Introduction
The arid landscape of the north coast region of Peru has been one of the most dynamic and productive areas of Peruvian archaeology (Shimada and Vega-Centeno, Reference Shimada, Vega-Centeno and Lozny2011; Tantaleán, Reference Tantaleán2014; Asensio, Reference Asensio2018). From the discovery of the Lord of Sipan in 1987 to the inscription of Caral in the World Heritage List in 2009, the archaeological community in Peru has undertaken numerous efforts to understand the cultural and natural dynamics of the populations that inhabited these desert areas (Alva and Donnan, Reference Alva and Donnan1993; Reindel, Reference Reindel, Reindel and Wagner2009; Shady et al. Reference Shady, Machacuay, Novoa, Quispe and Leyva2015; Chicoine, Reference Chicoine2017). This region stands out for the emergence of the valleys as oasis, adobe brick pyramids, large urban centres, and aqueducts built by state societies as the Moche and the Chimú, which are central elements of this archaeological scenery. Hydraulic technologies (Ortloff, Reference Ortloff2022), climatic catastrophes (Sandweiss et al. Reference Sandweiss, Maasch and Kelley2024), monumentality and the emergence of statal and imperial political entities have been the focus of Peruvian archaeological research (Stanish, Reference Stanish, Bondarenko, Kowalewski and Small2020; Tantaleán, Reference Tantaleán2021; Stanish et al. Reference Stanish, Earle, García Sanjuán, Tantaleán and Barrientos2024). In this work, a novel approach for the region is used, based on paleoparasitological research, which has the potential to add relevant information concerning the lifestyles of the ancient settlers, and the potential effects of the ecological relations between human and animal groups.
Paleoparasitological studies have allowed the identification of parasite structures from ancient times belonging to both humans and animals, through the analysis of diverse preserved materials commonly recovered in archaeological and, to a lesser extent, paleontological sites (Bouchet et al. Reference Bouchet, Guidon, Dittmar, Harter, Ferreira, Chaves, Reinhard and Araújo2003; Araújo et al. Reference Araújo, Reinhard, Ferreira, Pucu and Chieffi2013). These include coprolites, sediments from latrines, pits and the pelvic area of skeletonized individuals, soft tissues from mummified bodies, and hair, among others (Ferreira et al. Reference Ferreira, Reinhard, Araújo, Ferreira, Reinhard and Araújo2014; Mitchell, Reference Mitchell2015). A substantial body of works employing diverse adapted methodologies has provided valuable information regarding diet, health, paleoenvironmental scenario, and human-animal interactions of human past populations from several regions, including South America (Reinhard et al. Reference Reinhard, Confalonieri, Herrmann, Ferreira and Araújo1986; Araujo et al. Reference Araujo, Reinhard, Ferreira and Gardner2008; Araújo et al. Reference Araújo, Reinhard, Leles, Sianto, Iñiguez, Fugassa, Arriaza, Orellana and Ferreira2011; Ferreira et al. Reference Ferreira, Reinhard, Araújo, Ferreira, Reinhard and Araújo2014; Mitchell, Reference Mitchell2015; Novo and Ferreira, Reference Novo and Ferreira2016; Seo et al. Reference Seo, Chai, Kim, Shim, Ki and Shin2016). However, a few studies have been addressed from a paleoparasitological perspective in Peruvian archaeological contexts, which have evidenced gastrointestinal infection, haemoparasites, and tissular parasites in human groups, gastrointestinal infection in camelids, and ectoparasite infestation on cuyes or guinea pigs (Cavia porcellus) mummified corpses (Callen and Cameron, Reference Callen and Cameron1960; Allison et al. Reference Allison, Pezzia, Hasegawa and Gerszten1974; Patrucco et al. Reference Patrucco, Tello and Bonavia1983; Fornaciari et al. Reference Fornaciari, Castagna, Viacava, Tognetti, Bevilacqua and Segura1992; Leguía et al. Reference Leguía, Casas and Wheeler1995; Dittmar, Reference Dittmar2000; Guillen and Allison, Reference Guillen and Allison2005; Le Bailly et al. Reference Le Bailly, Goepfert, Prieto, Verano and Dufour2020). In this study, we investigated the presence of gastrointestinal parasites in animal coprolites from the Licapa II archaeological site, located in the Chicama Valley, Peru, associated with the Moche culture, and occupied between 450 and 900 AD (Koons, Reference Koons2012:371–372). Our aim is to explore parasite diversity in this ancient context and discuss their implications for understanding animal parasitism and human-animal interactions in the Moche culture.
Materials and methods
Licapa II: A Moche archaeological site
Licapa II (UTM: 692497E; 9148290 N) is a Moche archaeological complex located on the northern edge of the Chicama Valley (Figure 1). Located on the eastern slope of Cerro Azul mountain and the border of the productive area of the valley, it covers an area of about 240 000 m2, with six principal sub-architectural components: Huaca A, Residential Area, Huaca B, Funerary Platform, Storage facility, and secondary montículos (Koons, Reference Koons2012; Tavera Medina et al. Reference Tavera Medina, Tantaleán, Morales, Huachaca Montes, Quispe-Baquedano and Sanandres2025), aligned along a southwest-northeast axis. The complex is the product of a palimpsest landscape. According to Koons, Huaca A is the earliest structure, and the Residential Area, together with Huaca B, is the latest one. In other words, the complex has two phases ‘The first phase began sometime around 450–500 AD and was characterized by the presence of Huaca A. […] Sometime around 600 AD Huaca B and […] the area in between the Huacas [Residential Area] was now used for domestic/utilitarian activities. […] They abandoned the site around 900 AD’ (Koons, Reference Koons2012:371–372). For this study, we focus our attention on the lesser-known part of the complex: the Residential Area, as an effort to understand in depth the domestic conditions of this Moche settlement. The archaeological context of these samples, recovered during test excavations conducted in 2021 by the Chicama Archaeological Program (Tantaleán et al. Reference Tantaleán, Tavera, Gastello and Campos2021, Reference Tantaleán, Tavera, Campos, Gastello and Osores2022) in the area between the huacas, was interpreted as a guinea pig pen, that underwent remodelling and was subsequently sealed (Figure 2) (Tantaleán et al. Reference Tantaleán, Tavera, Gastello, Uribe and Roman2024). This occupation is connected with the second phase of the settlement, when Huaca B began to be constructed and the area between the two huacas started to be used for domestic and utilitarian activities (Koons, Reference Koons2012: 372; Tavera Medina et al. Reference Tavera Medina, Tantaleán, Morales, Huachaca Montes, Quispe-Baquedano and Sanandres2025).
On the left, North Coast of Peru showing the Chicama Valley and Moche sites mentioned in the text. On the right, two views of Licapa II site.

Our analysis centred on samples from Levels 11 and 13 of Unit 5 of the Residential Area (Figure 2). Although Level 13 was the deepest excavated context (Figure 2, detail E), it did not represent a sterile layer. Instead, it corresponds to the earliest identified guinea pig pen in this unit. This level was characterized by a yellowish clay floor containing coprolite deposits, botanical remains including a whole Psidium cattleianum fruit and fragments of Pouteria lucuma (Usca Méndez et al. Reference Usca Méndez, Flores Mancheno, Tello Flores and Navarro Ojeda2022:47) (Figure 2, details C and D), as well as non-diagnostic ceramic sherds. A second pen, constructed as a clay floor in Level 12, was built directly above the earlier pen. In this context, fragments of Moche-decorated ceramics were recovered (Figure 2, detail B). Finally, Level 11 consisted of a reddish clay matrix with a high concentration of coprolites. Above this, we identified a compact layer of yellowish clay (Level 10), interpreted as a floor sealing the uppermost phase of the pen (Figure 2, details F and G).
Stratigraphic correlation of Unit 5 of Licapa II site, and associated architectural and material details. (A) Variation in coprolite morphology; (B) Moche ceramic associated with the floor of the Guinea pig pen at level 12; (C) Psidium cattleianum seed associated with the floor of the guinea pig pen at level 13; (D) Pouteria lucuma seed associated with the floor of the guinea pig pen at level 13; (E) Floor of the guinea pig pen at level 13; (F) Closure of the guinea pig pens, coinciding with the construction of the floor corresponding to level 10; (G) Moche ceramic associated with the floor of level 10.

The samples
Thirty-six coprolites were recovered from a test pit at Licapa II after sieving the clay sediment (Figure 2). These specimens belonged to two stratigraphic units: N11 (n = 8) and N13 (n = 28). One coprolite from each stratum was preserved at −20ºC for future genetic analysis. Since they displayed different morphological characteristics, and considering the zooarcheological evidence for the site, they allowed us to suggest two different zoological origins (Chame, Reference Chame2003; Roman, Reference Roman2023). Coprolites recovered during the excavation of the N11 stratum were brown, with smooth surfaces and ovoid-shaped, and some were pointed at one or both ends. They measured between 1.0–1.5 cm in height and 0.5–0.9 cm in width, and were tentatively assigned to South American camelids (SAC). Coprolites from the N13 stratum were dark brown, cylindrical, with a regular surface. Their height ranged from 0.6 to 0.9 cm, and their width from 0.2 to 0.5 cm. Based on morphology and size, these samples were consistent with a rodent origin. However, given that the archaeological evidence indicates the presence of a pen in this layer, and that guinea pigs (Cavia porcellus) are the only rodent species known to have been domesticated in the region during that period, we consider it likely that they specifically belong to guinea pigs, or cuyes, the local name for this animal. Only one coprolite from N13 (sample 28) matched the pointed morphology and size (1.0 × 0.6 cm) characteristic of the SAC coprolites observed in N11.
Paleoparasitological techniques
Initially, each sample was individually photographed and processed. Coprolites were completely disaggregated in 15 mL tubes containing 0.5% trisodium phosphate solution using a glass bar and left to rehydrate for 72 h at 4 °C (Callen and Cameron, Reference Callen and Cameron1960) at constant motion in a mixer. Next, they were allowed to settle for 24 h at the same temperature, to concentrate all the microremains at the bottom of the tubes (Lutz, Reference Lutz1919). Then, aliquots of 200 µL were taken from each sample, and slides were mounted using rehydrated material and a drop of glycerine. Finally, the slides were observed in a light microscope (Labklass XSZ 107 CCD) at 100X (total magnification), and photographs of parasite remains were taken at 400X. Measures of the remains were taken with the software TCapture 4.3.0.605. Only well-preserved structures were measured. Diagnostics of parasite taxa were made based on published literature (Atías, Reference Atías1998; Acha and Szyfres, Reference Acha and Szyfres2003; Goater et al. Reference Goater, Goater and Esch2014) and resources available online.
Results
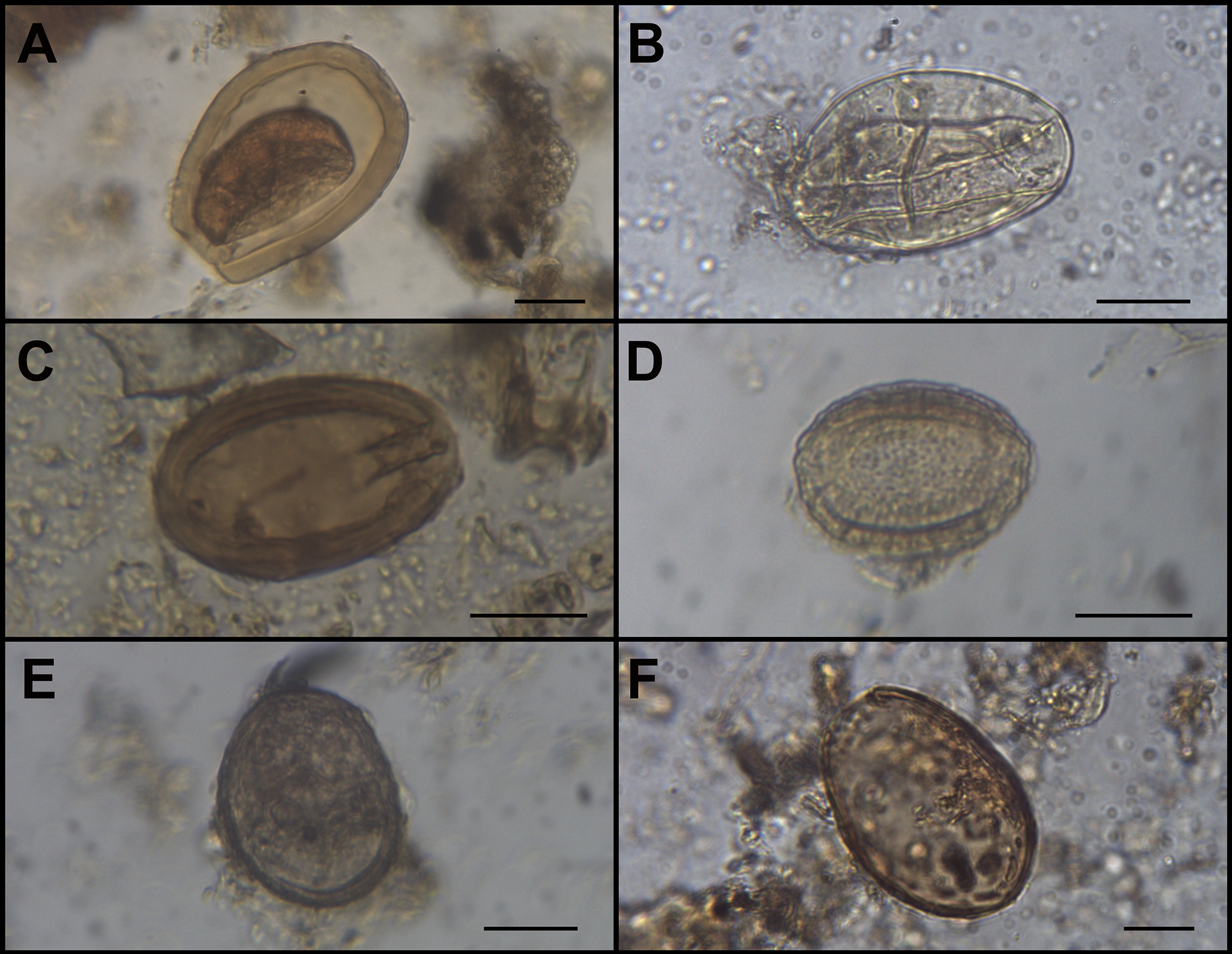
The analysis allowed us to identify six protozoan and helminth parasite taxa, at different levels of taxonomic resolution, in 13 of the 34 coprolites analysed from both strata (Figure 3; Tables 1 and 2). First, 68 oocysts of Eimeria macusaniensis (Apicomplexa, Eimeriidae, Eucoccidiorida) were identified in four coprolites, three of them from the N11 stratum, and one from the N13. They presented their characteristic ovoid shape, and were thick-walled and light brown, containing pyriform apparatus and micropyle, and measuring 64–103 µm in length by 51–78 µm in width (mean 86.87 µm length, 65.92 µm width, n = 40) (Figure 3A).
Paleoparasitological findings in animal coprolites from Licapa II site: (A) Eimeria macusaniensis oocyst; (B) Strongylid-type egg; (C) Acantocephalan egg; (D) Dioctophymatid egg; (E) Possible Paraspidodera sp. Egg; and (F) Dibothriocephalus sp./Adenocephalus sp. Egg. Bar = 20 μm.

Information of the samples analysed in this study from the Licapa II archaeological site, Chicama Valley, Peru: stratum, zoological origin, number of samples, sample ID, and paleoparasitological findings. SAC: South American camelids

Measurements of the parasite eggs found at Licapa II archaeological site, Chicama Valley, Peru and number (n) of measured structures

In addition, 21 eggs identified as Strongylid-type (Nematoda: Strongylida) were observed in samples from both strata. They were either thick or thin-walled, brown, with or without internal content, and measured 56–77 µm in length, by 38–56 µm in width (mean 63.29 µm length, 47.43 µm width, n = 14) (Figure 3B).
One acantocephalan egg (Acanthocephala: Archiacanthocephala, Oligacanthorhynchidae) was recovered in stratum N11. It is represented by a single brown, multilayered, elongated egg containing visible hooks, measuring 58 µm in length by 38 µm in width (Figure 3C).
Thirty-four structures identified as Dioctophymatidae eggs (Nematoda: Ascaridida, Dioctophymatide) were found in samples from N13. They were elliptic-shaped, thick and irregular-walled, and light brown to yellowish, and measured 33–48 µm in length by 22-41 µm in width (mean 41.76 µm length by 31.33 µm width, n = 21) (Figure 3D).
Six nematode eggs were found in one sample from the N13 stratum. They were ovoid-shaped, grey and thick-shelled, and measured 49–60 µm in length to 35–45 µm in width (mean 56.80 µm length by 42.20 µm width, n = 5) (Figure 3E). They were tentatively assigned to the genus Paraspidodera (Nematoda: Spirurida, Aspidoderidae).
Finally, two structures attributable to Dibothriocephalus sp./Adenocephalus sp. eggs (Platyhelminthes: Cestoda, Diphyllobothriidea, Diphyllobothriidae) were recovered from both strata. They were ovoid-shaped, either light or dark brown, without preserved operculum, and measured 61–75 µm in length by 44–52 µm in width (mean 68.00 µm length by 48.00 µm width, n = 2) (Figura 3F).
Discussion and conclusions
This paleoparasitological research led to the identification of one protozoan and five helminth parasite species, preserved in coprolites from both stratigraphic levels.
On the one hand, Eimeria macusaniensis is a protozoan parasite that frequently infects SAC and is considered of major importance to the health of these animals. It can cause parasitic disease and, in severe cases, can pose a threat to their lives (Aguirre and Cafrune, Reference Aguirre, Cafrune, Suárez, Olaechea, Rossanigo and Romero2007). There is a large record of findings of this protozoa in preserved feces from South American archaeological contexts, mainly in SAC coprolites (Leguía et al. Reference Leguía, Casas and Wheeler1995; Fugassa, Reference Fugassa2007; Taglioretti et al. Reference Taglioretti, Fugassa and Sardella2015, Reference Taglioretti, Fugassa, Rindel and Sardella2017; Amalfitano et al. Reference Amalfitano, Petrigh, Lobos and Fugassa2018, Reference Amalfitano, Petrigh and Fugassa2019; Petrigh et al. Reference Petrigh, Velázquez, Fugassa, Burry, Mondini and Korstanje2021; Tietze et al. Reference Tietze, Urquiza and Beltrame2021; Beltrame et al. Reference Beltrame, Tietze, Cañal, Barberena and Quintana2022; Ramirez et al. Reference Ramirez, Herrera‐Soto, González Andreu, Santana‐Sagredo, Uribe Rodríguez and Nores2023; Rodríguez et al. Reference Rodríguez, Cañal, Seguí, López, Silvana, Quintana, Tietze and Beltrame2025) with temporal depth of up to 10 000 yBP (Fugassa et al. Reference Fugassa, Sardella, Taglioretti, Reinhard and Araújo2008), as well as in human-origin coprolites associated to false parasitism (Vieira de Souza et al. Reference Vieira de Souza, da Silva, da Silva-pinto, Mendez-Quiros, de Miranda Chaves and Iñiguez2018). Of note, E. macusaniensis has been reported in camelid coprolites from the Huanchaquito site in Peru, in the current coastal homonymous city, dated 1400–1450 AD (Le Bailly et al. Reference Le Bailly, Goepfert, Prieto, Verano and Dufour2020).
On the other hand, helminth findings are represented by five different taxa. Eggs belonging to Strongylida parasites were found in both levels. Due to poor preservation of these structures and difficulties in recognizing strongylid species solely on egg features, they are just referred to as strongylid-type, which might belong to different species. This nematode order includes several helminth species, which can infect and cause disease in animals, sometimes leading to severe damage in the gastrointestinal tract (Aguirre and Cafrune, Reference Aguirre, Cafrune, Suárez, Olaechea, Rossanigo and Romero2007; Suárez, Reference Suárez, Suárez, Olaechea, Rossanigo and Romero2007). They have a wide range of definitive hosts, including mammals, birds, amphibians, and reptiles, and are considered of major importance for SAC and other herbivores (Urquhart et al. Reference Urquhart, Armour, Duncan, Dunn and Jennings1996). The paleoparasitological record has accounted for the presence of strongylids in different animal feces from archaeological contexts in several locations, including rodents and camelids (Araújo et al. Reference Araújo, Ferreira, Confalonieri, Chame and Ribeiro1989; Taglioretti et al. Reference Taglioretti, Fugassa and Sardella2015, Reference Taglioretti, Fugassa, Rindel and Sardella2017; Beltrame et al. Reference Beltrame, Tietze, Pérez, Bellusci and Sardella2017; Tietze et al. Reference Tietze, de Tommaso and Beltrame2020, Reference Tietze, Urquiza and Beltrame2021; Ramirez et al. Reference Ramirez, Herrera‐Soto, González Andreu, Santana‐Sagredo, Uribe Rodríguez and Nores2023), the latter also in Peru (Leguía et al. Reference Leguía, Casas and Wheeler1995).
An acanthocephalan egg was identified in N11 stratum. This is a large phylum formed by obligate parasites infecting a wide range of animals, like fish, amphibians, reptiles, birds, and mammals, through arthropods acting as intermediate hosts (Goater et al. Reference Goater, Goater and Esch2014). They can cause parasite disease in some of the definitive hosts, including humans, despite this disease being rather rare (Andres et al. Reference Andres, English and Greiner2014). Paleoparasitological research has shown that these parasites were widely distributed in ancient scenarios, with acanthocephalan eggs being found in samples of human and animal origin, including canid, felid and rodent feces (Moore et al. Reference Moore, Fry and Englert1969; Ferreira et al. Reference Ferreira, Araújo, Confalonieri and Chame1989; Noronha et al. Reference Noronha, Ferreira, Rangel, Araujo and Gomes1994; Fugassa et al. Reference Fugassa, Reinhard, Johnson, Gardner, Vieira and Araújo2011; Beltrame et al. Reference Beltrame, Sardella, Fugassa and Barberena2012, Reference Beltrame, Fernández and Sardella2015, Reference Beltrame, Bellusci, Fernández and Sardella2018; Sianto et al. Reference Sianto, de Souza, Chame, da Luz, Guidon, Pessis and Araújo2014; Mowlavi et al. Reference Mowlavi, Makki, Heidari, Rezaeian, Mohebali, Araujo, Boenke, Aali, Stollner and Mobedi2015; Ramirez et al. Reference Ramirez, Tavarone, Pérez Pincheira, Colobig, Basso, Beltrame and Nores2025). Here, since only one egg was found, a tentative identification based on morphological and morphometric characteristics is challenging. Nevertheless, its morphology resembles the eggs found previously in 9000-year-old feline coprolites from Brazilian contexts, identified as Oncicola sp. (Sianto et al. Reference Sianto, de Souza, Chame, da Luz, Guidon, Pessis and Araújo2014). A true infection in camelids with this parasite genus seems unlikely since it usually infects other animals, like canids and felids (Santos et al. Reference Santos, Chame, Chagas-Moutinho and Santos2017). Instead, the presence of acanthocephalan parasites in this sample might be related to a false parasitism event, resulting from contamination of the pastures from where camelids feed on with other animal faeces, probably belonging to rodents or canids, both of which have been identified in the zooarchaeological record of the site (Roman, Reference Roman2023). Alternatively, the egg found could result from the percolation from upper stratigraphic levels, where remains of the definitive hosts of this parasite might be present (Roman, Reference Roman2023).
Eggs tentatively assigned to the nematode Paraspidodera were found in one sample attributed to rodents. However, we cannot identify the species based on eggs features solely. Some species infect several rodent species from South America (Griffiths, Reference Griffiths1971). For instance, P. uncinata is considered one of the most frequently found parasites in guinea pigs, and even the parasitosis of major concern for this animal (Huamán et al. Reference Huamán, Killerby and Chauca2020), usually showing high prevalence of infection in both wild and domestic cuyes. The paleoparasitological record has provided evidence for the presence of P. uncinata in Argentinian archaeological contexts of up to 7900 years before the present (Sardella and Fugassa, Reference Sardella and Fugassa2009; Beltrame et al. Reference Beltrame, Fernández and Sardella2015).
Structures suggestive of eggs of Dioctophymatidae were found, also in both strata. This nematode family includes species infecting several animals as definitive hosts, mainly carnivores like mustelids, canids, felids, and, accidentally, humans (Eiras et al. Reference Eiras, Zhu, Yurlova, Pedrassani, Yoshikawa and Nawa2021). The parasite eggs are excreted in the urine of the host and are then ingested by annelids acting as intermediate hosts. Fishes or amphibians like frogs might ingest the infected annelid as a paratenic host, i.e. it does not allow the development of the parasite within its body but facilitates its transmission. Ingestion of the annelid or the infected paratenic host by the definitive host allows the development of the adult parasite in their kidney after migration from the intestines (Mascarenhas et al. Reference Mascarenhas, Müller, de Macedo, Henzel, Robaldo and Corrêa2019). The morphological features of the structures recovered in this work are suggestive of Dioctophymatidae eggs, but their size is smaller than that observed in parasitological literature (Pedrassani et al. Reference Pedrassani, Hoppe, Avancini and Nascimento2009). However, smaller eggs, comparable in size to those reported here, have also been found in zooarchaeological samples from small rodents (Tietze et al. Reference Tietze, de Tommaso and Beltrame2020). It has been suggested that eggs of different sizes can be laid according to the female development stage and the host, and that the smaller size could also be an effect of taphonomic processes (Fugassa et al. Reference Fugassa, Gonzalez Olivera and Petrigh2013). The paleoparasitological record has accounted for findings of dioctophymatid eggs in archaeological sites from South America, Europe, and Asia in human and animal-origin samples (Le Bailly et al. Reference Le Bailly, Leuzinger and Bouchet2003, Reference Le Bailly, Leuzinger, Schlichtherle and Bouchet2007; Fugassa et al. Reference Fugassa, Gonzalez Olivera and Petrigh2013; Beltrame et al. Reference Beltrame, Tietze, Pérez, Bellusci and Sardella2017; Ledger et al. Reference Ledger, Grimshaw, Fairey, Whelton, Bull, Ballantyne, Knight and Mitchell2019; Tietze et al. Reference Tietze, de Tommaso and Beltrame2020; Bizhani et al. Reference Bizhani, Najafi, Rokni, Sharifi, Fallah Kiapi, Paknezhad, Mohtasebi, Mowlavi, Otranto and Dupouy-Camet2023).
Finally, eggs of Dibothriocephalus sp. or Adenocephalus sp. were observed in camelid-origin coprolites. These parasites have been previously recorded in paleoparasitological investigations of South America, including Peru (Callen and Cameron, Reference Callen and Cameron1960; Baer et al. Reference Baer, Miranda, Fernandez and Medina1967; Ferreira et al. Reference Ferreira, Araújo, Confalonieri and Nuñez1984; Gonçalves et al. Reference Gonçalves, Araújo and Ferreira2003; Reinhard and Urban, Reference Reinhard and Urban2003; Santoro et al. Reference Santoro, Vinton and Reinhard2003; Tuemmers et al. Reference Tuemmers, Willgert and Serri2014; Kuchta et al. Reference Kuchta, Serrano-Martínez and Scholz2015; Ramirez et al. Reference Ramirez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021). This cestode genus includes species of parasites that infect several fish-eating mammals, including humans, after passage through two aquatic intermediate hosts (Wicht et al. Reference Wicht, Yanagida, Scholz, Ito, Jiménez and Brabec2010). In Licapa II, the existence of a canal or water channel in the site would have allowed the presence of the intermediate and/or paratenic hosts of both Dioctophymatidae and Dibothriocephalus sp./ Adenocephalus sp., which is further evidenced by the zooarchaeological record that accounted for both fish and frog remains (Koons, Reference Koons2012; Roman, Reference Roman2023; Tantaleán et al. Reference Tantaleán, Tavera, Gastello, Uribe and Roman2024). Although agriculturalism was the main subsistence strategy in these populations, hunting-gathering-fishing practices were also important, as mixed economies were proposed (Billman, Reference Billman2002; Chapdelaine, Reference Chapdelaine2011). In this sense, it is expected that people living here might have been exposed to these zoonotic parasitic infections, mainly if they consumed insufficiently cooked fish or frogs. While rodents but not SAC can occasionally become infected with dioctophymatids (Tokiwa et al. Reference Tokiwa, Harunari, Tanikawa, Akao and Ohta2011), it is unlikely that camelids and rodents can be infected with Dibothriocephalus sp./Adenocephalus sp. Therefore, the presence of their eggs in these coprolites might be related either to environmental contamination with eggs from definitive hosts, the intake of contaminated water sources, or the accidental ingestion of eggs present in pastures.
The analysed samples herein were interpreted as belonging to two different animals. On the one hand, the presence of eggs of the coccidian Eimeria macusaniensis allowed us to confirm the SAC origin of the coprolites from the N11 stratum, as well as one from N13, since SAC are its sole definitive host. On the other hand, although no parasite fauna specific to guinea pigs was identified, consideration of the size and morphology of the coprolites, together with the analysis of the complex stratigraphy of this test pit, which indicates that this structure was used as a cuyera, i.e. a guinea pig pen or enclosure where these animals were raised (Tantaleán et al. Reference Tantaleán, Tavera, Gastello, Uribe and Roman2024), supports the interpretation that the coprolites from N13 belong to guinea pigs or cuyes. This is further supported by zooarchaeological evidence from the upper strata of this test pit, including a bone of Cavia porcellus bearing cut marks (Roman, Reference Roman2023). Guinea pigs belong to the family Caviidae, within the suborder Hystricomorpha of the order Rodentia. The earliest archaeological records date back approximately 9000 years, and domestication is believed to have occurred between 5000 and 2500 BC (Sandweiss and Wing, Reference Sandweiss and Wing1997). Despite guinea pigs constituting a substantial source of meat in the central Andes (Morales, Reference Morales1994), the archaeological record frequently lacks sufficient evidence to substantiate their consumption (Valdez and Valdez, Reference Valdez and Valdez1997).
Parasitic disease is considered a major problem of both camelid and cuyes populations (de Lamo, Reference de Lamo2011; García et al. Reference García, Chávez, Pinedo and Suárez2013). Indeed, it has been suggested that more than a third of the morbidity and mortality of cuyes is due to parasite diseases (García et al. Reference García, Chávez, Pinedo and Suárez2013). Most of the species found here are not of major zoonotic importance, and cases of human infection with certain parasites, like acanthocephalan, are only occasionally reported. These cases are mainly a result of false parasitism events caused by the consumption of truly infected animals, usually their intermediate or definitive hosts (Sianto et al. Reference Sianto, Chame, Silva, Gonçalves, Reinhard, Fugassa and Araújo2009, Reference Sianto, de Souza, Chame, da Luz, Guidon, Pessis and Araújo2014). Nevertheless, the close contact of humans and animals, and the constant handling of feces, might have posed a threat to people’s health, since these practices allow the direct transmission of pathogens, not only parasites like the ones we aimed to explore here, but also bacteria and viruses.
The development of Moche state multivalley settlements (Stanish, Reference Stanish2001; Uceda Castillo et al. Reference Uceda Castillo, Morales Gamarra and Mujica Barreda2016; Tantaleán, Reference Tantaleán2021) marked a period of significant growth in the Central Andes. This period witnessed the increase of population density in settlements (Billman, Reference Billman1996) and the development of an economy based on agricultural production, making it an exceptional event in the region. Life in urban settlements gave rise to a new spatial system, which presented challenges to all activities, including the breeding and disposal of animal feces. This suggests that a variety of maintenance activities must be developed within the same spatial context, thereby engendering a more intimate cohabitation dynamic between domestic animals and humans, which had been previously suggested through the excavations of the Urban Core of Huacas de Moche (Uceda Castillo et al. Reference Uceda Castillo, Morales Gamarra and Mujica Barreda2016:56–57), and that has started to be observed in current excavations of the residential area of Licapa II. This initial study also demonstrated the remodelling dynamics of breeding animal spaces, a finding that, in conjunction with the current excavations, will facilitate the future development of a diachronic perspective on the health of domestic animals in urban settlements in the Central Andes.
Beyond the lack of accurate taxonomic identification in four of the six taxa identified, these first paleoparasitological analyses provide valuable information on the parasite diversity occurring in ancient times in the region and, in a broader sense, in Peru, where paleoparasitological data are scarce, compared with other South American regions. Furthermore, it contributes to understanding how certain practices carried out at the site by its inhabitants may have played a role in the transmission and persistence of specific parasitic infections. Also, it sheds light on the complex relationship between environment, parasites, and hosts, evidenced in certain parasites with indirect life cycles. We expect that future samplings of coprolites at this site and others in the region will allow us to expand our knowledge on ancient parasite ecology and diversity in South America, as well as the potential effect on both human and animal past populations.
Acknowledgements
We are also grateful to the research team, comprising students and recent graduates of the National University of San Marcos, for their commitment to the field, and to the Municipality of Rázuri for logistical assistance duing the fieldwork of 2021. We also acknowledge the Institute for Advanced Studies of Culture and the Environment, University of South Florida, for its longstanding support of our research over the years.
Author contributions
D.A.R, C.T.M. and R.N. conceptualized the study. C.T.M. and H.T. conducted archaeological surveys and provided samples. D.A.R. performed microscopy examinations and structure identification, and wrote the original draft of the manuscript with support from R.N. D.A.R. and C.M.T. prepared the figures. All authors reviewed and edited the manuscript, contributing to its structure and specific content.
Financial support
This research was supported by the Universidad Nacional Mayor de San Marcos - RR N° 01686-2020 and project number E20151261. Also, the Instituto Peruano de Estudios Arqueológicos (IPEA) supported economically this research.
Competing interests
The authors declare there are no conflicts of interest.
Ethical standards
Not applicable.