiwanitkit2005; Anantaphruti et al., Reference Anantaphruti, Nawa and Vanvanitchai2011; Saksirisampant et al., Reference Saksirisampant, Eamudomkarn, Jeon, Eom, Assavapongpaiboon, Sintuwong and Tulvatana2020), Myanmar (Chai et al., Reference Chai, Jung, Ryu, Kim, Hong, Htoon, Tin, Na and Sohn2020) and Indonesia (Prasetyo and Safitri, Reference Prasetyo and Safitri2019).
To date, no human sparganosis or spirometrosis cases have been reported in Lao PDR, Cambodia and Singapore. However, Spirometra infections have been confirmed in reptiles, amphibians and mammals in these countries (Okamoto et al., Reference Okamoto, Iseno, Shibahara, Sato, Wandra, Craig and Ito2007; Jongthawin et al., Reference Jongthawin, Intapan, Sanpool, Sadaow, Laymanivong, Thanchomnang and Maleewong2014; Kavana et al., Reference Kavana, Lim and Ambu2014; Sato et al., Reference Sato, Sato, Yoonuan, Pongvongsa, Sanguankiat, Kounnavong, Maipanich, Chigusa, Moji and Waikagul2017; Jeon et al., Reference Jeon, Park, Lee, Choe, Kang, Bia, Lee, Sohn, Hong, Chai and Eom2018b; Ahasan et al., Reference Ahasan, Azam, Mohanto, Rahman, Islam, Anisuzzaman, Khan, Begum and Chowdhury2020; Thanchomnang et al., Reference Thanchomnang, Sanpool, Yamasaki, Intapan, Maleewong, Petney, Saijuntha and Mehlhorn2021; Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021; Yudhana et al., Reference Yudhana, Praja and Kartikasari2021; Zendejas-Heredia et al., Reference Zendejas-Heredia, Colella, Huggins, Schaper, Schunack and Traub2023; Kuchta et al., Reference Kuchta, Phillips and Scholz2024; Nguyen et al., Reference Nguyen, Nguyen, Dong, Duong and Yoshida2024), Iran (Badria et al., Reference Badria, Eslahi, Majidiania and Pirestani2017), Guam Island (Holldorf et al., Reference Holldorf, Siers, Richmond, Klug and Reed2015) and New Zealand (Ugarte et al., Reference Ugarte, Thomas, Gasser, Hu, Scott and Collett2005), although the species identified as S. erinaceieuropaei in these countries has been re-assigned to S. mansoni (Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Sugiyama, Morishima and Kobayashi2024b; Kuchta et al., Reference Kuchta, Phillips and Scholz2024) (Figure 2).
Serodiagnosis of sparganosis
Various serological methods that detect specific IgG against Spirometra infections have been established using native and recombinant antigens. Platforms used for these include enzyme-linked immunosorbent assay (ELISA) (Lee et al., Reference Lee, Bae, Kim, Deung and Ryang2002; Cui et al., Reference Cui, Li, Wang, Jiang and Lin2011; Rahman et al., Reference Rahman, Kim, Hong and Choi2014; Choi, Reference Choi2024), multidot-ELISA (Nakamura-Uchiyama, Reference Nakamura-Uchiyama2005) and western blot (Chung et al., Reference Chung, Kong, Yang and Cho2000; Rahman et al., Reference Rahman, Lee and Bae2011; Lu et al., Reference Lu, Sun, Lu, Chen, Song, Ai, Cai, Li and Chen2021). These methods are labour-intensive and require sophisticated equipment and skilled technicians. Recently, innovative immunochromatography-based diagnostic tools (ICT) are being developed as alternatives to the conventional methods (Yamasaki et al., Reference Yamasaki, Nakamura, Intapan, Maleewong, Morishima, Sugiyama, Matsuoka, Kobayashi, Takayama and Kobayashi2014, Reference Yamasaki, Nakamura, Intapan, Maleewong, Morishima, Sugiyama, Kobayashi, Takayama and Kobayashi2018, Reference Yamasaki, Morishima and Sugiyama2024a; Harasan et al., Reference Harasan, Sadaow, Boonroumkaew, Rodpai, Sanpool, Yamasaki, Intapan and Maleewong2025). Detailed accounts of the principles of the ICT are available in Janwan et al. (Reference Janwan, Intapan, Sadaow, Rodpai, Yamasaki, Boonroumkaew, Sanpool, Thanchomnang, Sadee and Maleewong2021) and Rodpai et al. (Reference Rodpai, Sadaow, Boonroumkaew, Phupiewkham, Thanchomnang, Limpanont, Chusongsang, Sanpool, Ohmae, Yamasaki, Intapan and Maleewong2021).
We initially developed an ICT kit for rapid diagnosis of sparganosis (Yamasaki et al., Reference Yamasaki, Nakamura, Intapan, Maleewong, Morishima, Sugiyama, Matsuoka, Kobayashi, Takayama and Kobayashi2014), but later the kit was improved to test for larval migrans due to sparganosis, toxocariasis and gnathostomiasis on a single panel (Figure 4A) (Yamasaki et al., Reference Yamasaki, Nakamura, Intapan, Maleewong, Morishima, Sugiyama, Kobayashi, Takayama and Kobayashi2018). The kit has perforations so that individual tests can be removed and used alone (Figure 4B,C). Such tests are simple to perform: briefly, 5 uL of serum diluted in running buffer is applied into a well that is marked ‘S’. Next, 60 uL of running buffer is applied into the well marked ‘B’. ICT results are read at 15 min with the naked eye and judged as positive or negative by reference to a colour board, with the cutoff for positivity set at an intensity of 0.5. The appearance of red bands at the T-line and the C-line indicate a positive test (Figure 4), whereas only the C-line appears in a negative case.

Figure 4. Immunochromatography-based point-of-care tool for the serodiagnosis of species causing larva migrans. (A) Device for toxocariasis (left), sparganosis (middle) and gnathostomiasis (right). Diagnostic results for Spirometra mansoni (B) and Spirometra asiana infections (C) using separate devices (Yamasaki et al., Reference Yamasaki, Morishima and Sugiyama2024a). (D) Colour board for evaluation. Wells S and B are for applying serum (or whole blood) and running buffer, respectively. An arrow at the T-lines (B, C) indicates the appearance of the positive bands.
The kit is useful not only for the diagnosis of S. mansoni and S. asiana infections and for follow-up after treatment but is also applicable for field surveys for sparganosis cases (Yamasaki et al., Reference Yamasaki, Sanpool, Rodpai, Sadaow, Laummaunwai, Un, Thanchomnang, Laymanivong, Aung, Intapan and Maleewong2021, Reference Yamasaki, Morishima and Sugiyama2024a; Harasan et al., Reference Harasan, Sadaow, Boonroumkaew, Rodpai, Sanpool, Yamasaki, Intapan and Maleewong2025). Since the kit occasionally shows cross-reactions with cysticercosis, careful consideration of factors such as dietary history, history of overseas travel and imaging findings is required when determining a diagnosis (Yamasaki et al., Reference Yamasaki, Nakamura, Intapan, Maleewong, Morishima, Sugiyama, Matsuoka, Kobayashi, Takayama and Kobayashi2014).
It is expected that the ICT kits can also be used to diagnose proliferative sparganosis in South America, although this has not yet been tested to date. The ICT format has recently been modified to use whole blood instead of serum (Harasan et al., Reference Harasan, Sadaow, Boonroumkaew, Rodpai, Sanpool, Yamasaki, Intapan and Maleewong2025) but this format is not yet commercially available.
Conclusions and perspectives
This review has summarized the latest information about the identities of Spirometra species and the current situation of sparganosis and spirometrosis in Asia and Oceania. Despite their importance as foodborne diseases, sparganosis and spirometrosis are neglected parasitic diseases in both developing and developed countries, and few public-health strategies exist for their prevention and control. Recently, it has been confirmed that S. mansoni can cause proliferative sparganosis in the Asia–Oceania region. Therefore, physicians, veterinarians and related professionals should be aware of the following: (i) the Spirometra species present in Asia–Oceania region are S. mansoni and S. asiana, but not S. erinaceieuropaei, (ii) if proliferative sparganosis is suspected, the causative species should not be identified to be Spa. proliferum based on morphological characteristics alone of the plerocercoids. Heavy or multiple infections caused by S. mansoni and/or S. asiana should be considered. Molecular identification is indispensable for diagnosing the causative species, (iii) S. asiana is a recently discovered species, and little is known about its biology, geographical distribution and pathogenicity in humans, further data are needed, and (iv) since wild boars serve as paratenic hosts for both S. mansoni and S. asiana, it is important to thoroughly cook wild boar meat to prevent infection.
Acknowledgements
We thank David Blair (James Cook University, Australia) for valuable suggestions, English editing and assistance with this paper’s presentation through the National Research Council of Thailand (NRCT): High-Potential Research Team Grant Program (Contract no. N42A670561 to WM) and Roman Kuchta (Czech Academy of Sciences) for valuable comments on Sparganum proliferum. We also express our deep gratitude Oranuch Sanpool, Lakkhana Sadaow, Patcharaporn Boonroumkaew (Khon Kaen University and Mekong Health Science Research Institute, Thailand), Rutchanee Rodpai (Nakhonratchasima College, Thailand), Tongjit Thanchomnang (Mahasarakham University, Thailand), Penchom Janwan (Walailak University, Thailand), Sakhone Laymanivong (Ministry of Health, Lao PDR), Win Pa Pa Aung (University of Medicine 2, Myanmar), Yasuyuki Morishima and Hiromu Sugiyama (Japan Institute of Health Security, Japan) for collaborative research, and Naoko Wakiya, Miou Hisada and Mariko Yamashita (Japan Institute of Health Security, Japan) for their efforts in obtaining many references.
Author contributions
HY wrote the first draft of the manuscript and prepared the figures and table. PMI and WM reviewed and edited the manuscript. HY, PMI and WM approved the final version of the manuscript for submission.
Financial support
We have no financial support for preparing this review.
Competing interests
The authors declare no competing interests.
Ethical standards
Not applicable.