Non-technical Summary
We present an overview of conservation paleobiology and the directions in which the field could progress in the next 50 years to aid conservation. To do so, we use elasmobranchs (sharks, rays, and skates), one of the mostly highly marine endangered groups today, as a model. The perspectives we share are guided by current conservation priorities and recent advances in elasmobranch paleobiology and are developed around four main topics. For each topic, we outline knowledge gaps, discuss the potential of near- and deep-time records to contribute relevant information, highlight examples, and suggest research directions. Ultimately, we aim at focusing conservation paleobiology research agendas, encouraging collaborations across timescales, and distilling lessons that could be transferred to other threatened but understudied taxa in conservation paleobiology.
Introduction
Over the previous millennia, humans have dramatically transformed ecosystems and pushed species to the edge of extinction (Vitousek et al. Reference Vitousek, Mooney, Lubchenco and Melillo1997; Richardson et al. Reference Richardson, Steffen, Lucht, Bendtsen, Cornell, Donges and Drüke2023). These anthropogenic impacts have accelerated since the 1950s (Steffen et al. Reference Steffen, Broadgate, Deutsch, Gaffney and Ludwig2015) and have potentially triggered a sixth mass extinction event comparable in magnitude to the other five that shaped the history of life (Barnosky et al. Reference Barnosky, Matzke, Tomiya, Wogan, Swartz, Quental and Marshall2011; Dirzo et al. Reference Dirzo, Young, Galetti, Ceballos, Isaac and Collen2014; Ceballos et al. Reference Ceballos, Ehrlich, Barnosky, García, Pringle and Palmer2015). Change has progressed over different timescales on land and in the ocean, with marine impacts beginning more recently, so we sit at a critical moment where recoveries remain within reach (McCauley et al. Reference McCauley, Pinsky, Palumbi, Estes, Joyce and Warner2015). Because resources are finite, decision makers must prioritize what and where to protect as well as which benchmarks to use when tracking progress toward conservation goals. Information about natural ecosystem states and vulnerabilities can help guide these decisions. However, human impacts often preceded the instrumental records and biological monitoring programs used to collect this information, shifting the reference points used for conservation (Pauly Reference Pauly1995; Jackson Reference Jackson2001).
The geological record captures a long-term view of environmental change that can inform conservation and inspire action. Geohistorical records (e.g., fossil records, sediment cores, tree rings, and archaeological middens) extend our understanding of ecosystem trajectories beyond the time frame of direct human observation (National Research Council 2005). They are commonly divided into near time (last 2.58 Myr, known as the Quaternary Period) and deep time (older than 2.58 Myr), which vary in scale, resolution, and completeness, and reflect different eco-evolutionary processes (Dietl and Flessa Reference Dietl and Flessa2011; Fig. 1). Collectively, geohistorical records can be used to reconstruct natural variability before human disturbance (Willis and Birks Reference Willis and Birks2006; Keane et al. Reference Keane, Hessburg, Landres and Swanson2009; O'Dea et al. Reference O'Dea, Lepore, Altieri, Chan, Morales-Saldaña, Muñoz, Pandolfi, Toscano, Zhao and Dillon2020; Fig. 1C–G), document the patterns and drivers of biotic change over millennia to millions of years (Jablonski and Sepkoski Reference Jablonski and Sepkoski1996; Jackson and Blois Reference Jackson and Blois2015; Duda et al. Reference Duda, Allen-Mahé, Barbraud, Blais, Boudreau, Bryant and Delord2020; Fig. 1A–G), estimate extinction risk (Harnik et al. Reference Harnik, Lotze, Anderson, Finkel, Finnegan, Lindberg and Liow2012; Finnegan et al. Reference Finnegan, Anderson, Harnik, Simpson, Tittensor, Byrnes and Finkel2015; Collins et al. Reference Collins, Edie, Hunt, Roy and Jablonski2018; Fig. 1B), and anticipate species’ responses to future climatic conditions that have yet to be observed in the instrumental record (Davis Reference Davis1989; Willis et al. Reference Willis, Bailey, Bhagwat and Birks2010; Fordham et al. Reference Fordham, Jackson, Brown, Huntley, Brook, Dahl-Jensen and Gilbert2020; Fig. 1A). The potential for geohistorical records to tackle conservation challenges has accrued over decades (e.g., Martin and Wright Reference Martin and Wright1967; Graham Reference Graham1988; Smol Reference Smol1992; Birks Reference Birks1996; Swetnam et al. Reference Swetnam, Allen and Betancourt1999; Jackson et al. Reference Jackson, Kirby, Berger, Bjorndal, Botsford, Bourque and Bradbury2001), setting the stage for conservation paleobiology to emerge as a named subfield in 2002 (Flessa Reference Flessa2002).
Examples of conservation paleobiology in action. Conservation paleobiology uses a variety of near-time (blue; last 2.58 Myr) and deep-time (yellow; older than 2.58 Myr) geohistorical records to extend the temporal span of direct observations (green; ca. last century). The application of deep-time geohistorical records to conservation practice remains unrealized, although it has promise: A, insect herbivory increased in North America during the Paleocene–Eocene thermal maximum (rapid global warming ca. 56 Ma), offering an analogue for how future warming might precipitate heightened insect damage to plants (Labandeira and Currano Reference Labandeira and Currano2013); B, morphological traits of fossil Caribbean corals during the Plio-Pleistocene were used to predict the extinction risk of extant corals and validate their conservation status (Raja et al. Reference Raja, Lauchstedt, Pandolfi, Kim, Budd and Kiessling2021). Examples of conservation paleobiology studies resulting in tangible conservation outcomes are accumulating: C, caribou antlers exposed on landscapes dating back decades to millennia have supported spatial management plans (Miller et al. Reference Miller, Crowley, Bataille, Wald, Kelly, Gaetano, Bahn and Druckenmiller2021, Reference Miller, Wald and Druckenmiller2023); D, excavations from Makauwahi Cave Reserve on Kaua‘i have informed forest restoration and the introduction of giant tortoises to fill lost ecological roles (Burney et al. Reference Burney, James, Burney, Olson, Kikuchi, Wagner and Burney2001); E, intertidal death assemblages, archaeological shell middens, and modern clams have guided Indigenous-led ecosystem restoration goals and traditional clam gardening practices in the Salish Sea of British Columbia, Canada (Toniello et al. Reference Toniello, Lepofsky, Lertzman-Lepofsky, Salomon and Rowell2019); F, Holocene-age coral subfossils defined spatially explicit historical baselines for coral outplanting in Hong Kong (Cybulski et al. Reference Cybulski, Husa, Duprey, Mamo, Tsang, Yasuhara, Xie, Qiu, Yokoyama and Baker2020); G, estimates of pre-alteration (before 1900 CE) hydrology reconstructed from pollen and mollusks were used to set salinity targets in the Florida Bay and manage freshwater flow through the Greater Everglades ecosystem (Marshall et al. Reference Marshall, Wingard and Pitts2014; Wingard et al. Reference Wingard, Bernhardt and Wachnicka2017). Illustrations by Ian Cooke Tapia (Cooked Illustrations).
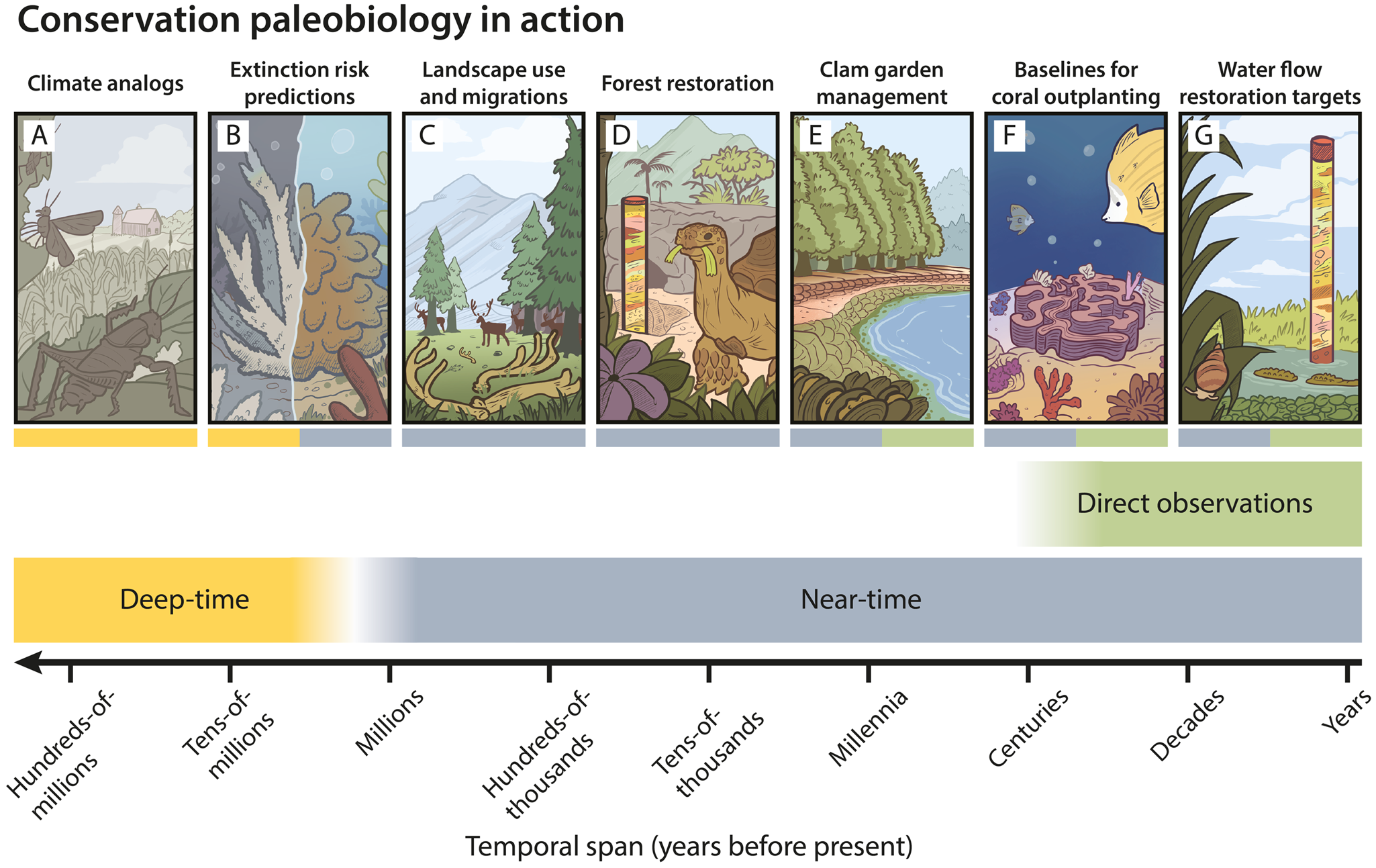
Conservation paleobiology aims to apply geohistorical records to the conservation, management, and restoration of biodiversity and ecosystem services (Dietl and Flessa Reference Dietl and Flessa2011; Dietl et al. Reference Dietl, Kidwell, Brenner, Burney, Flessa, Jackson and Koch2015). It integrates information from a variety of related disciplines, such as archaeology, paleoecology, historical ecology, and conservation biology (Louys Reference Louys2012; Barnosky et al. Reference Barnosky, Hadly, Gonzalez, Head, Polly, Lawing and Eronen2017; Dillon et al. Reference Dillon, Pier, Smith, Raja, Dimitrijević, Austin and Cybulski2022b), and it has benefited from recent advances in paleontological and analytical methods (Dillon et al. Reference Dillon, Dunne, Womack, Kouvari, Larina, Claytor and Ivkić2023; Tomašových et al. Reference Tomašových, Dominici, Nawrot and Zuschin2023) as well as interdisciplinary collaborations (e.g., Conservation Paleobiology Network, https://conservationpaleorcn.org). Over the last two decades, conservation paleobiology has attracted a growing research community interested in leveraging geohistorical records to help address the climate and biodiversity crises (Dillon et al. Reference Dillon, Pier, Smith, Raja, Dimitrijević, Austin and Cybulski2022b). Examples of conservation paleobiology research in action are mounting: geohistorical records have supported caribou management in Alaska (Miller et al. Reference Miller, Crowley, Bataille, Wald, Kelly, Gaetano, Bahn and Druckenmiller2021, Reference Miller, Wald and Druckenmiller2023; Fig. 1C), forest restoration on Kaua‘i (Burney et al. Reference Burney, James, Burney, Olson, Kikuchi, Wagner and Burney2001; Fig. 1D), traditional clam gardening practices in British Columbia (Toniello et al. Reference Toniello, Lepofsky, Lertzman-Lepofsky, Salomon and Rowell2019; Fig. 1E), coral outplanting in Hong Kong (Cybulski et al. Reference Cybulski, Husa, Duprey, Mamo, Tsang, Yasuhara, Xie, Qiu, Yokoyama and Baker2020; Fig. 1F), and freshwater flow restoration in the Florida Everglades (Marshall et al. Reference Marshall, Wingard and Pitts2014; Wingard et al. Reference Wingard, Bernhardt and Wachnicka2017; Fig. 1G). These case studies have begun to instill optimism in the field's ability to “put the dead to work” (Dietl and Flessa Reference Dietl and Flessa2011) and provide a framework for others to follow (Groff et al. Reference Groff, McDonough MacKenzie, Pier, Shaffer and Dietl2023).
However, most geohistorical records do not influence conservation decisions. A review of the conservation paleobiology literature found that only ~10% of research resulted in tangible conservation outcomes, with all successful examples using near-time records of extant species (Groff et al. Reference Groff, McDonough MacKenzie, Pier, Shaffer and Dietl2023). Although not all conservation paleobiology research needs to have direct real-world impacts, the field is still grappling with the research–implementation “gap” as it comes to terms with how applied it is, and should be, in practice (Boyer et al. Reference Boyer, Brenner, Burney, Pandolfi, Savarese, Dietl, Flessa, Dietl and Flessa2017; Kelley et al. Reference Kelley, Dietl, Visaggi, Tyler and Schneider2018; Savarese Reference Savarese, Tyler and Schneider2018; Dillon et al. Reference Dillon, Pier, Smith, Raja, Dimitrijević, Austin and Cybulski2022b). Implementation is, in part, impeded by disconnects between research and conservation priorities, such as when researchers collect data without first targeting a relevant conservation need or defining project goals with practitioners (Knight et al. Reference Knight, Cowling, Rouget, Balmford, Lombard and Campbell2008; Beier et al. Reference Beier, Hansen, Helbrecht and Behar2017; Toomey et al. Reference Toomey, Knight and Barlow2017). For example, conservation paleobiology research focuses on a few taxonomic groups, such that many taxa are underrepresented relative to their extinction risk (Dillon et al. Reference Dillon, Pier, Smith, Raja, Dimitrijević, Austin and Cybulski2022b). If conservation paleobiology aspires to increase its salience in conservation, reframing research agendas and collaborating with practitioners could bring the field closer to generating actionable science that is aligned with conservation needs.
One taxonomic group that has received little attention in conservation paleobiology research is marine vertebrates—in particular, the most threatened marine vertebrate group today: elasmobranchs (sharks, rays, and skates) (Dulvy et al. Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021; Dillon et al. Reference Dillon, Pier, Smith, Raja, Dimitrijević, Austin and Cybulski2022b; Sherman et al. Reference Sherman, Simpfendorfer, Pacoureau, Matsushiba, Yan, Walls and Rigby2023; Fig. 1). Elasmobranchs are found throughout all major marine (as well as some brackish and freshwater) ecosystems, where they play important ecological roles such as structuring food webs as upper trophic level consumers (Ferretti et al. Reference Ferretti, Worm, Britten, Heithaus and Lotze2010; Hammerschlag et al. Reference Hammerschlag, Schmitz, Flecker, Lafferty, Sih, Atwood, Gallagher, Irschick, Skubel and Cooke2019; Flowers et al. Reference Flowers, Heithaus and Papastamatiou2021; Heithaus et al. Reference Heithaus, Dunn, Farabaugh, Lester, Madin, Meekan, Papastamatiou, Roff, Vaudo, Wirsing, Carrier, Simpfendorfer, Heithaus and Yopak2022; Fig. 2A). Elasmobranchs are also economically beneficial for ecotourism (Gallagher and Hammerschlag Reference Gallagher and Hammerschlag2011; Cisneros-Montemayor et al. Reference Cisneros-Montemayor, Barnes-Mauthe, Al-Abdulrazzak, Navarro-Holm and Sumaila2013), contribute to food security (Glaus et al. Reference Glaus, Adrian-Kalchhauser, Piovano, Appleyard, Brunnschweiler and Rico2019), and have cultural significance (de Borhegyi Reference de Borhegyi1961; Skubel et al. Reference Skubel, Shriver-Rice and Maranto2019; Fig. 2B). However, elasmobranchs are threatened by overfishing (Dulvy et al. Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021; Fig. 2C), which has drastically reduced their populations in the last decades (e.g., Pacoureau et al. Reference Pacoureau, Rigby, Kyne, Sherley, Winker, Carlson and Fordham2021). If elasmobranch species go extinct, not only would biodiversity be lost but also millions of years of evolutionary history and ecological functions that could not be replaced (Stein et al. Reference Stein, Mull, Kuhn, Aschliman, Davidson, Joy, Smith, Dulvy and Mooers2018; Pimiento et al. Reference Pimiento, Leprieur, Silvestro, Lefcheck, Albouy, Rasher, Davis, Svenning and Griffin2020, Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023). Despite how important they are to protect (Fig. 2D), elasmobranchs have only recently entered policy agendas (Dulvy et al. Reference Dulvy, Baum, Clarke, Compagno, Cortés, Domingo and Fordham2008, Reference Dulvy, Simpfendorfer, Davidson, Fordham, Bräutigam, Sant and Welch2017; Fordham et al. Reference Fordham, Lawson, Koubrak, Cronin, Carrier, Simpfendorfer, Heithaus and Yopak2022), so effective management is hindered by a lack of information about their biology, ecology, and conservation status (Jorgensen et al. Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022).
The potential of the elasmobranch fossil record to inform conservation. A, Elasmobranchs structure food webs, with their elimination potentially resulting in cascading effects (Heithaus et al. Reference Heithaus, Dunn, Farabaugh, Lester, Madin, Meekan, Papastamatiou, Roff, Vaudo, Wirsing, Carrier, Simpfendorfer, Heithaus and Yopak2022). B, They are socioeconomically important, as they drive tourism (Cisneros-Montemayor et al. Reference Cisneros-Montemayor, Barnes-Mauthe, Al-Abdulrazzak, Navarro-Holm and Sumaila2013) and are a source of protein for artisanal fishers (Glaus et al. Reference Glaus, Adrian-Kalchhauser, Piovano, Appleyard, Brunnschweiler and Rico2019). They also hold cultural significance, as evidenced by archaeological artifacts and motifs found in Central America, among other regions (de Borhegyi Reference de Borhegyi1961). C, Overfishing is the primary threat to elasmobranch populations (Dulvy et al. Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021). Given the antiquity of fishing, most systematic monitoring studies are predicated on a shifted baseline, so a long-term perspective is needed to reconstruct natural variation in elasmobranch communities. D, Elasmobranchs are the most threatened marine vertebrate group today (Dulvy et al. Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021) and need protection. E, Elasmobranchs have a rich fossil record because their teeth and dermal denticles are shed continuously and are composed of hard materials that easily fossilize. F, Modern elasmobranchs have a long evolutionary history, with extant taxa being well represented in the fossil record (Paillard et al. Reference Paillard, Shimada and Pimiento2021). G, Fossil elasmobranch teeth can often be identified to the species level, and their morphological traits enable ecological inferences (Cooper et al. Reference Cooper, Griffin, Kindlimann and Pimiento2023). H, The elasmobranch fossil record is well documented in the paleontological literature. Illustrations by Ian Cooke Tapia (Cooked Illustrations).

The elasmobranch fossil record is often overlooked when addressing these knowledge gaps. There has been a dwindling presence of paleobiological work at relevant biology conferences (Shiffman et al. Reference Shiffman, Ajemian, Carrier, Daly-Engel, Davis, Dulvy and Grubbs2020) and little buy-in for including geohistorical records in elasmobranch conservation assessments (McClenachan et al. Reference McClenachan, Ferretti and Baum2012; Engelhard et al. Reference Engelhard, Thurstan, MacKenzie, Alleway, Bannister, Cardinale and Clarke2016). Paleobiology has a lot to contribute to these conversations. First, elasmobranchs have a rich fossil record, with their teeth being the most abundant vertebrate fossil (Maisey Reference Maisey1984; Cappetta Reference Cappetta and Schultze2012). This is because their teeth (and dermal denticles) are shed continuously and are composed of hard materials that easily fossilize (Fig. 2E). Second, the fossil record of modern elasmobranchs and their extinct relatives (Neoselachii) is geologically extensive, dating back to the Triassic (250 Ma; Maisey et al. Reference Maisey, Naylor and Ward2004; Cappetta Reference Cappetta and Schultze2012; Fig. 2F). Importantly, many extant taxa are represented in the fossil record, including all orders, 88% of families, 75% of genera, and 10% of modern species (Pimiento and Benton Reference Pimiento and Benton2020; Paillard et al. Reference Paillard, Shimada and Pimiento2021). Third, elasmobranch fossils provide valuable biological information (Naylor and Marcus Reference Naylor and Marcus1994; Cooper et al. Reference Cooper, Griffin, Kindlimann and Pimiento2023; Fig. 2G). Fourth, they are well-documented in the scientific literature, with around 17,000 occurrences in the Paleobiology Database (Fig. 2H). Finally, iconic specimens such as Otodus megalodon teeth capture the public's imagination and tell stories about ancient animals (Clements et al. Reference Clements, Atterby, Cleary, Dearden and Rossi2022). Elasmobranchs therefore represent a ripe opportunity to incorporate geohistorical records into conservation practice (Fig. 2).
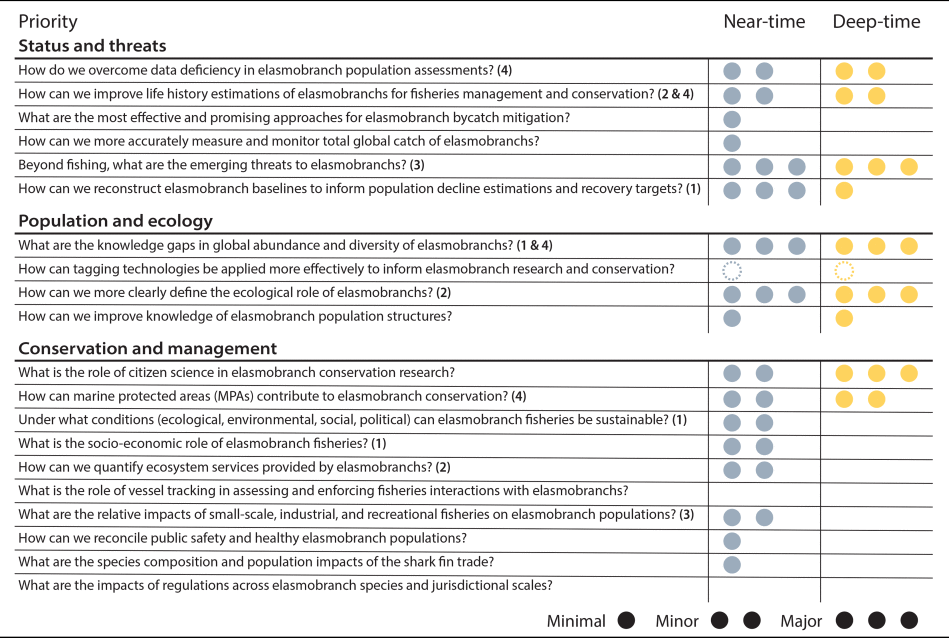
Here, we use the elasmobranch fossil record as an example to present research directions in which conservation paleobiology could progress to aid conservation in the coming decades. We share our perspectives as conservation paleobiologists working on elasmobranchs in the near- and deep-time fossil record. To frame our perspectives, we assessed the intersection between an expert-led list of current elasmobranch conservation priorities (Jorgensen et al. Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022) and available fossil and historical records (Table 1, Supplementary Material). Four broad topics emerged for which paleobiological research could address knowledge gaps in elasmobranch science and conservation: (1) baselines, (2) ecological roles, (3) threats, and (4) conservation priorities (referred to hereafter as Topics 1–4) (Fig. 3). We pose each as a question, discuss the potential of near- and deep-time records to contribute relevant information, and suggest how they could become more actionable. Our intent is to focus conservation paleobiology research agendas, encourage collaboration across timescales to inform elasmobranch conservation, and distill lessons that could be transferred to other threatened but understudied taxa in conservation paleobiology.
The potential of near- and deep-time geohistorical records to address priority questions in elasmobranch conservation identified by Jorgensen et al. (Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022). Three circles indicate a major contribution (e.g., relevant data are available and directly applicable); two circles indicate a minor contribution (e.g., some relevant data exist, but their application is less tangible or hindered by biases and/or mismatches in resolution); one circle indicates minimal contribution (e.g., few relevant data exist, but they could hypothetically contribute); and an empty cell indicates that no viable contribution is envisioned (e.g., no relevant data are available or the question is out of scope). A circle with a dotted outline indicates that the question could be reframed to incorporate geohistorical data but is not applicable as written. The bold numbers correspond to headings within the article (Topics 1–4) where examples are presented. See the Supplementary Material for the rationale behind each assessment.
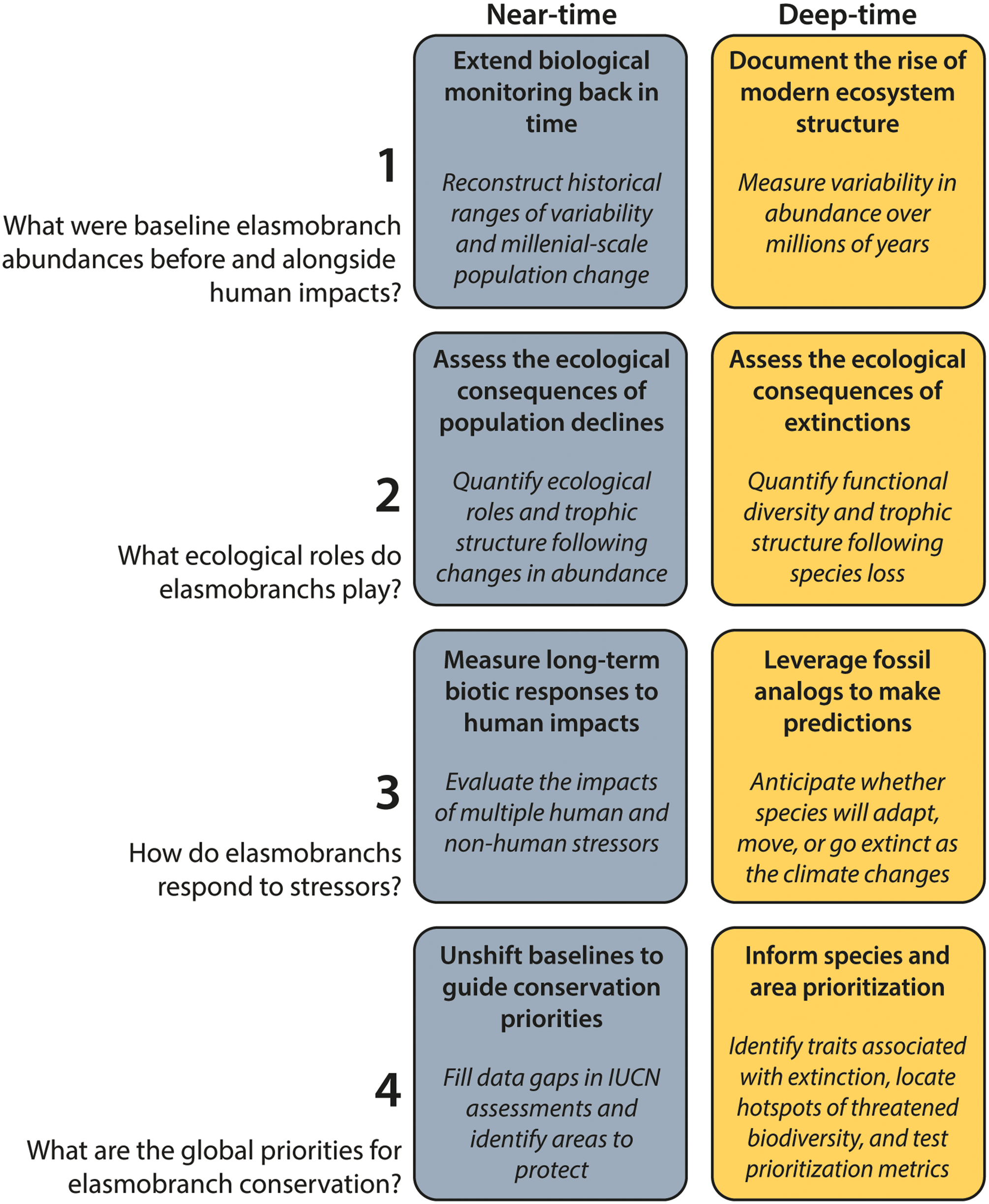
The elasmobranch fossil record offers a model to align conservation paleobiology research agendas with conservation. Research topics for which near- and deep-time geohistorical records have potential to address knowledge gaps in elasmobranch science and conservation include: (1) baselines, (2) ecological roles, (3) threats, and (4) conservation priorities. IUCN, International Union for Conservation of Nature.
Topic 1: What Were Baseline Elasmobranch Abundances before and alongside Human Impacts?
Elasmobranch populations are severely depleted across many ecosystems today (MacNeil et al. Reference MacNeil, Chapman, Heupel, Simpfendorfer, Heithaus, Meekan and Harvey2020; Pacoureau et al. Reference Pacoureau, Rigby, Kyne, Sherley, Winker, Carlson and Fordham2021; Simpfendorfer et al. Reference Simpfendorfer, Heithaus, Heupel, MacNeil, Meekan, Harvey, Sherman, Currey-Randall, Goetze and Kiszka2023). Yet systematic monitoring has only captured the recent history of elasmobranch exploitation, so historical baselines preceding human impacts are scarce (Jackson Reference Jackson1997; Jackson et al. Reference Jackson, Kirby, Berger, Bjorndal, Botsford, Bourque and Bradbury2001). These baselines define ranges of natural variability that provide benchmarks to measure the timing, magnitude, and drivers of ecological change over human timescales (Willis and Birks Reference Willis and Birks2006; Lotze and Worm Reference Lotze and Worm2009; Fig. 1C–G). Without accurate baselines, our understanding of elasmobranch ecology and conservation status can become skewed, leading to unrealistic or unambitious conservation targets (Lotze and Worm Reference Lotze and Worm2009; McClenachan et al. Reference McClenachan, Ferretti and Baum2012).
Elasmobranch biologists recognize the value of baselines (Heupel et al. Reference Heupel, Papastamatiou, Espinoza, Green and Simpfendorfer2019; Jorgensen et al. Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022; Table 1) and have employed creative approximations to compensate for the lack of historical data. Elasmobranch baselines have been inferred using space-for-time comparisons on remote islands (Sandin et al. Reference Sandin, Smith, DeMartini, Dinsdale, Donner, Friedlander and Konotchick2008; Bradley et al. Reference Bradley, Conklin, Papastamatiou, McCauley, Pollock, Pollock, Kendall, Gaines and Caselle2017), population models (Ferretti et al. Reference Ferretti, Curnick, Liu, Romanov and Block2018), model-estimated regional averages (MacNeil et al. Reference MacNeil, Chapman, Heupel, Simpfendorfer, Heithaus, Meekan and Harvey2020; Simpfendorfer et al. Reference Simpfendorfer, Heithaus, Heupel, MacNeil, Meekan, Harvey, Sherman, Currey-Randall, Goetze and Kiszka2023), and predictions based on environmental variables like primary productivity (Nadon et al. Reference Nadon, Baum, Williams, Mcpherson, Zgliczynski, Richards, Schroeder and Brainard2012; Valdivia et al. Reference Valdivia, Cox and Bruno2017; Fig. 4A). Available time-series data have also been collated in databases (e.g., Mull et al. Reference Mull, Pacoureau, Pardo, Ruiz, García-Rodríguez, Finucci and Haack2022). Collectively, these studies have mapped spatial variation in modern elasmobranch abundance to quantify depletion and understand the ecology of less disturbed systems.
Geohistorical and historical data can contextualize the extent of ongoing elasmobranch declines. A, Elasmobranch population trends are estimated using a variety of methods, including fisheries and monitoring data, historical and archaeological records, and near-time fossil records. A collection of available data from the Atlantic Ocean, including the Caribbean Sea—a region in which dramatic declines in shark abundance in the late twentieth century portended their dire conservation status—are compared here. These include: (1) the Living Planet Index calculated from abundance time-series data for 14 oceanic shark and ray species (Pacoureau et al. Reference Pacoureau, Rigby, Kyne, Sherley, Winker, Carlson and Fordham2021); (2) a comparison of longline fisheries catch rates for four oceanic shark species in the Gulf of Mexico from the 1950s and 1990s (Baum and Myers Reference Baum and Myers2004); (3) relative abundances of two resident shark species on Caribbean reefs recorded from Baited Remote Underwater Video Stations (BRUVS) compared with a model-predicted regional baseline (Simpfendorfer et al. Reference Simpfendorfer, Heithaus, Heupel, MacNeil, Meekan, Harvey, Sherman, Currey-Randall, Goetze and Kiszka2023); (4) perceived abundances of sharks inferred from archaeological, historical, ecological, and fisheries records in Caribbean Panama (Dillon et al. Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021); and 5) a comparison of shark dermal denticle accumulations from mid-Holocene and modern reefs in Caribbean Panama and the Dominican Republic (Dillon et al. Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021). These methods have different biases and temporal spans, with fishing impacts preceding most observational records. Baselines are either inferred from time-series data (1) or historical observations (2 and 4) when available, predicted using a model where all parameters are set those expected with no human impacts (3), or measured from fossil accumulations before major human impact (5). Each method reports large declines ranging from 46% to 79%, with some of the higher estimates produced when the baseline is extended farther back in time (although note that the data span multiple species and areas). B, In Caribbean Panama, BRUVS deployed from 2016 to 2019 overwhelmingly recorded nurse sharks (Ginglymostoma cirratum), a demersal reef-associated species (upper green bar; Chevis and Graham Reference Chevis and Graham2022). Dermal denticle accumulations sampled from modern (middle blue bar) and mid-Holocene (lower blue bar) reefs in the same area suggest that the current dominance of nurse sharks likely does not reflect the historical state of shark communities (Dillon et al. Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021). The shading indicates the relative abundances of pelagic (light), demersal (darker), and other (darkest) sharks in each record. Illustrations by Ashley Diedenhofen.

However, relying on modern survey data alone to shape expectations of elasmobranch baselines might be misleading given the ubiquity of human activities, which can overwrite the environmental drivers of elasmobranch abundance (MacNeil et al. Reference MacNeil, Chapman, Heupel, Simpfendorfer, Heithaus, Meekan and Harvey2020; Clementi et al. Reference Clementi, Babcock, Valentin-Albanese, Bond, Flowers, Heithaus and Whitman2021). Discrepancies also exist across survey methods, because elasmobranchs are challenging to count (McCauley et al. Reference McCauley, McLean, Bauer, Young and Micheli2012a). Geohistorical records can therefore complement ecological and fisheries surveys to track long-term population change. In deep time, fossils track the rise of modern ecosystem structure, whereas near-time records reconstruct historical variability over shorter time periods to contextualize modern elasmobranch communities (Fig. 3).
Over millions of years, fossils chronicle how elasmobranch abundances have shifted alongside their prey base, marking transitions between ecosystem states (Fig. 3). Relating these biological changes to major events in Earth's history can, in turn, evidence the environmental, climatic, and ecological factors that have shaped the emergence of ecosystems that we study and manage today (see Topics 3 and 4). For example, Sibert et al. (Reference Sibert, Norris, Cuevas and Graves2016) used fossil dermal denticles and fish teeth accumulating in deep-sea sediment cores to demonstrate that open-ocean ecosystems have been restructured multiple times over the last 85 Myr. Importantly, they observed a geologically abrupt disappearance of elasmobranchs at 19 Ma, which might offer insight into the eco-evolutionary implications of recent declines (Sibert and Rubin Reference Sibert and Rubin2021). Baseline carrying capacities of elasmobranchs today are the legacy of these past events, underscoring the value of measuring variability in deep time.
The near-time fossil record, alongside archaeological and historical records, can extend biological monitoring back in time to reconstruct elasmobranch baselines throughout human history and before extensive human impact (Fig. 3). For example, fossil dermal denticles accumulating in coral reef sediments have been used as a proxy for relative shark abundance to reconstruct pre-exploitation baselines and millennial-scale change along Panama's Caribbean coast (Dillon et al. Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021; Fig. 4A). When compared with monitoring data (Chevis and Graham Reference Chevis and Graham2022), these fossil assemblages suggest that the current dominance of demersal species like nurse sharks (Ginglymostoma cirratum) deviates from the historical state of shark communities in the area (Fig. 4B). Likewise, archaeological records (Fossile et al. Reference Fossile, Herbst, McGrath, Toso, Giannini, Milheira and Gilson2023) and cultural artifacts (Drew et al. Reference Drew, Philipp and Westneat2013) have revealed shifts in shark community composition through the lens of harvesting as well as demonstrated their socioeconomic significance as a food resource and cultural symbol (López de la Lama et al. Reference López de la Lama, Puente, Sueiro and Chan2021). Over more recent timescales, historical records—including photographs, archival landings data, and local ecological knowledge—have documented elasmobranch population vulnerability and trajectories before industrial fishing (e.g., Ferretti et al. Reference Ferretti, Myers, Serena and Lotze2008; McClenachan Reference McClenachan2009; Bom et al. Reference Bom, van de Water, Camphuysen, van der Veer and van Leeuwen2020; Martínez-Candelas et al. Reference Martínez-Candelas, Pérez-Jiménez, Espinoza-Tenorio, McClenachan and Méndez-Loeza2020; Herbst et al. Reference Herbst, Rampon, Baleeiro, Silva, Fossile and Colonese2023). These examples demonstrate how historical elasmobranch baselines can be pieced together using a variety of methods to fill an established knowledge gap. However, this information is not yet commonly applied in elasmobranch conservation.
What makes a baseline actionable? We see at least three avenues for increasing the salience of historical baselines in elasmobranch conservation. First, because baselines vary with environmental context (Valdivia et al. Reference Valdivia, Cox and Bruno2017), geohistorical records could be collected from additional regions, habitats, and time points to inform local conservation targets and model the biophysical drivers of natural variability. Additionally, elasmobranch populations could be reconstructed during cultural periods with different fishing intensities or management strategies to track their responses to human activities over millennia (see Topic 3). This work is being facilitated by the development of new methods, such as lab protocols to access lesser-known dermal denticle records (Dillon et al. Reference Dillon, Norris and O'Dea2017; Sibert et al. Reference Sibert, Cramer, Hastings and Norris2017), machine learning to classify fossil material (Mimura et al. Reference Mimura, Nakamura, Yasukawa, Sibert, Ohta, Kitazawa and Kato2023), paleoecological time-series analyses (Simpson Reference Simpson2018; Mottl et al. Reference Mottl, Grytnes, Seddon, Steinbauer, Bhatta, Felde, Flantua and Birks2020), and time-series databases (Smith et al. Reference Smith, Rillo, Kocsis, Dornelas, Fastovich, Huang and Jonkers2023). As data accumulate, a next step could include developing data synthesis workflows akin to those for other fossil assemblages (e.g., fossil pollen; Flantua et al. Reference Flantua, Mottl, Felde, Bhatta, Birks, Grytnes, Seddon and Birks2023).
Second, baselines derived from geohistorical records could be reported using formats compatible with modern data to create a common currency for translating changes in elasmobranch populations across timescales. For example, Rodrigues et al. (Reference Rodrigues, Monsarrat, Charpentier, Brooks, Hoffmann, Reeves, Palomares and Turvey2019) proposed a framework for comparing modern species’ abundances to historical reference points to quantify long-term human impacts. Their classification system consisted of categories defined by percentage change intervals relative to a baseline before major human impact, enabling standardized comparisons despite patchy data. To deal with uncertainty when mobilizing diverse datasets, they specified the likelihood of each category and provided conservative estimates of the magnitude of change (Rodrigues et al. Reference Rodrigues, Monsarrat, Charpentier, Brooks, Hoffmann, Reeves, Palomares and Turvey2019). Communicating uncertainty around baseline estimates is critical to build trust in the methods, clarify their biases, and propagate the effects of that uncertainty into downstream conservation decisions (Dietl Reference Dietl2019; Cooke et al. Reference Cooke, Rytwinski, Taylor, Nyboer, Nguyen, Bennett and Young2020; White et al. Reference White, Stoffels and Whitehead2023).
Series of paleobiological data measured in the same units are more readily compared. The magnitude of change from a baseline could be calculated as a percentage or effect size along with confidence intervals, sample ages and chronological uncertainty, and either sensitivity analyses or models that account for the loss of skeletal material through taphonomic and depositional processes (e.g., Tomašových and Kidwell Reference Tomašových and Kidwell2017; Kiessling et al. Reference Kiessling, Smith and Raja2023; Tomašových et al. Reference Tomašových, Dominici, Nawrot and Zuschin2023). For example, Dillon et al. (Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021) compared both absolute and relative dermal denticle abundances across time points and sampling locations to characterize elasmobranch community change. Denticle accumulations can also be compared with contemporaneous records (e.g., fish teeth or otoliths) after accounting for differences in taphonomy and production rates (Sibert et al. Reference Sibert, Norris, Cuevas and Graves2016, Reference Sibert, Cramer, Hastings and Norris2017). Converting denticle accumulations into absolute shark densities poses a greater challenge, as denticle abundances are affected by depositional, taphonomic, and biological processes. At a minimum, information about denticle densities and shedding rates, shark body sizes, sedimentation rates, and taphonomic alteration would be needed to produce such estimates (Sibert et al. Reference Sibert, Cramer, Hastings and Norris2017; Dillon et al. Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021, Reference Dillon, Bagla, Plioplys, McCauley, Lafferty and O'Dea2022a). In the absence of this information, ground-truthing studies have attempted to correlate elasmobranch fossil accumulations (e.g., dermal denticles) with modern surveys (e.g., shark densities) in wild (Dillon et al. Reference Dillon, Lafferty, McCauley, Bradley, Norris, Caselle, DiRenzo, Gardner and O'Dea2020) and aquarium (Dillon et al. Reference Dillon, Bagla, Plioplys, McCauley, Lafferty and O'Dea2022a) settings to evaluate their ecological fidelity.
Finally, baselines should be framed in terms of conservation needs. This process begins with investing in partnerships with conservation practitioners to ensure that the end goal, users, and timeline of a conservation need inform data collection strategies (Beier et al. Reference Beier, Hansen, Helbrecht and Behar2017; Cooke et al. Reference Cooke, Rytwinski, Taylor, Nyboer, Nguyen, Bennett and Young2020; Dietl et al. Reference Dietl, Durham, Clark and Prado2023). When building collaborations, partners could discuss how a baseline is selected and what success might look like in relation to that baseline (Campbell et al. Reference Campbell, Gray, Hazen and Shackeroff2009; Redford et al. Reference Redford, Amato, Baillie, Beldomenico, Bennett, Clum and Cook2011; MacKeracher et al. Reference MacKeracher, Diedrich and Simpfendorfer2019; Cooke et al. Reference Cooke, Rytwinski, Taylor, Nyboer, Nguyen, Bennett and Young2020), consider the interplay between baselines and human cultural practices (Simpfendorfer et al. Reference Simpfendorfer, Heupel and Kendal2021; Hoel et al. Reference Hoel, Chin and Lau2022), and design multiple anticipatory recovery goals that situate a baseline in its current sociopolitical context (Dietl Reference Dietl2019; Ingeman et al. Reference Ingeman, Samhouri and Stier2019). These conversations are important given the ecological and political challenges of recovering large predators (Marshall et al. Reference Marshall, Stier, Samhouri, Kelly and Ward2016; Stier et al. Reference Stier, Samhouri, Novak, Marshall, Ward, Holt and Levin2016; Ingeman et al. Reference Ingeman, Zhao, Wolf, Williams, Ritger, Ripple and Kopecky2022) and the potential for human–wildlife conflict if recovery is successful (Carlson et al. Reference Carlson, Heupel, Young, Cramp and Simpfendorfer2019). In addition to sourcing research questions from practitioners, historical baselines could help iteratively shape elasmobranch conservation priorities by flagging populations that are either vulnerable or have high recovery potential (see Topic 4). Baselines might also uncover elasmobranch populations that have resisted decline, thus creating opportunities to learn from these “bright spots” (Lotze et al. Reference Lotze, Coll, Magera, Ward-Paige and Airoldi2011; O'Dea et al. Reference O'Dea, Dillon, Altieri and Lepore2017; Ingeman et al. Reference Ingeman, Zhao, Wolf, Williams, Ritger, Ripple and Kopecky2022). In sum, geohistorical records offer an exciting research avenue to contextualize recent elasmobranch population change and retroactively fill monitoring gaps, especially when integrated with modern datasets in conservation assessments.
Topic 2: What Ecological Roles Do Elasmobranchs Play?
When organisms are lost from an ecosystem—either through population declines, extirpations, or extinctions—their ecological functions can also be lost. Conservation efforts have traditionally focused on species diversity, but there is a growing interest in preserving species’ contributions to ecosystem processes (Soulé et al. Reference Soulé, Estes, Berger and Del Rio2003; Sanderson Reference Sanderson2006; Akçakaya et al. Reference Akçakaya, Rodrigues, Keith, Milner-Gulland, Sanderson, Hedges and Mallon2020). One way to measure these contributions is through species’ ecological traits, such as body size, diet, and mobility (Petchey and Gaston Reference Petchey and Gaston2002; Villéger et al. Reference Villéger, Mason and Mouillot2008; Gagic et al. Reference Gagic, Bartomeus, Jonsson, Taylor, Winqvist, Fischer and Slade2015). Ecological traits reflect how energy or other resources are assimilated and moved across ecosystems (Done et al. Reference Done, Ogden, Weibe, Rosen, Mooney, Cushman, Medina, Sala and Schulze1996; Bellwood et al. Reference Bellwood, Streit, Brandl and Tebbett2019). These traits, in turn, constitute the functional diversity of a community or assemblage, apart from its taxonomic composition. Functional diversity has been measured in both modern and fossil assemblages to quantify their responses to disturbances (Mouillot et al. Reference Mouillot, Graham, Villéger, Mason and Bellwood2013), identify functions that disappear or emerge when species’ configurations shift (Graham et al. Reference Graham, Cinner, Norström and Nyström2014; Pimiento et al. Reference Pimiento, Griffin, Clements, Silvestro, Varela, Uhen and Jaramillo2017; Bellwood et al. Reference Bellwood, Streit, Brandl and Tebbett2019; Fig. 1D), and predict future ecosystem states using traits as proxies (Streit and Bellwood Reference Streit and Bellwood2022).
Elasmobranchs are at greater risk of losing functional diversity than any other marine megafauna group under simulated extinction scenarios, likely as a result of their threatened status and because species with extreme trait combinations are selectively fished (Pimiento et al. Reference Pimiento, Leprieur, Silvestro, Lefcheck, Albouy, Rasher, Davis, Svenning and Griffin2020). Indeed, sharks are already thought to be “functionally extinct” in some areas where their numbers are too low to maintain their ecological functions (Jackson et al. Reference Jackson, Kirby, Berger, Bjorndal, Botsford, Bourque and Bradbury2001; MacNeil et al. Reference MacNeil, Chapman, Heupel, Simpfendorfer, Heithaus, Meekan and Harvey2020). However, the ecological consequences of elasmobranch declines are still being unraveled (Stevens Reference Stevens2000; Heithaus et al. Reference Heithaus, Frid, Wirsing and Worm2008, Reference Heithaus, Dunn, Farabaugh, Lester, Madin, Meekan, Papastamatiou, Roff, Vaudo, Wirsing, Carrier, Simpfendorfer, Heithaus and Yopak2022; Ferretti et al. Reference Ferretti, Worm, Britten, Heithaus and Lotze2010; Roff et al. Reference Roff, Doropoulos, Rogers, Bozec, Krueck, Aurellado, Priest, Birrell and Mumby2016), and their functional diversity is often overlooked in current conservation priorities (Pimiento et al. Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023).
These knowledge gaps arise because we are still learning about elasmobranchs’ ecological roles as they are concurrently being eliminated or modified by human activities (Table 1). For example, we know that great sharks (i.e., large apex predators) structure food webs through direct predation (Ferretti et al. Reference Ferretti, Worm, Britten, Heithaus and Lotze2010; Heupel et al. Reference Heupel, Knip, Simpfendorfer and Dulvy2014; Fig. 2A). However, whether these top-down effects cascade to lower trophic levels is still debated, particularly in complex ecosystems like coral reefs (Bascompte et al. Reference Bascompte, Melián and Sala2005; Estes et al. Reference Estes, Terborgh, Brashares, Power, Berger, Bond and Carpenter2011; Frisch et al. Reference Frisch, Ireland, Rizzari, Lönnstedt, Magnenat, Mirbach and Hobbs2016; Roff et al. Reference Roff, Doropoulos, Rogers, Bozec, Krueck, Aurellado, Priest, Birrell and Mumby2016; Casey et al. Reference Casey, Baird, Brandl, Hoogenboom, Rizzari, Frisch, Mirbach and Connolly2017; Desbiens et al. Reference Desbiens, Roff, Robbins, Taylor, Castro-Sanguino, Dempsey and Mumby2021). Because historical overfishing depleted large apex sharks (Jackson et al. Reference Jackson, Kirby, Berger, Bjorndal, Botsford, Bourque and Bradbury2001), it is unclear whether the lack of robust evidence stems from food web properties (e.g., omnivory, functional redundancy, or diffuse predation), buffering by bottom-up processes (e.g., environmental variation controlling prey availability), or potential confounds (e.g., simultaneous fishing of predators and prey), or alternatively, whether trophic cascades occurred before biological monitoring (Roff et al. Reference Roff, Doropoulos, Rogers, Bozec, Krueck, Aurellado, Priest, Birrell and Mumby2016). Modern ecological studies are typically limited to testing these hypotheses after apex predators have already been removed from food webs, and they sometimes arrive at different conclusions (Heithaus et al. Reference Heithaus, Dunn, Farabaugh, Lester, Madin, Meekan, Papastamatiou, Roff, Vaudo, Wirsing, Carrier, Simpfendorfer, Heithaus and Yopak2022).
Elasmobranchs are more than just predators. They alter prey behavior (Heithaus et al. Reference Heithaus, Frid, Wirsing and Worm2008; Sherman et al. Reference Sherman, Heupel, Moore, Chin and Simpfendorfer2020), compete with or create feeding opportunities for other species (Papastamatiou et al. Reference Papastamatiou, Wetherbee, Lowe and Crow2006; Oliver et al. Reference Oliver, Hussey, Turner and Beckett2011), energetically connect resource pools across habitats (McCauley et al. Reference McCauley, Young, Dunbar, Estes, Semmens and Micheli2012b), transport nutrients (Williams et al. Reference Williams, Papastamatiou, Caselle, Bradley and Jacoby2018), and are prey themselves (Ford et al. Reference Ford, Ellis, Matkin, Wetklo, Barrett-Lennard and Withler2011; Mourier et al. Reference Mourier, Planes and Buray2013). These lesser-studied functions are presumed to promote ecosystem health, yet the mechanistic linkages remain untested or are similarly predicated on a shifted baseline (Roff et al. Reference Roff, Doropoulos, Rogers, Bozec, Krueck, Aurellado, Priest, Birrell and Mumby2016; Jorgensen et al. Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022).
For each of these cases, recent methodological developments have improved our ability to extract relevant ecological information from the elasmobranch fossil record. We discuss three approaches here: (1) traits inferred from morphology, (2) biomechanical reconstructions, and (3) geochemistry. These approaches can be used in conjunction with occurrence data to reconstruct elasmobranchs’ ecological roles, biotic interactions, and functional diversity in both near and deep time. Near-time studies typically apply these methods to detect the ecological consequences of elasmobranch population change following fishing, whereas deep-time studies aim to infer the trophic ecology of extinct species and assess how disturbances like past extinction events alter food web structure and functional diversity (Fig. 3).
Traits Inferred from Morphology
Some ecological traits can be inferred from shark tooth morphology (e.g., body size, prey preference, and feeding mechanism; Fig. 2G), although the associations between tooth measurements and functional traits are not always one to one (Frazzetta Reference Frazzetta1988; Ciampaglio et al. Reference Ciampaglio, Wray and Corliss2005; Cooper et al. Reference Cooper, Griffin, Kindlimann and Pimiento2023). Because shark teeth can be identified to species (Naylor and Marcus Reference Naylor and Marcus1994; Cappetta Reference Cappetta and Schultze2012; Paillard et al. Reference Paillard, Shimada and Pimiento2021), biological processes related to these traits can be explored over microevolutionary scales. Shark dermal denticles also encode biological information (e.g., body size, mobility, position in the water column, schooling behavior, and bioluminescence), although they are less taxonomically resolved than teeth (Reif Reference Reif1985; Raschi and Tabit Reference Raschi and Tabit1992; Dillon et al. Reference Dillon, Norris and O'Dea2017; Ferrón and Botella Reference Ferrón and Botella2017; Ferrón and Palacios-Abella Reference Ferrón and Palacios-Abella2022). Additionally, elasmobranch vertebral rings preserve life-history traits such as growth rates and age (Daiber Reference Daiber1960; Shimada Reference Shimada2008). Together, these traits reflect functional ecology, as they relate to how species use resources (Tavares et al. Reference Tavares, Moura, Acevedo-Trejos and Merico2019). For example, analyses of fossil shark tooth morphology across the Cretaceous/Paleogene (K/Pg) mass extinction documented a reduction in the body size and ecological diversity of Lamniformes (Belben et al. Reference Belben, Underwood, Johanson and Twitchett2017) and a proliferation of similar tooth morphologies within Carcharhiniformes (Bazzi et al. Reference Bazzi, Kear, Blom, Ahlberg and Campione2018), suggesting morphological turnover. Other studies use traits to constrain inferences from food web networks. A. Shipley et al. (Reference Shipley, Aze, Pimiento, Beckerman, Dunne, Shaw and Dunhill2023) incorporated traits into a metacommunity web to model how trophic dynamics shifted after Otodus megalodon went extinct at the end of the Pliocene, foreshadowing the potential ecological consequences of losing extant apex predators like white sharks (Carcharodon carcharias). Beyond measuring individual traits, shark teeth and dermal denticles can be ascribed to functional groups based on their morphologies, which correspond with ecological life modes (Reif Reference Reif1985; Cappetta Reference Cappetta1986; Dillon et al. Reference Dillon, Norris and O'Dea2017; Ferrón and Botella Reference Ferrón and Botella2017) and can be used to infer the ecological structure of shark communities (Dillon et al. Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021; Fig. 4B).
Biomechanical Reconstructions
When interpreting traits based on morphology, it is important to establish the relationship between each trait and its ecological function (Streit and Bellwood Reference Streit and Bellwood2022; Brown et al. Reference Brown, Bunting, Carvalho, de Bello, Mander, Marcisz, Mottl, Reitalu and Svenning2023). One way to test hypothesized relationships between form and function is through biomechanical reconstructions. Much of this work for elasmobranchs has focused on locomotion and feeding kinematics (Motta and Wilga Reference Motta, Wilga, Tricas and Gruber2001; Dean and Bhushan Reference Dean and Bhushan2010). Dermal denticles’ hydrodynamic properties have been experimentally tested using pieces of shark skin (Oeffner and Lauder Reference Oeffner and Lauder2012; Afroz et al. Reference Afroz, Lang, Habegger, Motta and Hueter2016), 3D printed biomimetic foils (Wen et al. Reference Wen, Weaver and Lauder2014; Lauder et al. Reference Lauder, Wainwright, Domel, Weaver, Wen and Bertoldi2016; Domel et al. Reference Domel, Domel, Weaver, Saadat, Bertoldi and Lauder2018), or simplified replicas (Bechert et al. Reference Bechert, Bruse and Hage2000; Lang et al. Reference Lang, Motta, Hidalgo and Westcott2008). Computational fluid dynamics models have also been applied to simulate water flow over denticles (Díez et al. Reference Díez, Soto and Blanco2015). These biomechanical studies support the placement of denticles into functional groups. In contrast, biomechanical tests of shark tooth performance show that some tooth morphotypes used to infer diet are hazy (Whitenack and Motta Reference Whitenack and Motta2010; Corn et al. Reference Corn, Farina, Brash and Summers2016; Ballell and Ferrón Reference Ballell and Ferrón2021), motivating reassessments of their value as ecological proxies to determine which tooth measurements are most informative (Cooper et al. Reference Cooper, Griffin, Kindlimann and Pimiento2023). Collectively, this work has refined our understanding of the functional significance of shark tooth and dermal denticle morphology, allowing insights to be extended back in time. Additional promise lies in applying these methods to fossil morphologies that lack extant analogues.
Geochemistry
Geochemical approaches such as stable isotope analysis augment what we can learn from fossil morphology. Notably, they have revolutionized studies of elasmobranch diet and habitat use (Vennemann et al. Reference Vennemann, Hegner, Cliff and Benz2001; Boecklen et al. Reference Boecklen, Yarnes, Cook and James2011; Hussey et al. Reference Hussey, MacNeil, Olin, McMeans, Kinney, Chapman and Fisk2012; Kim and Koch Reference Kim and Koch2012). Elasmobranch diet in the fossil record was traditionally deduced from either tooth morphology (e.g., Cappetta Reference Cappetta1986) or from infrequent bite marks, coprolites, or preserved stomach contents (e.g., Aguilera and de Aguilera Reference Aguilera and de Aguilera2004; Benites-Palomino et al. Reference Benites-Palomino, Velez-Juarbe, Altamirano-Sierra, Collareta, Carrillo-Briceño and Urbina2022). Minute signatures of enameloid-bound nitrogen isotopes (δ15N) can now be measured in fossil shark teeth using the oxidation–denitrifer method (Kast et al. Reference Kast, Griffiths, Kim, Rao, Shimada, Becker and Maisch2022), which has begun to reveal the diet, trophic position, and energetic requirements of ancient sharks. In parallel, bulk nitrogen (δ15N) and carbon (δ13C) isotopes have been extracted from collagen in teeth sampled from living sharks (Polo-Silva et al. Reference Polo-Silva, Galván-Magaña and Delgado-Huertas2012; O. N. Shipley et al. Reference Shipley, Henkes, Gelsleichter, Morgan, Schneider, Talwar and Frisk2021, Reference Shipley, Matich, Hussey, Brooks, Chapman, Frisk and Guttridge2023) and zooarchaeological remains (Burg Mayer and de Freitas Reference Burg Mayer and de Freitas2023) to document resource use and connectivity—highlighting conceptual overlap across timescales. Other promising trophic indicators include zinc isotopes (δ66Zn; McCormack et al. Reference McCormack, Griffiths, Kim, Shimada, Karnes, Maisch and Pederzani2022, Reference McCormack, Karnes, Haulsee, Fox and Kim2023) and calcium isotopes (δ44/42Ca; Martin et al. Reference Martin, Tacail, Adnet, Girard and Balter2015; Assemat et al. Reference Assemat, Adnet, Bayez, Hassler, Arnaud-Godet, Mollen, Girard and Martin2022). Nitrogen and zinc isotope analyses have provided new insights into the trophic evolution of apex predators, suggesting that large Cenozoic megatooth sharks (genus Otodus) reached higher trophic positions than any extant marine species (Kast et al. Reference Kast, Griffiths, Kim, Rao, Shimada, Becker and Maisch2022) and/or potentially competed for resources with C. carcharias (McCormack et al. Reference McCormack, Griffiths, Kim, Shimada, Karnes, Maisch and Pederzani2022). Additionally, stable isotopes can trace elasmobranch movement across isotopically distinct waterbodies (i.e., δ18O and 87Sr/86Sr; Fischer et al. Reference Fischer, Schneider, Voigt, Joachimski, Tichomirowa, Tütken, Götze and Berner2013). As these isotope systems are applied to ever-smaller fossils with increasing precision, it could be fruitful to characterize trophic niches using multiple complementary isotopes (Cybulski et al. Reference Cybulski, Skinner, Wan, Wong, Toonen, Gaither, Soong, Wyatt and Baker2022; Lüdecke et al. Reference Lüdecke, Leichliter, Aldeias, Bamford, Biro, Braun and Capelli2022) and to track changes in trophic niches across disturbance events.
How can we better leverage ecological data preserved in the elasmobranch fossil record to anticipate and manage their future functional ecology? As we have shown, ecological traits convey information about ecosystem functioning that can be translated across timescales (Fig. 3). Consequently, opportunity lies in combining fossil and modern trait datasets (Brown et al. Reference Brown, Bunting, Carvalho, de Bello, Mander, Marcisz, Mottl, Reitalu and Svenning2023). One path forward could be to develop a conceptual framework that unites elasmobranch functional ecology through time. Such a framework could establish best practices for data collection, archiving in online trait databases, and reporting. To encourage collaboration across potential users, this framework could be codeveloped by paleobiologists, biologists, and conservationists asking similar trait-based questions over varying temporal and spatial scales. Ultimately, if conservation success is framed as restoring functionally viable populations (Akçakaya et al. Reference Akçakaya, Rodrigues, Keith, Milner-Gulland, Sanderson, Hedges and Mallon2020), then information about past ecosystem processes could play a role in setting and measuring progress toward recovery goals.
Topic 3: How Do Elasmobranchs Respond to Stressors?
Understanding how species respond to threats is important for guiding conservation strategies. Elasmobranchs provide a useful case study for gleaning conservation lessons from a lineage's past failures and successes. Elasmobranchs and their relatives have endured major extinction events and weathered episodes of global cooling and warming in the geological past (Kriwet et al. Reference Kriwet, Kiessling and Klug2008; Whitenack et al. Reference Whitenack, Kim, Sibert, Carrier, Simpfendorfer, Heithaus and Yopak2022), marking millions of years of stability punctuated by pronounced change (Sibert et al. Reference Sibert, Norris, Cuevas and Graves2016). Elasmobranchs now face a new threat. Over mere decades, overfishing has pushed their populations to the brink of collapse (Ward and Myers Reference Ward and Myers2005; Dulvy et al. Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021; Pacoureau et al. Reference Pacoureau, Rigby, Kyne, Sherley, Winker, Carlson and Fordham2021), and its effects are compounded by habitat degradation, pollution, and climate change (Dulvy et al. Reference Dulvy, Fowler, Musick, Cavanagh, Kyne, Harrison and Carlson2014; Sherman et al. Reference Sherman, Simpfendorfer, Pacoureau, Matsushiba, Yan, Walls and Rigby2023).
However, key questions remain about how these anthropogenic and environmental stressors affect elasmobranchs (Table 1). We have more to learn about how climate change will interact with other stressors to impact elasmobranch populations (Chin et al. Reference Chin, Kyne, Walker and McAuley2010; Rummer et al. Reference Rummer, Bouyoucos, Wheeler, Santos, Rosa, Carrier, Simpfendorfer, Heithaus and Yopak2022), where to focus management efforts as their ranges shift (Dulvy et al. Reference Dulvy, Simpfendorfer, Davidson, Fordham, Bräutigam, Sant and Welch2017; Tanaka et al. Reference Tanaka, Van Houtan, Mailander, Dias, Galginaitis, O'Sullivan, Lowe and Jorgensen2021; Diaz-Carballido et al. Reference Diaz-Carballido, Mendoza-González, Yañez-Arenas and Chiappa-Carrara2022), and which species are most sensitive (Jorgensen et al. Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022). Because elasmobranchs are large and mobile, it is generally not feasible to manipulate them in real time, hindering mechanistic insight into how stressors control their populations. Instead, the fossil record spans a range of scenarios that can be leveraged to study how elasmobranchs responded to combinations of stressors in the past and, in turn, assess their susceptibility to future change. Because threats to elasmobranch populations operate over different timescales, both near- and deep-time records are pertinent.
Geohistorical records spanning centuries to millennia demonstrate elasmobranchs’ responses to long-term anthropogenic stressors such as fishing and habitat loss. These records can help evaluate the relative impacts of multiple human and nonhuman stressors and identify conservation actions with the biggest potential to mitigate their effects (Fig. 3). For example, Dillon et al. (Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021) documented a 71% decline in shark dermal denticle accumulation since the mid-Holocene in Caribbean Panama, suggesting a significant drop in shark abundance (Fig. 4A; see Topic 1). To better understand the mechanisms, they examined differences in the amount of decline across denticle functional groups and found that commercially valuable sharks were preferentially affected, implicating fishing as the dominant cause (Fig. 4B). Yet they also observed declines in sharks that are infrequently fished, evidencing the additional contribution of indirect human pressures such as habitat degradation (Dillon et al. Reference Dillon, McCauley, Morales-Saldaña, Leonard, Zhao and O'Dea2021). Other examples come from historical ecology, where studies have aligned time series of elasmobranch abundance derived from archival sources with putative stressors in locations such as Hawai'i (Kittinger et al. Reference Kittinger, Pandolfi, Blodgett, Hunt, Jiang, Maly, McClenachan, Schultz and Wilcox2011) and the Adriatic Sea (Fortibuoni et al. Reference Fortibuoni, Libralato, Raicevich, Giovanardi and Solidoro2010). By comparing the timing, direction, and magnitude of ecological change with cultural events in each location, these studies disentangled the relative contributions of different human stressors (e.g., fishing vs. habitat degradation vs. climate change) and often showed a progression of impacts starting with historical small-scale harvesting (a pattern documented more broadly by Jackson et al. [Reference Jackson2001], Pandolfi et al. [Reference Pandolfi, Bradbury, Sala, Hughes, Bjorndal, Cooke and McArdle2003], and Lotze et al. [Reference Lotze, Lenihan, Bourque, Bradbury, Cooke, Kay, Kidwell, Kirby, Peterson and Jackson2006]). Additional insight could be gained from pairing these studies with modern surveys to sharpen their temporal resolution given the fast pace of elasmobranch population declines and conservation action.
Deep-time geohistorical records are well suited to addressing elasmobranchs’ responses to climate change. Over millions of years, the fossil record offers valuable parallels for future projected environmental conditions that have not yet been experienced in human history (Fig. 3). Earth is heading toward a climate similar to the Paleocene–Eocene thermal maximum (56 Ma) (Burke et al. Reference Burke, Williams, Chandler, Haywood, Lunt and Otto-Bliesner2018; Fig. 1A) and an extinction crisis not seen since the K/Pg mass extinction (66 Ma) (Barnosky et al. Reference Barnosky, Matzke, Tomiya, Wogan, Swartz, Quental and Marshall2011; Ceballos et al. Reference Ceballos, Ehrlich, Barnosky, García, Pringle and Palmer2015). Although these protracted time frames might appear juxtaposed with conservation's forward-facing gaze (Dietl et al. Reference Dietl, Smith and Durham2019), they foreshadow how species might respond to future climate change based on how they fared during past episodes of rapid global warming. Natural experiments in the fossil record could anticipate elasmobranch range shifts in response to climate perturbations or describe refugia to guide adaptive spatial management (see Topic 4). For example, Villafaña and Rivadeneira (Reference Villafaña and Rivadeneira2018) tested how ecological and life-history traits modulated elasmobranch distributions following environmental change during the Neogene. The fossil record could also reveal the drivers of elasmobranch diversity dynamics. For example, Condamine et al. (Reference Condamine, Romieu and Guinot2019) found that Lamniformes experienced a significant decline in diversity over the last 20 Myr due to climatic cooling and competition with other clades. Although the climatic stressors faced by elasmobranchs today differ (i.e., a warming, acidified, and deoxygenating ocean), this study illustrates how diversity covaries with abiotic and biotic factors through deep time. These examples demonstrate how deep-time analogues can be leveraged to anticipate vulnerability in extant species and plan for resilience under future climatic conditions.
Topic 4: What Are the Global Priorities for Elasmobranch Conservation?
Current efforts to halt extinction typically use prioritization tools that identify species and areas that are in most need of protection (Arponen Reference Arponen2012; Butchart et al. Reference Butchart, Scharlemann, Evans, Quader, Aricò, Arinaitwe and Balman2012). These priorities exist because extinctions do not occur randomly on the tree of life (Wang and Bush Reference Wang and Bush2008) and because resources to protect biodiversity are limited (Murdoch et al. Reference Murdoch, Polasky, Wilson, Possingham, Kareiva and Shaw2007). The International Union for Conservation of Nature (IUCN) Red List of Threatened Species provides a robust framework to categorize species according to their extinction risk and is widely used for conservation prioritization (Mace and Lande Reference Mace and Lande1991; Rodrigues et al. Reference Rodrigues, Pilgrim, Lamoreux, Hoffmann and Brooks2006; Hoffmann et al. Reference Hoffmann, Brooks, da Fonseca, Gascon, Hawkins, James and Langhammer2008; Mace et al. Reference Mace, Collar, Gaston, Hilton-Taylor, Akçakaya, Leader-Williams, Milner-Gulland and Stuart2008; Betts et al. Reference Betts, Young, Hilton-Taylor, Hoffmann, Rodríguez, Stuart and Milner-Gulland2020). IUCN statuses have also been combined with various dimensions of biodiversity (e.g., phylogenetic and functional) in prioritization metrics to identify endangered species for which extinction will result in irreplaceable losses of evolutionary history or ecological functions. These metrics include EDGE (Evolutionarily Distinct and Globally Endangered; Isaac et al. Reference Isaac, Turvey, Collen, Waterman and Baillie2007) and FUSE (Functionally Unique, Specialized, and Endangered; Pimiento et al. Reference Pimiento, Leprieur, Silvestro, Lefcheck, Albouy, Rasher, Davis, Svenning and Griffin2020) (for a complete list, see Pimiento and Antonelli Reference Pimiento and Antonelli2022). The EDGE metric prioritizes evolutionary history by combining species’ IUCN statuses with a phylogeny-based calculation of their evolutionary distinctiveness (Isaac et al. Reference Isaac, Turvey, Collen, Waterman and Baillie2007), whereas the FUSE metric prioritizes species’ contributions to functional diversity by combining their IUCN statuses with a trait-based calculation of their functional uniqueness and specialization (Pimiento et al. Reference Pimiento, Leprieur, Silvestro, Lefcheck, Albouy, Rasher, Davis, Svenning and Griffin2020). These data-driven prioritizations can then be fed into policy and management decisions such as the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES).
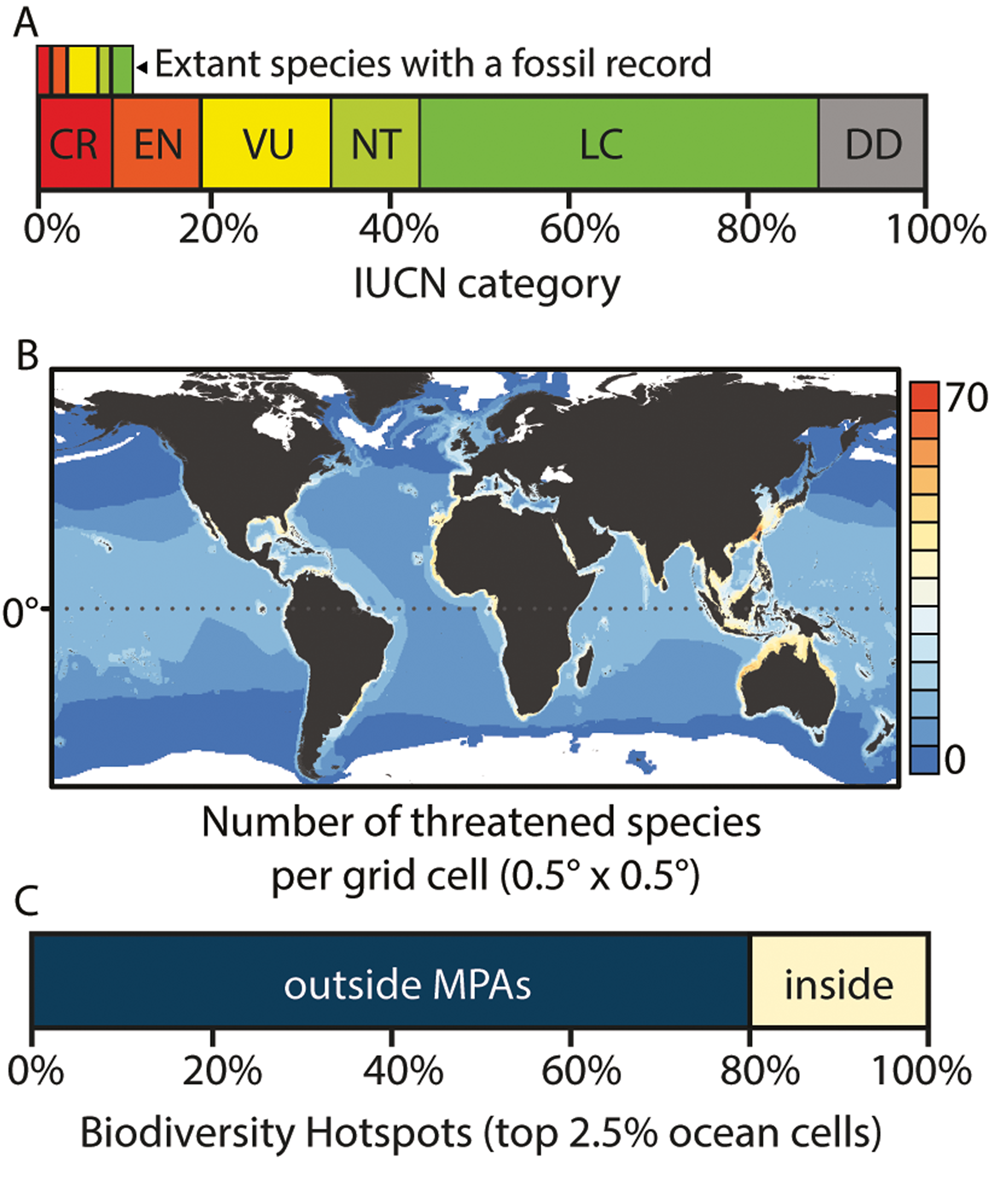
According to the IUCN, 37% of elasmobranch species are at risk of extinction (Fig. 5A), making them the most threatened marine vertebrate group today (Dulvy et al. Reference Dulvy, Fowler, Musick, Cavanagh, Kyne, Harrison and Carlson2014, Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021). Threatened elasmobranch species are both evolutionarily (Stein et al. Reference Stein, Mull, Kuhn, Aschliman, Davidson, Joy, Smith, Dulvy and Mooers2018) and functionally (Pimiento et al. Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023) distinct. However, the EDGE and FUSE metrics highlight different elasmobranch species, demonstrating the value of considering multiple metrics (Pimiento et al. Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023). Additionally, prioritization tools have been used to identify hotspots of threatened elasmobranch biodiversity (Fig. 5B) as well as to assess the spatial congruence between different dimensions of their biodiversity (Lucifora et al. Reference Lucifora, García and Worm2011; Stein et al. Reference Stein, Mull, Kuhn, Aschliman, Davidson, Joy, Smith, Dulvy and Mooers2018; Derrick et al. Reference Derrick, Cheok and Dulvy2020; Pimiento et al. Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023). Although some hotspots overlap, functional diversity appears to have a distinct spatial fingerprint (Pimiento et al. Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023). Importantly, many biodiversity hotspots coincide with industrial fishing, and most fall outside the existing marine protected area network (Davidson and Dulvy Reference Davidson and Dulvy2017; Pimiento et al. Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023; Fig. 5C). Together, these studies underscore the urgency to protect elasmobranchs (Fig. 2D) but suggest that no single metric epitomizes the global priorities for elasmobranch conservation.
Elasmobranch threats and conservation status. A, Percentage of elasmobranch species across the International Union for Conservation of Nature (IUCN) statuses: CR, Critically Endangered; EN, Endangered; VU, Vulnerable; NT, Near Threatened; LC, Least Concern; DD, and Data Deficient. Data downloaded from the IUCN Red List of Threatened Species (http://www.iucnredlist.org, last accessed November 2023). Adapted from Dulvy et al. (Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021). The top panel shows the same information for just extant species with a fossil record (Paillard et al. Reference Paillard, Shimada and Pimiento2021). B, Global distribution of threatened species (represented by number of species categorized by the IUCN as CR, EN, VU) per grid cell. Adapted from Pimiento et al. (Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023). C, Mean percentage of hotspot cells (top 2.5%) defined using various dimensions of biodiversity (i.e., species richness, functional richness, phylogenetic diversity, threatened species (see B), EDGE (Evolutionarily Distinct and Globally Endangered), FUSE (Functionally Unique, Specialized, and Endangered), evolutionary distinctiveness, functional specialization, and functional uniqueness) falling inside or outside existing marine protected areas (MPAs). Adapted from Pimiento et al. (Reference Pimiento, Albouy, Silvestro, Mouton, Velez, Mouillot, Griffin and Leprieur2023).
Elasmobranch conservation has benefited from new information about their biology and threats (e.g., Dulvy et al. Reference Dulvy, Fowler, Musick, Cavanagh, Kyne, Harrison and Carlson2014, Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021; Osgood and Baum Reference Osgood and Baum2015; Mull et al. Reference Mull, Pacoureau, Pardo, Ruiz, García-Rodríguez, Finucci and Haack2022), resulting in additional species protections (Fordham et al. Reference Fordham, Lawson, Koubrak, Cronin, Carrier, Simpfendorfer, Heithaus and Yopak2022) and priority area designations (Hyde et al. Reference Hyde, Notarbartolo di Sciara, Sorrentino, Boyd, Finucci, Fowler and Kyne2022). However, many species remain poorly understood, leaving taxonomic and geographic gaps in our knowledge of their extinction risk (Dulvy et al. Reference Dulvy, Simpfendorfer, Davidson, Fordham, Bräutigam, Sant and Welch2017; Guy et al. Reference Guy, Cox, Williams, Brown, Eckelbecker, Glassic, Lewis, Maskill, McGarvey and Siemiantkowski2021) and the biological outcomes of management efforts (Ferraro and Pressey Reference Ferraro and Pressey2015; Daly et al. Reference Daly, Smale, Singh, Anders, Shivji, Daly and Lea2018; Pacoureau et al. Reference Pacoureau, Carlson, Kindsvater, Rigby, Winker, Simpfendorfer and Charvet2023; Table 1). Furthermore, elasmobranch conservation priorities primarily react to ongoing losses rather than anticipate future vulnerabilities. A longer-term perspective is needed to ground current conservation priorities in an understanding of past extinctions. Here, we discuss how near- and deep-time geohistorical records can optimize elasmobranch conservation priorities by: (1) contextualizing IUCN assessments, (2) identifying correlates of extinction risk, (3) evaluating prioritization metrics, and (4) mapping spatial distributions of threatened elasmobranch biodiversity (Fig. 3).
Contextualizing IUCN Assessments
Near-time fossil and historical records spanning the last century can contextualize IUCN assessments (McClenachan et al. Reference McClenachan, Ferretti and Baum2012; Leonetti et al. Reference Leonetti, Sperone, Travaglini, Mojetta, Signore, Psomadakis, Dinkel and Bottaro2020; Kowalewski et al. Reference Kowalewski, Nawrot, Scarponi, Tomašových and Zuschin2023). These data are especially informative for elasmobranchs, because the IUCN measures population changes over a period of 10 years or three generations (up to 100 years; Mace and Lande Reference Mace and Lande1991), which for long-lived species usually exceeds modern survey data. For example, Ferretti et al. (Reference Ferretti, Myers, Serena and Lotze2008) used historical records to reconstruct population trends of five shark species in the Mediterranean Sea since the early nineteenth century. They found that these species had declined >96–99.9% from their historical abundances, suggesting an ~2.5-fold larger decline than estimates based solely on available fisheries data spanning the last two decades (Cavanagh and Gibson Reference Cavanagh and Gibson2007; Ferretti et al. Reference Ferretti, Myers, Serena and Lotze2008). Equally large declines went undetected in oceanic whitetip sharks (Carcharhinus longimanus) between the 1950s and 1990s in the Gulf of Mexico (Baum and Myers Reference Baum and Myers2004; Fig. 4A). By extending the time window over which extinction risk was evaluated, historical records contributed to the oceanic whitetip shark's classification as Critically Endangered on the IUCN Red List (Rigby et al. Reference Rigby, Barreto, Carlson, Fernando, Fordham, Francis and Herman2019) and a bycatch ban (PEW Environment Group 2012). Finally, historical records are now considered in IUCN Green List assessments, which measure species’ recoveries against historical baselines (Akçakaya et al. Reference Akçakaya, Bennett, Brooks, Grace, Heath, Hedges and Hilton-Taylor2018; Grace et al. Reference Grace, Akçakaya, Bennett, Hilton-Taylor, Long, Milner-Gulland, Young and Hoffmann2019; see Topic 1). These examples show that by documenting changes in species’ abundances or geographic ranges over multiple decades, historical records in particular can unshift the baselines used in IUCN assessments and flag species that might be poorly monitored or not thought to be at risk (Fig. 3).
Identifying Correlates of Extinction Risk
Over deeper timescales, the fossil record can reveal ecological and life-history traits that make species prone to extinction. In turn, these correlates of extinction risk can be used to test the accuracy of IUCN assessments (Raja et al. Reference Raja, Lauchstedt, Pandolfi, Kim, Budd and Kiessling2021; Fig. 1B), predict the extinction risk of Data Deficient species (Dulvy et al. Reference Dulvy, Fowler, Musick, Cavanagh, Kyne, Harrison and Carlson2014; Walls and Dulvy Reference Walls and Dulvy2020), and identify intrinsically vulnerable species before they start to decline (McKinney Reference McKinney1997; Pimiento and Antonelli Reference Pimiento and Antonelli2022). Although correlations between traits and extinction risk can be inferred from extant taxa (e.g., Ripple et al. Reference Ripple, Wolf, Newsome, Hoffmann, Wirsing and McCauley2017; Dulvy et al. Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021), this information is often restricted to short ecological timescales and is not available for all regions or species. The fossil record offers a wealth of complementary insight into extinction selectivity patterns over long evolutionary timescales. For example, intrinsic traits such as geographic range, body size, diet, and thermoregulation have been associated with extinction risk across different clades and extinction events (Harnik et al. Reference Harnik, Lotze, Anderson, Finkel, Finnegan, Lindberg and Liow2012; Finnegan et al. Reference Finnegan, Anderson, Harnik, Simpson, Tittensor, Byrnes and Finkel2015; Payne et al. Reference Payne, Bush, Heim, Knope and McCauley2016; Pimiento et al. Reference Pimiento, Griffin, Clements, Silvestro, Varela, Uhen and Jaramillo2017; Reddin et al. Reference Reddin, Kocsis, Aberhan and Kiessling2021). In some clades, intrinsic traits predict extinctions better than abiotic factors alone (Boyer Reference Boyer2010). Consequently, the fossil record could facilitate more proactive conservation by applying lessons from past extinctions to better understand which species might be most vulnerable—or resilient—in the current biodiversity crisis (Pimiento and Antonelli Reference Pimiento and Antonelli2022).
Elasmobranchs have suffered at least two major extinction events over the last 150 Myr. Tracing the traits that influence extinction selectivity across these events could help contextualize their current vulnerabilities and corroborate extinction risk correlates inferred from extant species (i.e., body size and depth; Dulvy et al. Reference Dulvy, Fowler, Musick, Cavanagh, Kyne, Harrison and Carlson2014, Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021). During the K/Pg mass extinction, at least 62% of elasmobranch species went extinct, with diet, habitat, and geographic range determining extinction selectivity (Kriwet and Benton Reference Kriwet and Benton2004; Guinot and Condamine Reference Guinot and Condamine2023). Another important extinction event occurred during the early Miocene Epoch, when oceanic elasmobranchs declined in abundance by >90% and morphological diversity by >70% (Sibert and Rubin Reference Sibert and Rubin2021; see Topic 1). During this event, habitat again predicted extinction, with elasmobranchs restricted to deep-sea habitats being most affected. Although ecological specialization appears to be a common correlate of extinction risk across these two events, it is difficult to compare them, because they were studied using different analytical approaches and taxonomic resolutions.
Analytical advances have improved efforts to model extinction rates and their correlates more consistently. For example, macroevolutionary dynamics can be estimated using R packages like DivDyn (Kocsis et al. Reference Kocsis, Reddin, Alroy and Kiessling2019; used by Villafaña et al. Reference Villafaña, Rivadeneira, Pimiento and Kriwet2023), capture–mark–recapture approaches (Liow and Nichols Reference Liow and Nichols2010; used by Sibert et al. Reference Sibert, Friedman, Hull, Hunt and Norris2018), and Bayesian process-based birth–death models (Silvestro et al. Reference Silvestro, Salamin and Schnitzler2014; used by Guinot and Condamine Reference Guinot and Condamine2023), some of which combine fossil occurrences with phylogenetic information (Heath et al. Reference Heath, Huelsenbeck and Stadler2014; used by Brée et al. Reference Brée, Condamine and Guinot2022). Machine learning algorithms are increasingly used to model relationships between extinction risk and traits, allowing the vulnerability of extant taxa to be predicted based on their traits (e.g., Finnegan et al. Reference Finnegan, Anderson, Harnik, Simpson, Tittensor, Byrnes and Finkel2015; Raja et al. Reference Raja, Lauchstedt, Pandolfi, Kim, Budd and Kiessling2021). New methods have also facilitated measurements of traits like trophic position, which are hypothesized to predict extinction risk but are challenging to infer from fossils (see Topic 2). With these tools in hand, it has become more possible to analyze deep-time fossil trait and occurrence data to identify which intrinsic traits are shared across extinction events with different magnitudes and causes (Pimiento and Antonelli Reference Pimiento and Antonelli2022) or where discrepancies emerge (i.e., mismatches between trait-based predictions of extinction risk and IUCN statuses; Raja et al. Reference Raja, Lauchstedt, Pandolfi, Kim, Budd and Kiessling2021).
Evaluating Prioritization Metrics
Geohistorical records can also be incorporated into species-based conservation prioritization metrics like EDGE and FUSE to test their performance. For example, Cavin and Kemp (Reference Cavin and Kemp2011) showed that when fossils are included in the lungfish (Sarcopterygii: Dipnoi) phylogeny, the rankings of top EDGE species change. The fossil record has also been used to evaluate the evolutionary implications of conservation actions guided by the EDGE metric—namely whether it prioritizes species representing the “seeds” of future radiations or whether it preserves the tail ends of depauperate clades now “doomed” to extinction. Bennett et al. (Reference Bennett, Sutton and Turvey2019) found that, when applied to mammals, the EDGE metric does not prioritize seeds or doomed clades but rather slow-evolving “living fossils” that disproportionately contribute to their clade's evolutionary history. This approach could be applied to elasmobranchs to simulate how phylogenetic diversity and EDGE prioritizations might change after hypothetically losing or saving threatened species with unique evolutionary histories (Pimiento and Antonelli Reference Pimiento and Antonelli2022). Additionally, the fossil record could test the extent to which implementing the FUSE metric would preserve ecosystem functioning over time. Such tests could show whether certain components of elasmobranchs’ ecological distinctiveness (uniqueness vs. specialization) are more vulnerable to extinction or important for maintaining functional diversity (Pimiento and Antonelli Reference Pimiento and Antonelli2022). At least 10% of extant elasmobranch species (~100 species) have a fossil record (Paillard et al. Reference Paillard, Shimada and Pimiento2021; Fig. 5A), making them a good testing ground for evaluating these prioritization metrics and predicting the evolutionary and functional legacies if they are used to guide conservation decisions (Fig. 3).
Mapping Spatial Distributions of Threatened Elasmobranch Diversity
Fossil occurrences archive spatially explicit relationships between biodiversity and environmental conditions through geological time, which could help guide area-based conservation. For example, Carrillo-Briceño et al. (Reference Carrillo-Briceño, Carrillo, Aguilera and Sanchez-Villagra2018) used the Neogene elasmobranch fossil record to catalog geographic patterns of paleodiversity in Tropical America, a study that has since contextualized regional conservation priorities (Ehemann et al. Reference Ehemann, González-González, Tagliafico and Weigmann2019; Becerril-García et al. Reference Becerril-García, Arauz, Arellano-Martínez, Bonfil, Ayala-Bocos, Castillo-Géniz and Carrera-Fernández2022). Similarly, macroevolutionary analyses could demonstrate whether diversity hotspots are conserved and might represent resilient areas worth protecting (Pimiento and Antonelli Reference Pimiento and Antonelli2022). Another example comes from Finnegan et al. (Reference Finnegan, Anderson, Harnik, Simpson, Tittensor, Byrnes and Finkel2015), in which the geographic distribution of extinction risk predicted from the Neogene–Pleistocene fossil record was overlaid with maps of human impacts and climate velocity to locate present-day hotspots of extinction vulnerability. Although fossil-informed maps of extinction vulnerability have yet to be incorporated into decision-making processes (Kiessling et al. Reference Kiessling, Raja, Roden, Turvey and Saupe2019), there is a precedent for similar maps of current anthropogenic impacts to prioritize areas for protection (e.g., Queiroz et al. Reference Queiroz, Humphries, Couto, Vedor, da Costa, Sequeira and Mucientes2019).
Fossil and historical occurrences are also increasingly used in conjunction with modern occurrences in species distribution models. These models have been implemented to hindcast ecological niches (Nogués-Bravo Reference Nogués-Bravo2009; Myers et al. Reference Myers, Stigall and Lieberman2015; Monsarrat et al. Reference Monsarrat, Novellie, Rushworth and Kerley2019; Skroblin et al. Reference Skroblin, Carboon, Bidu, Chapman, Miller, Taylor, Taylor, Game and Wintle2021), evaluate area-based prioritization strategies (Williams et al. Reference Williams, Kharouba, Veloz, Vellend, McLachlan, Liu, Otto-Bliesner and He2013), and identify potential refugia or range shifts under future projected climate scenarios for a variety of taxa (Maguire et al. Reference Maguire, Nieto-Lugilde, Fitzpatrick, Williams and Blois2015; Lima-Ribeiro et al. Reference Lima-Ribeiro, Moreno, Terribile, Caten, Loyola, Rangel and Diniz-Filho2017; Jones et al. Reference Jones, Mannion, Farnsworth, Valdes, Kelland and Allison2019; see Topic 3). There is great potential to apply these methods to elasmobranch occurrence data (e.g., Klippel et al. Reference Klippel, Amaral and Vinhas2016; Birkmanis et al. Reference Birkmanis, Freer, Simmons, Partridge and Sequeira2020; Sabadin et al. Reference Sabadin, Lucifora, Barbini, Figueroa and Kittlein2020; Diaz-Carballido et al. Reference Diaz-Carballido, Mendoza-González, Yañez-Arenas and Chiappa-Carrara2022), especially for large migratory species, to describe their spatial distributions and protect areas that harbor threatened diversity (Fig. 3). At the same time, because elasmobranch distributions do not fall neatly within the geopolitical boundaries in which conservation policies are implemented (Dulvy et al. Reference Dulvy, Simpfendorfer, Davidson, Fordham, Bräutigam, Sant and Welch2017; Fordham et al. Reference Fordham, Lawson, Koubrak, Cronin, Carrier, Simpfendorfer, Heithaus and Yopak2022), incorporating fossils into area-based conservation begins with investing in collaborative international science that is coproduced by researchers and decision makers.
Limitations
Conservation paleobiology can contribute valuable temporal perspective to current elasmobranch conservation priorities (Fig. 3, Table 1), although certain attributes of geohistorical records limit their application (Kidwell Reference Kidwell2013, Reference Kidwell2015; Kidwell and Tomašových Reference Kidwell and Tomašových2013; Kowalewski et al. Reference Kowalewski, Nawrot, Scarponi, Tomašových and Zuschin2023). The fossil record is inherently incomplete due to a combination of geological, sampling, and taphonomic biases that influence what is preserved, when, and where (Raup Reference Raup1972, Reference Raup1976; Sepkoski et al. Reference Sepkoski, Bambach, Raup and Valentine1981; Benson et al. Reference Benson, Butler, Close, Saupe and Rabosky2021). Furthermore, although elasmobranch teeth are diagnostic, other components of their fossil record (e.g., vertebrae) cannot be identified to species or even genera, limiting taxonomic insight (Reif Reference Reif1985; Cappetta Reference Cappetta and Schultze2012). Paleobiological work on elasmobranchs also appears to be biased toward sharks, leaving rays, a highly threatened clade (Dulvy et al. Reference Dulvy, Pacoureau, Rigby, Pollom, Jabado, Ebert and Finucci2021), less studied. Some limitations can be addressed by investing in taxonomic expertise, specimen and data digitization, and additional field or collections work, which could expand available datasets and improve our understanding of elasmobranch systematics and traits. Additionally, future work could assess whether sharks could serve as taxonomic surrogates for rays. At the same time, striking differences in the pace and scale of human impacts on elasmobranch populations today relative to the time-averaged eco-evolutionary dynamics captured by the fossil record remain an impasse. This often manifests as misalignment between the spatiotemporal scale of a conservation problem and available geohistorical records (Dietl et al. Reference Dietl, Smith and Durham2019; Kiessling et al. Reference Kiessling, Raja, Roden, Turvey and Saupe2019). Although no silver bullet, pairing one record's limitations with another's strengths could help improve data coverage and resolution (Buma et al. Reference Buma, Harvey, Gavin, Kelly, Loboda, McNeil and Marlon2019).
Other elasmobranch conservation needs fall outside the scope of conservation paleobiology. For example, Jorgensen et al. (Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022) identified several priorities that are not answerable using information preserved in geohistorical records, such as those involving tourism, vessel tracking, trade laws, and bycatch mitigation policies (Table 1). Effective governance and community buy-in are critical for achieving conservation successes but cannot be tackled with fossils alone. Other priorities identified by the authors would require creative reframing for geohistorical records to be relevant (e.g., tagging technologies, which track species’ movement), highlighting the value of jointly developing these priority questions. To increase the conservation relevance of paleobiological data, conservation paleobiologists could collaborate across disciplinary boundaries, such that geohistorical records are matched with conservation problems that they are well suited to address in complement with approaches from other fields. Doing so could help ensure that paleobiological research, when applicable, considers the multidimensional nature of conservation problems and provides actionable results targeted to specific conservation needs that are voiced by practitioners or other decision makers (Savarese Reference Savarese, Tyler and Schneider2018; Dietl et al. Reference Dietl, Durham, Clark and Prado2023). This collaborative approach could ultimately help cement geohistorical records’ place in conservation problem-solving toolboxes.
Coda
Throughout this article, we have highlighted research directions in conservation paleobiology that have potential to address emergent priorities in elasmobranch science and conservation. These priorities were broadly assembled from expert opinions (Jorgensen et al. Reference Jorgensen, Micheli, White, Van Houtan, Alfaro-Shigueto, Andrzejaczek and Arnoldi2022), whereas on-the-ground conservation problems may be more context specific and evolve as needs change. Nonetheless, this exercise could serve as a model for understanding which geohistorical records are most useful for conservation practice. Evaluating alignment between conservation needs and geohistorical records could lead to more tangible conservation paleobiology research goals that are designed with implementation in mind, for elasmobranchs as well as other clades. We find that these areas of overlap are often best addressed with information spanning many timescales and branches of paleontology and biology, creating opportunities for interdisciplinary collaboration and synthesis. For conservation paleobiologists seeking to increase the practical applications of their work, we encourage finding similar horizon-scanning lists of priority questions (or similar venues where such priorities are exchanged) as a potential starting point for initiating conversations with practitioners working in their study systems. These two-way interactions could define shared questions that conservation paleobiology could help answer and, in doing so, coproduce more actionable science.
Acknowledgments
We thank A. O'Dea for conceptual discussions, J. Cybulski for his valuable feedback on the manuscript draft and figures, I. Cooke Tapia and Cooked Illustrations for their creative assistance with figure design, and G. Mathes for assistance with figure creation. We also thank E. Sibert, W. Kiessling, and one anonymous reviewer for their insightful comments, which have greatly improved the article. E.M.D. is supported by an Earl S. Tupper Postdoctoral Fellowship from the Smithsonian Tropical Research Institute. C.P. is funded by a PRIMA grant (no. 185798) from the Swiss National Science Foundation.
Competing Interests
The authors declare no competing interests.
Data Availability Statement
Supplementary Material is available from the Zenodo Digital Repository: https://doi.org/10.5281/zenodo.10681908.








