Introduction
Digenean trematodes of the Liolopidae Dollfus, 1934 infect the gastrointestinal tract of reptiles and amphibians associated with aquatic habitats (Skrjabin, Reference Skrjabin1962; Brooks and Overstreet, Reference Brooks and Overstreet1978; Niewiadomska, Reference Niewiadomska, Gibson, Jones and Bray2002; Baba et al., Reference Baba, Hosoi, Urabe, Shimazu, Tochimoto and Hasegawa2011; Dutton et al., Reference Dutton, DuPreez, Urabe and Bullard2022, Reference Dutton, Bullard, Brule and Kelly2024). Higher classification for the Liolopidae remains problematic. The family unambiguously belongs to the Diplostomida but has variously been treated within each of the 3 diplostomidan superfamilies recognized at present, the Brachylaimoidea Joyeux & Foley, 1930, Diplostomoidea Poirier, 1886 and Schistosomatoidea Stiles & Hassall, 1898, as well in the previous concept of the Clinostomoidea Lühe, 1901 (Niewiadomska, Reference Niewiadomska, Gibson, Jones and Bray2002). Phylogenetic analyses have found the family to be genetically distinct and basal among the Diplostomida, resolving sister to either the Diplostomoidea or Schistosomatoidea (Baba et al., Reference Baba, Hosoi, Urabe, Shimazu, Tochimoto and Hasegawa2011; Hernández-Mena et al., Reference Hernández-Mena, García-Varela and Pérez-ponce de León2017) or even sister to all other Diplostomida (Cutmore et al., Reference Cutmore, Littlewood, Arellano-Martínez, Louvard and Cribb2023).
The Liolopidae currently comprises 6 genera, of which only Harmotrema Nicoll, Reference Nicoll1914 includes parasites of snakes: Liolope Cohn, 1902 comprises the type-species Liolope copulans Cohn, 1902 from giant salamanders in East Asia as well as L. dollfusi Skrjabin, Reference Skrjabin1962 from a freshwater pelomedusid turtle in Gabon; Helicotrema Odhner 1912 comprises Neotropical species from iguanas, freshwater turtles and tortoises; Dracovermis Brooks and Overstreet, Reference Brooks and Overstreet1978 and Ngubuvangandu Dutton & Bullard, 2026 comprise species infecting crocodilians; and the monotypic Paraharmotrema karinganiense Dutton & Bullard, 2022, like L. dollfusi, infects freshwater pelomedusid turtles and is known from southeastern Africa (Chin et al., Reference Chin, Ku, Jin and Gu1974; Brooks and Overstreet, Reference Brooks and Overstreet1978; Niewiadomska, Reference Niewiadomska, Gibson, Jones and Bray2002; Dutton et al., Reference Dutton, DuPreez, Urabe and Bullard2022, Reference Dutton, Bullard, Brule and Kelly2024, Reference Dutton, Jacobs, Beytell, Netherlands, DuPreez and Bullard2026). Recently, Numdi and Aisien (Reference Numdi and Aisien2021) reported an unidentified species of Harmotrema from the West African mud turtle Pelusios castaneus (Schweigger) (Pelomedusidae) in Nigeria, but that worm appears to be consistent with P. karinganiense.
Life cycle information for the Liolopidae is known from the type-species L. copulans in Japan (Ozaki and Okuda, Reference Ozaki and Okuda1951; Baba et al., Reference Baba, Hosoi, Urabe, Shimazu, Tochimoto and Hasegawa2011) and for P. karinganiense in southeastern Africa (Donough et al., Reference Donough, Truter, Wepener, Brendonck, Thoré and Smit2025). For both species, natural infections have been reported from freshwater snails as first-intermediate hosts, and freshwater fishes as second-intermediate hosts. Presumably all liolopids require trophic transmission from vertebrate second-intermediate hosts (fishes or conceivably also amphibians), with the possible exception of Helicotrema spp. which are known from mostly herbivorous definitive hosts.
The only liolopid genus with species known from snakes, Harmotrema, currently comprises 5 recognized species. The type species H. infecundum Nicoll, Reference Nicoll1914 is known only from Smith’s African water snake Grayia smythii Günther (Colubridae), from western Africa (Nicoll, Reference Nicoll1914; Dollfus, Reference Dollfus1950). Considering the habits of this semi-aquatic snake, transmission of H. infecundum presumably occurs in freshwater, likely involving fish or amphibian intermediate hosts. Conversely, 3 of the remaining 5 species are known only from marine elapid snakes: H. laticaudae Yamaguti, Reference Yamaguti1933 is known from the blue-lipped sea krait Laticauda laticaudata (Linnaeus) (Elapidae: Hydrophiinae: Laticaudini) in Japan (Yamaguti, Reference Yamaguti1933; Telford, Reference Telford1967), the black-banded sea krait L. semifasciata (Reinwardt) in the Republic of Korea (Choe et al., Reference Choe, Kim, Kim, Lee, Kim and Eom2020; Dutton et al., Reference Dutton, DuPreez, Urabe and Bullard2022), and the olive sea snake Aipysurus laevis Lacépède (Hydrophiinae: Hydrophinii) and the olive-headed sea snake Hydrophis major (Shaw) (Hydrophinii) in Australia (Brooks and Overstreet, Reference Brooks and Overstreet1978), whereas Ha. indicum Chattopadhyaya, Reference Chattopadhyaya1970 emend. and Ha. linguiforme Wang, Reference Wang1987 are known only from the beaked sea snake Hy. schistosus (Daudin) in India and the annulated sea snake Hy. cyanocinctus Daudin in Fujian, China, respectively (Chattopadhyaya, Reference Chattopadhyaya1970; Wang, Reference Wang1987). Presumably, these 3 species are transmitted to their definitive hosts via predation of marine fishes. Predicting the life cycle ecology for Ha. eugari Tubangui & Masilungan, Reference Tubangui and Masilungan1936 is less straightforward. It is known in the Philippines from both a terrestrial snake, the Philippine cobra Naja philippinensis Taylor (Elapidae: Elapinae), and a snake which forages substantially in marine habitats, the Southeast Asian bockadam Cerberus schneiderii (Schlegel) (as C. rynchops) (Homalopsidae) (Tubangui and Masilungan, Reference Tubangui and Masilungan1936; Tubangui, Reference Tubangui1947; Fischthal and Kuntz, Reference Fischthal and Kuntz1967; see also Murphy et al., Reference Murphy, Voris and Karns2012; Bernstein et al., Reference Bernstein, Bautista, Clores, Brown, Ruane, Sanguila, Alis-Besenio, Pejo and Cuesta2024). Finally, Ha. microrchis Bhutta & Khan, Reference Bhutta and Khan1975, from the gharial (Bhutta and Khan, Reference Bhutta and Khan1975), is morphologically and ecologically inconsistent with the genus concept and has been considered a species inquirenda by Dutton et al. (Reference Dutton, Jacobs, Beytell, Netherlands, DuPreez and Bullard2026).
This study reports on liolopids recovered during the first parasitological investigation of marine snakes in Sri Lanka. The specific identities of the recovered specimens were assessed using an integrated approach, and a new classification hypothesis is presented to reflect the significance of marine liolopids.
Materials and methods
Host and parasite collection
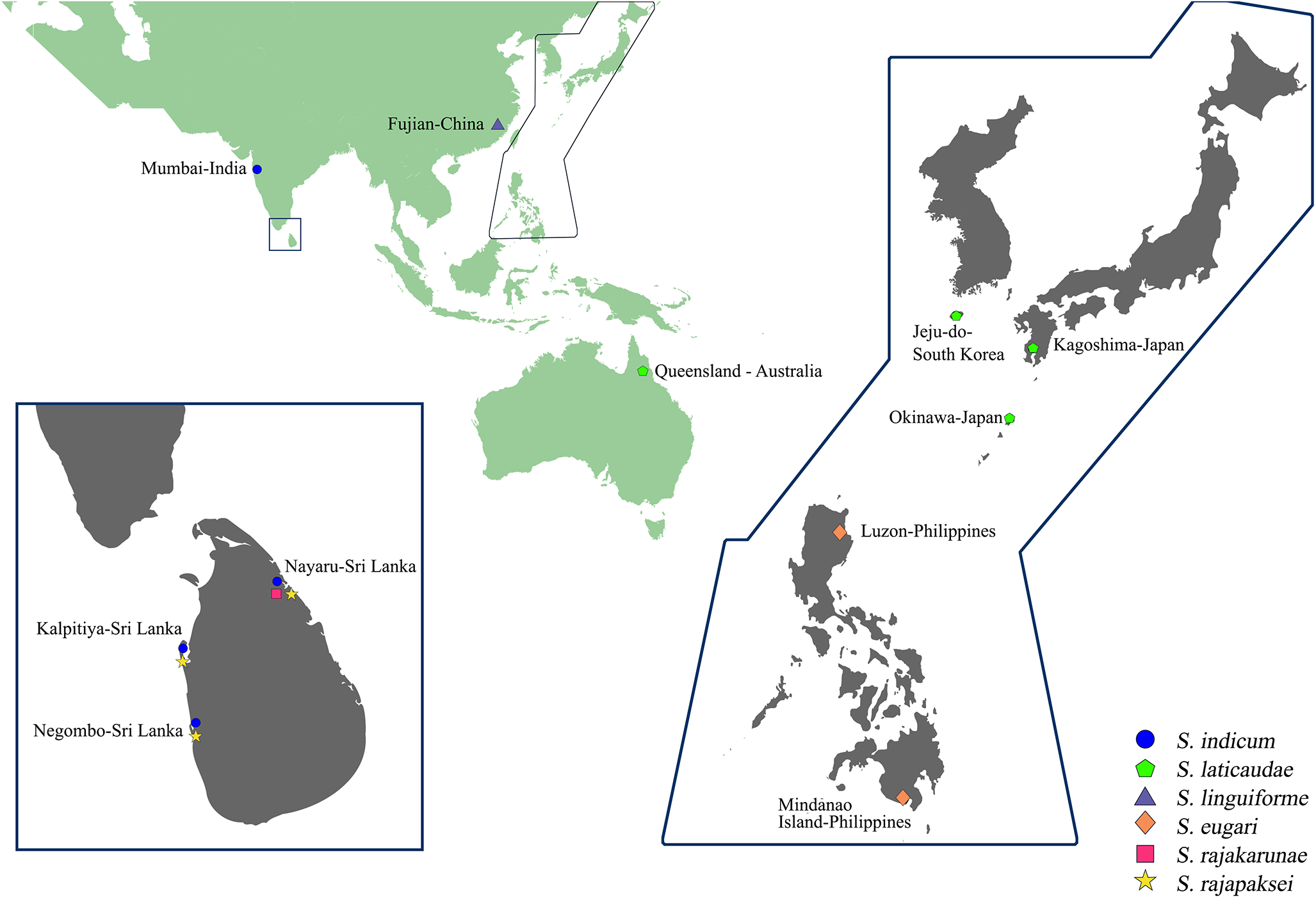
Marine snakes from fishery bycatch were collected between August 2021 and August 2022 in Sri Lanka, from coastal waters of both the Gulf of Mannar, mostly from Portugal Bay, Northwestern Province and the Bay of Bengal, in the vicinity of Nayaru, Northern Province (see Figure 1). Examined snakes comprised 22 individuals belonging to 7 species: 7 little filesnakes Acrochordus granulatus (Schneider) (Acrochordidae) trawled in Portugal Bay, 2 landed at Kalpitiya, 4 landed at Baththalangunduwa and 1 landed at Negombo, 1 South Asian bockadam Cerberus rynchops (Schneider) (Homalopsidae) trawled in Portugal Bay and landed at Baththalangunduwa, 2 ornate reef sea snakes Hydrophis ornatus (Gray) from the Laccadive Sea landed at Dehiwala, 7 beaked sea snakes Hy. schistosus trawled in Portugal Bay and landed at Kalpitiya and Baththalangunduwa and from the Laccadive Sea landed at Negombo, 1 Shaw’s sea snake Hy. curtus (Shaw) and 2 annulated sea snakes Hy. cyanocinctus from the Bay of Bengal landed at Nayaru and 2 yellow sea snakes Hy. spiralis (Shaw), 1 each from the Gulf of Mannar landed at Kalpitiya and the Bay of Bengal landed at Nayaru. Fresh faecal samples were collected from Hy. schistosus by gently massaging/pressing the lower coelom towards the vent. Snakes and faecal samples were isolated in sealable bags, stored on ice and transported to the Division of Parasitology, Department of Veterinary Pathobiology, Faculty of Veterinary Medicine and Animal Science, University of Peradeniya, Sri Lanka. Snakes were examined and necropsied as detailed in Martin et al. (Reference Martin, De Silva, Pathirana and Rajapakse2023), and recovered trematodes were preserved in 70–95% ethanol or 10% formalin.

Figure 1. Landing locations for the marine snakes caught through fisheries bycatch in Sri Lanka and examined for parasites.
Morphological study
Fresh parasite specimens and faecal samples were prepared as temporary mounted slides to observe gross morphology under dissecting and light microscopes. Permanent mounts of whole parasites were prepared, without flattening, from preserved specimens washed in distilled water, stained with Mayer’s haematoxylin and destained with diluted HCl solution (1%), neutralized with diluted NH3 solution (1%), dehydrated in a series of ethanol solutions of 50, 70, 90, 95 and twice in 100%, cleared in methyl salicylate, initially in a 50% solution with 100% ethanol and subsequently in a 100% solution, and finally mounted in Canada balsam. Some specimens preserved in formalin were stained in Van Cleave’s haematoxylin with several drops of Ehrlich’s haematoxylin.
Morphometric data were acquired through live feed using cellSens Standard v1.13, from an Olympus BX50 microscope equipped with Nomarski interference contrast and an Olympus DP71 digital camera with a UCMAD3 adaptor (Olympus Inc., Tokyo, Japan). Line drawings were drawn using a camera lucida from an Olympus BHA phase contrast microscope and subsequently digitized in Adobe Illustrator CC. Measurements are provided in micrometres (μm), with the range followed by mean and standard error in parentheses, length followed by width where applicable, and measurements for eggs averaged from 10 per specimen. Type specimens are lodged in the National Museum of Sri Lanka, Colombo (NMSL), with some paratypes deposited in the Crustacea and Worms collection of the Western Australian Museum, Perth (WAM).
Molecular and phylogenetic study
Novel genetic sequence data were generated from hologenophores to prospect for genetic diversity and potential species richness. Sequences were generated for the cytochrome c oxidase subunit 1 mitochondrial marker (COI mtDNA), the noncoding second internal transcribed spacer unit of ribosomal DNA (ITS2 rDNA) and the large ribosomal subunit gene (28S rDNA). Specimens for molecular analysis were processed, and sequences generated and assembled, in accordance with the protocols in Martin et al. (Reference Martin, De Silva, Pathirana and Rajapakse2023). The partial COI region was amplified and sequenced using the primers Dig_cox1Fa (Wee et al., Reference Wee, Cribb, Bray and Cutmore2017) and Dig_cox1R (Wee et al., Reference Wee, Cribb, Bray and Cutmore2017), the ITS2 region using 3S (Morgan and Blair, Reference Morgan and Blair1995) and ITS2.2 (Cribb et al., Reference Cribb, Adlard and Bray1998), and the partial 28S region using LSU5 (Littlewood, Reference Littlewood1994) and 1500R (Snyder and Tkach, Reference Snyder and Tkach2001).
For each of the 3 markers, the novel sequences were aligned with MUSCLE (Edgar, Reference Edgar2004) in MEGA 11 (Tamura et al., Reference Tamura, Stecher and Kumar2021) using default parameters and compared using simple distance matrices, unrooted neighbour-joining trees and manual inspection of alignments and corresponding chromatograms. The only relevant published sequence included in these alignments was GenBank OL413009 of Dutton et al. (Reference Dutton, DuPreez, Urabe and Bullard2022), representative of 2 identical replicate 28S sequences generated from a collection of 5 specimens identified as Harmotrema laticaudae from a single Laticauda semifasciata at Ishigakai Island, Okinawa Prefecture, Japan, with corresponding morphological vouchers available in the Kyoto University Museum (KUZ Z3989) (M. Urabe, pers. comm.).
A phylogenetic hypothesis based on partial 28S rDNA was reconstructed for the Diplostomida via maximum likelihood analysis. The alignment was trimmed to 1276 nucleotides without masking ambiguously aligned regions and included 1 sequence each for all representatives of the Liolopidae with available data, as well as 1 sequence each per family with available data within the Brachylaimoidea, Diplostomatoidea and Schistosomatoidea (Table 1); a representative of the Transversotrematoidea (Plagiorchiida) was used as the outgroup. The analysis was performed using the implementation of RAxML v8.2.12 (Stamatakis, Reference Stamatakis2014) in the CIPRES portal (Miller et al., Reference Miller, Pfeiffer and Schwartz2010). The analysis assumed the GTR + Γ model of nucleotide substitution and ran 1000 bootstrap pseudoreplicates as determined with the majority rule bootstopping criterion (autoMRE) (Pattengale et al., Reference Pattengale, Alipour, Bininda-Emonds, Moret and Stamatakis2009).
Table 1. 28S rDNA sequence data representative of the taxonomic breadth of Diplostomida and used in the phylogenetic analysis

a Registered name in GenBank as Sanguinicola occidentalis.
Results
Recovered material
Out of 22 marine snakes across 7 species examined, liolopid specimens were recovered from the small intestine of 1 of 1 Hy. curtus, 2 of 2 Hy. cyanocinctus, 6 of 7 Hy. schistosus and 2 of 2 Hy. spiralis. No liolopid infections were detected in 2 Hy. ornatus, 7 A. granulatus and 1 C. rynchops. The recovered liolopids were pale-white, attached firmly to the intestine epithelium, and were already dead despite necropsy of fresh snakes. Yellow-brown, oval eggs were recovered in faecal samples from Hy. schistosus and were presumed to be those of the liolopids as no other trematodes were detected in those particular individual snakes.
Partially digested and undigested prey fishes were identified in 5 species of marine snakes: Hy. curtus contained sprats (Clupeidae), eel tail catfishes (Plotosidae) and garden eels (Congridae); Hy. cyanocinctus contained gobies (Gobidae), Hy. schistosus contained rabbit fishes (Siganidae), croakers (Sciaenidae), terapons (Terapontidae) and puffers (Tetradontidae); and both C. rynchops and A. granulatus contained gobies (Gobiidae). Because these snakes were procured from bycatch, these prey fishes, especially those undigested, might have been predated from fishing nets.
Species recognition
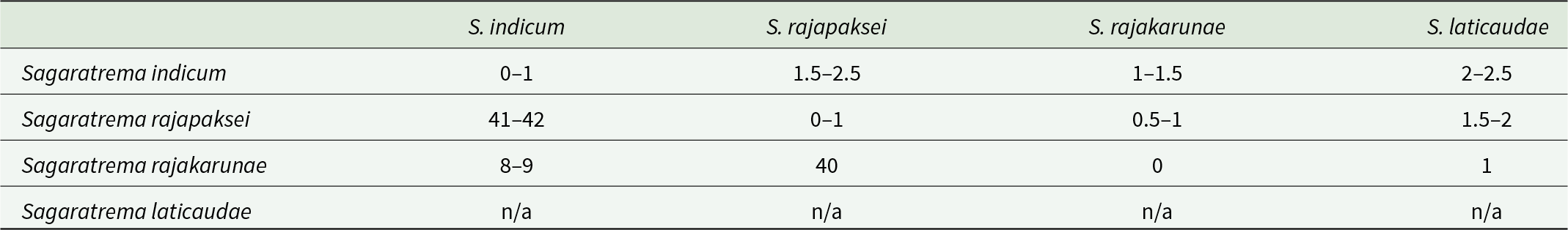
Following iterative investigation of morphology and genetic data, especially from hologenophore specimens, the novel collection from Sri Lanka is interpreted to represent 3 distinct species, all consistent with the current concept of Harmotrema. These species exhibit overlapping host and geographic ranges, including coinfections (Table 2). Generated molecular data comprised 11 partial COI mtDNA sequenced 485 base-positions in length, 9 ITS2 rDNA sequences 485 base-positions in length (including partial flanking 5.8S and 28S) and 8 partial 28S rDNA sequences 1,120 base-positions in length. These data were generated from 18 specimens from Hy. cyanocinctus caught in the Bay of Bengal and Hy. schistosus caught in the Gulf of Mannar. The ITS2 sequences were all identical but the 3 putative species were distinguished in both COI and 28S (Table 3). In COI, the 3 species corresponded to 3 haplogroups with intra-group variation of 0–1 base-positions (Figure 2). The 2 most similar haplogroups differed at 8–9 base-positions (2–2.2%), with a corresponding difference at 1 base-position in 28S. The third COI haplogroup differed to those 2 by 40–42 base positions (10.1–10.6%), with corresponding differences at 1 and 2 base-positions in 28S (some isolates also exhibited intraspecific polymorphisms, see Table 3). Importantly, these 3 genotypes in 28S, differing at 1–2 base-positions in sympatry in Sri Lanka, differed similarly by 1–2 base-positions from the available sequence identified as Harmotrema laticaudae from Japan (GenBank: OL413009; Dutton et al., Reference Dutton, DuPreez, Urabe and Bullard2022, see Figure 2). These 3 putative species are corroborated by morphological distinction, including relative to H. laticaudae. On the basis of morphology, one of the 3 recovered species is identified as H. indicum and the other 2 are considered species new to science.

Figure 2. Unrooted neighbour-joining trees based on COI mtDNA (left) and 28S rDNA (right). Dotted horizontal lines indicate which data were generated from each specimen. The sequence representing S. laticaudae is GenBank OL413009 of Dutton et al. (Reference Dutton, DuPreez, Urabe and Bullard2022); all other sequences are novel. The scale bars measure distance in number of base-positions.
Table 2. Host-parasite-locality combinations for novel material of Sagaratrema n. gen. spp. from Hydrophis spp. reported herein. Dots are positive combinations. Both instances of overlapping combinations included coinfections

Table 3. Pairwise interspecific genetic differences between species of Sagaratrema, in number of base-positions, for partial 28S rDNA (1,120 base-positions) above the diagonal, and partial COI mtDNA (485 base-positions) below the diagonal. Intraspecific genetic variation is included for COI on the diagonal. For 28S, intragenomic polymorphic sites are counted as a difference of 0.5 if one of the 2 nucleotides present is common

Taxonomy
Descriptions for the 3 liolopid species recovered from sea snakes in Sri Lanka are provided below. Following consideration for morphology, biogeography and ecology, a new genus is proposed for liolopids from Indo-West Pacific marine snakes, and Harmotrema is revised.
Phylum Platyhelminthes Minot, 1876
Class Trematoda Rudolphi, 1808
Subclass Digenea Carus, 1863
Order Diplostomida Olson, Cribb, Tkach, Bray & Littlewood, Reference Olson, Cribb, Tkach, Bray and Littlewood2003
Suborder Diplostomata Olson, Cribb, Tkach, Bray & Littlewood, Reference Olson, Cribb, Tkach, Bray and Littlewood2003
Superfamily uncertain
Family Liolopidae Odhner, 1912
Harmotrema Nicoll, Reference Nicoll1914
Type-species: Harmotrema infecundum Nicoll, Reference Nicoll1914
Revised diagnosis (marita): Body dorsoventrally flattened, elongate elliptical, broadest in mid third, gently tapered to bluntly pointed ends, 3–4.5 times longer than wide. Tegument unarmed. Suckers small, feeble; oral sucker slightly larger; ventral sucker in anterior fifth to third of body. Pharynx smaller than oral sucker. Oesophagus short but distinct. Intestine bifurcates in forebody; caeca blind, extend near to posterior end of body. Testes 2, tandem, medial (anterior testis may be dextro-submedial), with regular to irregular margins, well separated, in posterior two-fifths of body; post-testicular zone less than one-fifth of body length. Cirrus-sac large, oblique, spans intercaecal space, in hindbody and posterior half of body, substantially separated from ventral sucker, anteriad of gonads, contains bipartite seminal vesicle, pars prostatica and (presumed) eversible cirrus. Genital pore sinistro-submedial. Ovary dextro-submedial, globular with regular to slightly irregular margin, substantially smaller than and between testes. Mehlis gland large, similar in size to testes. Vitellarium follicular with 2 lateral fields; fields of follicles co-distributed with and strongly constrained about intestinal caeca, extend length of caeca in hindbody and well beyond ventral sucker into forebody but fall well short of intestinal bifurcation, may become confluent in post-testicular zone. Uterus short, intercaecal, between posterior testis and genital pore; oviduct extends posteriorly from ovary. Eggs very few (reported 2–5), very large, larger than ovary, 150–180 long. Excretory duct extends from terminal posterior excretory pore, bifurcates immediately into lateral branches near level of caeca termini, each lateral branch then bifurcates in midbody at the level of anterior testis to form loop with 1 extracaecal and 1 intercaecal branch extending anteriad and reuniting near to level of intestinal bifurcation. In intestine of African colubrid snakes frequenting freshwater.
Remarks: Harmotrema is here proposed to be reduced to monotypy. The revised diagnosis is based on the initial description of the type-species H. infecundum by Nicoll (Reference Nicoll1914) as well as an excellent supplemental description by Dollfus (Reference Dollfus1950) in which additional details were provided, most notably for the excretory system. The 5 species previously recognized in the genus are known only from snakes and are the only liolopids known from snakes. However, of these 5 species, the type-species H. infecundum is comparatively distinctive morphologically and ecologically; a new genus is proposed below for the remaining 4 species, as well as 2 new species, also proposed below. Harmotrema infecundum is known only from Smith’s African water snake Grayia smythii in Western Africa, a colubrid associated with freshwater systems, whereas all the remaining species are known from the Indo-West Pacific and mostly from marine snakes, specifically the viviparous sea snakes (Elapidae: Hydrophinae: Hydrophinii) and the amphibious sea kraits (Hydrophinae: Laticaudini: Laticauda spp.), with 1 species, H. eugari, known from the South Asian bockadam Cerberus schneiderii (Homalopsidae) (as C. rynchops, see Murphy et al., Reference Murphy, Voris and Karns2012) as well as a terrestrial snake, the Philippine cobra Naja philippinensis (Elapidae: Elapinae). Relative to these Indo-West Pacific species, in H. infecundum the loops of the lateral excretory ducts reach posteriorly only to the midbody near to the level of the anterior testis vs span the length of the caeca, the body is smaller and less elongate, the eggs are fewer and larger, the testes more posterior and irregular vs smooth, the forebody is longer (i.e. the ventral sucker is situated more posteriorly), the vitelline zone is longer (the follicles extend significantly into the forebody) and the post-testicular zone is shorter.
Sagaratrema De Silva, Pathirana & Martin n. g.
Type-species: S. laticaudae (Yamaguti, Reference Yamaguti1933) n. comb. (= Harmotrema laticaudae)
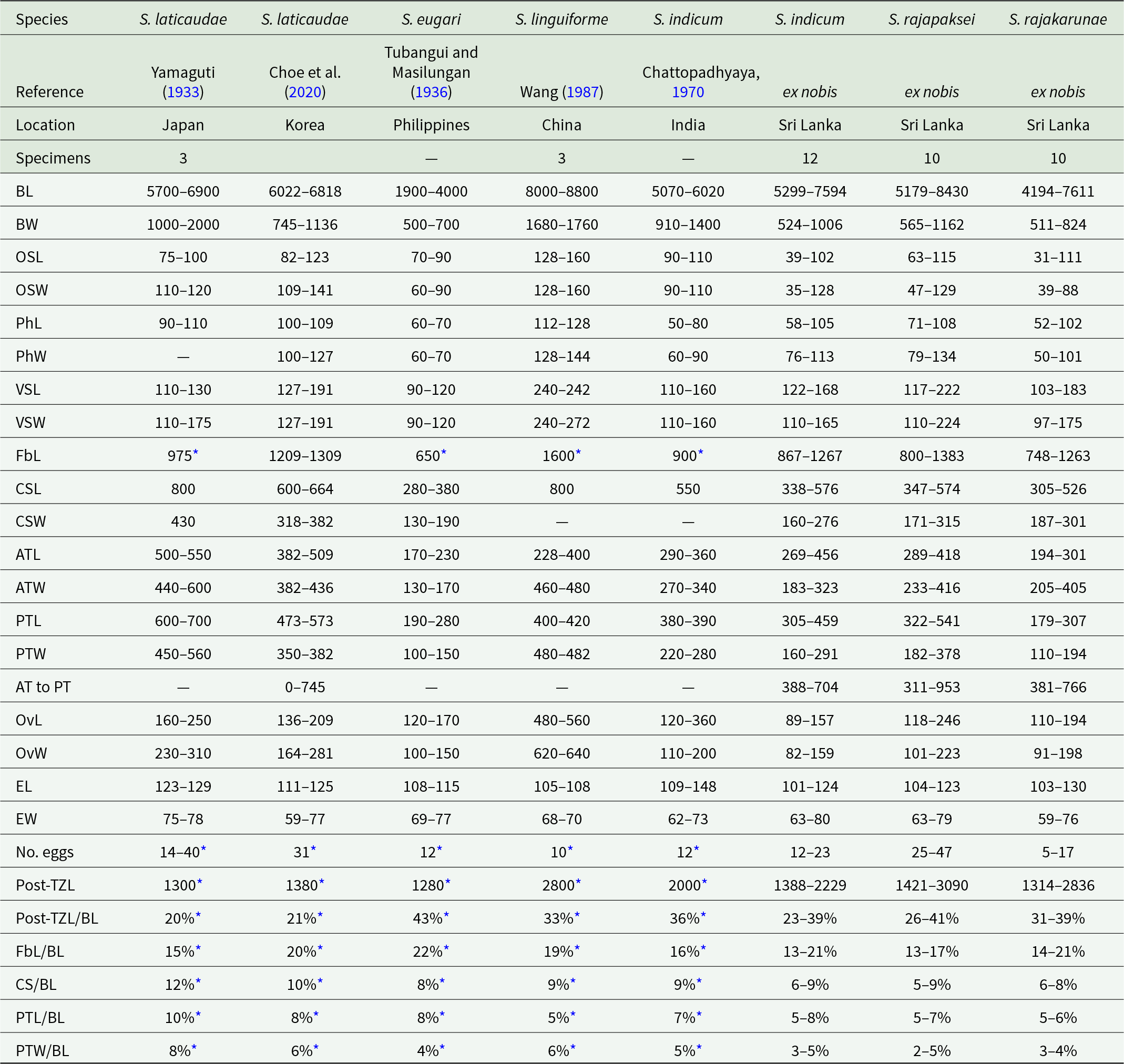
Other recognized species: S. eugari (Tubangui and Masilungan, Reference Tubangui and Masilungan1936) n. comb. (= H. eugari), S. indicum (Chattopadhyaya, Reference Chattopadhyaya1970) n. comb. (= H. indica, H. indicum), S. linguiforme (Wang, Reference Wang1987) n. comb. (= H. linguiforme), S. rajapaksei De Silva, Pathirana & Martin n. sp., S. rajakarunae De Silva, Pathirana & Martin n. sp. (comparative measurement data in Table 4)
Table 4. Morphometric data from reports of Sagaratrema spp

Abbreviations: L, length; W, width; B, body; OS, oral sucker; Ph, pharynx; VS, ventral sucker; Fb, forebody; CS, cirrus-sac; AT, anterior testis; PT, posterior testis; Ov, ovary; E, egg; Post-TZ, post-testis zone.
An asterisk indicates measurement not provided but taken from illustration. A dash indicates data unavailable. Units in micrometres.
Etymology: From Sinhala සාගර (sāgara, =ocean) and trema from trematode, because we hypothesize that at least some species of the genus have marine life cycles, and in honour of the Ocean University of Sri Lanka, where M.L.I. De Silva and E. Pathirana first conceived of this research. The genus is neuter.
Diagnosis (marita): Body dorsoventrally flattened, elongate linguiform, gently tapered to bluntly pointed ends, 4.5–7.5 times longer than wide. Tegument may have minute spines/scales. Suckers small, feeble; oral sucker weakly muscular, terminal to subterminal, smaller than ventral sucker; ventral sucker reduced to circular, flattened disc with central concave depression surrounded by flimsy skirt of muscle, in anterior fifth to quarter of body. Pharynx small, similar in size to oral sucker. Oesophagus short but distinct. Intestine bifurcates in forebody; caeca blind, narrow, extend near to posterior extremity of body. Testes 2, tandem, medial, ellipsoidal, smooth, well separated, in mid hindbody; post-testicular zone substantial, one-fifth to one-third body length. Cirrus-sac large, oblique, spans intercaecal space, in hindbody but anterior half of body, substantially separated from ventral sucker, anterior to gonads, contains bipartite seminal vesicle, pars prostatica and spined, eversible cirrus. Genital pore sinistro-submedial. Ovary medial to dextro-submedial, spherical, smooth, smaller than and between testes. Vitellarium follicular with 2 lateral fields; vitelline reservoir small, posterior to ovary; fields of follicles co-distributed with intestinal caeca, restricted to hindbody (or reach level of ventral sucker or just anteriad), confluent, fill much of intercaecal space in post-testicular zone and between ventral sucker and cirrus sac, mostly excluded from intercaecal space between cirrus-sac and posterior testis. Uterus short, intercaecal, between posterior testis and genital pore, passes ventral to anterior testis; oviduct extends posteriorly from ovary; uterine seminal receptacle in proximal coils; metraterm well-developed. Eggs operculate, many (approximately 20–50), large, smaller than ovary, 105–130 long. Excretory pore subterminal. Excretory vessel divided into 4 parallel vessels; primary vessel bifurcates immediately anterior to excretory pore into paired lateral vessels; lateral vessels each bifurcate near termini of caeca into 1 extracaecal and 1 intercaecal vessel; extracaecal and intercaecal vessels run length of intestinal caeca, reunite in forebody at level of intestinal bifurcation or oesophagus; lateral vessels reconnect via narrow, transverse loop dorsal to pharynx. In intestine of Indo-West Pacific and mostly marine snakes.
Differential diagnosis: Body elongate linguiform with length 4.5–7.5 times width vs ovoid, pyriform or linguiform with length <4.5 width in Liolope, Dracovermis, Ngubuvangandu. Ventral sucker feeble vs strongly muscular in Liolope. Testes entire vs deeply lobed in Paraharmotrema. Post-testicular zone one-fifth to one-third body vs less than one-fifth in Harmotrema, greater than one-half in Helicotrema. Cirrus-sac separated from ventral sucker vs immediately posteriad in Liolope. Vitelline fields confluent vs separate in Harmotrema, Paraharmotrema; restricted to hindbody vs enter forebody substantially in Harmotrema, Liolope, Ngubuvangandu; extend anteriorly near to level of ventral sucker vs at most to level of cirrus-sac in Helicotrema. Eggs large (105–130 µm long) but smaller than in Harmotrema (150–186 µm long), Paraharmotrema (130–160 µm long), many (approximately 5–47) vs fewer in Harmotrema (2–5), Dracovermis (1–7), Ngubuvangandu (< 15), Liolope (up to 23). Excretory vessels with loops spanning length of intestinal caeca vs anterior half in Harmotrema, Paraharmotrema.
Remarks: Sagaratrema is proposed for the liolopids known from Indo-West Pacific and marine snakes; the known geographic range for the recognized species is depicted in Figure 3. These species are united by, and distinguished from, all other liolopids by the combination of the arrangement of the excretory system and the distribution of the vitellarium. In mounted specimens, the ventral sucker sometimes appears muscular with sucker walls folded inwards due to the cover slip pressure. Chattopadhyaya (Reference Chattopadhyaya1970) briefly described minute tegumental spines in S. indicum. We did not detect tegument spines or scales in any of the Sri Lankan specimens when examined under light microscope; imaging with scanning electron microscopy is warranted to understand the nature of the tegument.

Figure 3. Known geographic range for recognized species of Sagaratrema.
Key to species of Sagaratrema
1a. 1 Post-testicular zone one-fifth to one-quarter of body length; cirrus-sac occupies > one-tenth body length …….…………………………………………………............ S. laticaudae
1b. 2 Post-testicular zone approximately one-third body length; cirrus-sac occupies < one-tenth body length ………….... 2
2a. 3 Vitelline follicles constrained laterally, mostly restricted to intercaecal zone (especially anterior to cirrus-sac and posterior to testes)………………………………………….... 3
2b. 4 Vitelline follicles extend laterally beyond caeca near to body margins………………………………………………........ 4
3a. 5 Body approximately 2–4 mm long, vitelline follicles do not reach level of ventral sucker ………………………………………………………………….......... S. eugari
3b. 6 Body approximately 5–6.5 mm long, vitelline follicles always reach level of ventral sucker ……………………………………………….……………….................. S. indicum
4a. 7 Posterior testis wider than longer; egg length <110µm…………………………………………………………………………………………................ S. linguiforme
4b. 8 Posterior testis longer than wide, egg length usually >110 µm…………………………………………............ 5
5a. 9 Eggs number >20, vitelline follicles reach level of ventral sucker ………………………………………... S. rajapaksei
5b. 10 Eggs number <20, vitelline follicles do not reach level of ventral sucker.....................................................S. rajakarunae
Sagaratrema indicum (Chattopadhyaya, Reference Chattopadhyaya1970) n. comb.
Synonyms: Harmotrema indicum Chattopadhyaya, Reference Chattopadhyaya1970; Harmotrema indica Chattopadhyaya, Reference Chattopadhyaya1970
Type-host: Hydrophis schistosus (Daudin) (as Enhydrina schistosa).
Type-locality: Off the coast of Mumbai (as Bombay), India.
Other previous reports: none.
Novel hosts, localities: Hydrophis schistosus from Portugal Bay and the Laccadive Sea landed at Kalpitiya, Baththalangunduwa and Negombo, Sri Lanka; Hydrophis cyanocinctus Daudin from the Bay of Bengal, landed at Nayaru, Sri Lanka.
Material examined: 27 novel specimens, comprising 23 from Hy. schistosus and 4 from 2 Hy. cyanocinctus.
Material deposited: 10 voucher specimens deposited, 4 in NMSL and 6 in WAM, V13583–88 including 4 hologenophores (WAM V13585–88).
Genetic sequences: COI (partial): 3 sequences varying at 0–1 base-positions with 2 deposited, PX979754 and PX979755 representative of 2 and 1 replicates, respectively; ITS2 (with flanking 5.8S and 28S): 3 identical sequences with 1 deposited, PX981823; 28S (partial): 2 identical replicates with 1 deposited, PX981820. All sequences from Hy. schistosus.
Supplementary description (Figure 4A): Based on 12 gravid specimens. Body dorsoventrally flattened, elongate linguiform, broadest in region between ventral sucker and anterior testis, gently tapered to bluntly pointed ends, 5299–7594 (6016 ± 728) × 524–1006 (805 ± 141), length 6.3–11 (7.4 ± 1.5) times width. Forebody 867–1267 (1061 ± 101) or 13–20 (18 ± 2)% of body length. Tegument seemingly unarmed. Oral sucker small, circular, terminal to subterminal, 39–102 (71 ± 21) × 35–128 (101 ± 30). Ventral sucker small, feeble, flattened disc with flimsy muscular walls, 122–168 (146 ± 16.9) × 110–165 (129 ± 18). Pharynx muscular, slightly smaller than oral sucker, 58–105 (73 ± 15) × 76–113 (91 ± 15). Prepharynx indistinct. Oesophagus short. Intestine bifurcated; caeca narrow, mostly straight, terminate near posterior end of body.

Figure 4. Species of Sagaratrema recovered from marine snakes of Sri Lanka. All illustrations from ventral perspective. (A) Sagaratrema indicum (Chattopadhyaya, Reference Chattopadhyaya1970) n. comb., voucher from Hydrophis cyanocinctus. (B) Sagaratrema rajapaksei n. sp., holotype, from Hydrophis curtus. (C) Sagaratrema rajakarunae n. sp., holotype, from Hydrophis cyanocinctus. (D) Sagaratrema rajapaksei n. sp., holotype, terminal genitalia. (E) Sagaratrema rajapaksei n. sp., holotype, ovarian complex. Terminal genitalia and ovarian complex similar among species. Abbreviations: cs, cirrus-sac; ed, ejaculatory duct; gp, genital pore; mg, Mehlis’ gland; o, ovary; pp, pars prostatica; s, spines; sv, seminal vesicle; u, uterus with eggs; um, uterine metraterm; usr, uterine seminal receptacle; vr, vitelline reservoir. Scale bars: A–C, 1 cm; D–E, 200 µm.
Testes 2, tandem, medial, smooth, longer than wide, span intercaecal space, well separated; anterior testis subspherical, 269–456 (356 ± 55) × 183–323 (272 ± 40), separated from cirrus-sac by short distance 196–441 (279 ± 66) or 4–6 (5 ± 0.67)% of body length; posterior testis elongate ellipsoidal, often with transverse notch so as to appear weakly bilobed, 305–459 (375 ± 43) × 160–291 (251 ± 39), separated from anterior testis by 388–704 (558 ± 121) or 7–9 (9 ± 1.5)% of body length; post-testicular zone 1388–2229 (1784 ± 244) or 23–39 (30 ± 5)% of body length. Cirrus-sac large, slightly oblique, 338–576 (448 ± 66) × 160–276 (217 ± 31.7), occupies 6–9 (7 ± 0.9)% of body length, situated 1951–3360 (2235 ± 376) or 34–44 (38 ± 3)% of body length from anterior body extremity, situated 734–1925 (1047 ± 332) or 14–25 (17 ± 3.93)% of body length from ventral sucker. Seminal vesicle internal, bipartite with anterior part slightly smaller. Pars prostatica short, simple; prostatic cells fill space within cirrus-sac around seminal vesicle and cirrus. Cirrus spined. Common genital atrium indistinct. Genital pore sinistro-submedial, intercaecal.
Ovary spherical, dextro-submedial, between testes, closer to posterior testis, 89–157 (120 ± 20) × 82–159 (123 ± 22). Vitelline reservoir postero-dorsal to ovary. Vitelline follicles small, irregularly rounded, co-distributed throughout hindbody along intestinal caeca dorsally and ventrally in 2 lateral, confluent fields, extend anteriorly to level of ventral sucker or sometimes just anterior, laterally more extensive dorsally than ventrally, predominately confined to intercaecal space in zone between ventral sucker and cirrus-sac and in post-testicular zone, excluded from intercaecal space and in asymmetrical patches about caeca in zone between cirrus-sac and posterior testis, in some specimens limited to 1 or 2 patches. Uterus short, intercaecal, constrained between posterior testis and genital pore, ventral to anterior testis; uterine seminal receptacle prominent in proximal part of uterus; metraterm muscular, well developed. Eggs oval, 12–23 (n = 17) in number, 101–124 (110 ± 6.10) × 63–80 (71 ± 5). Excretory pore subterminal. Excretory vessel lateral loops span length of caeca.
Remarks: The novel material is consistent with H. indicum as described by Chattopadhyaya (Reference Chattopadhyaya1970) and is reported here from the same host species, among others, in proximity to the type-locality, at least relative to reports of other known species recognized here in Sagaratrema. Morphologically, the species is recognized by its laterally constrained vitelline distribution. Chattopadhyaya (Reference Chattopadhyaya1970) also reported a transverse notch on the posterior testis, which we observed in some but not all specimens, but also in some specimens of other species. Chattopadhyaya (Reference Chattopadhyaya1970) differentiated S. indicum, in part, for the presence of minute, backward facing tegumental spines. We did not observe spines in our specimens for any species of Sagaratrema and so do not rely on these as a distinguishing characteristic for S. indicum.
Sagaratrema indicum closely resembles S. eugari in which the vitelline follicles are similar by being mostly constrained to the intercaecal space. Chattopadhyaya (Reference Chattopadhyaya1970) distinguished the 2 by the anterior extent of the vitelline follicles, extending to the level of the ventral sucker in S. indicum vs falling short by a small distance in S. eugari. It is not clear for how many specimens Tubangui and Masilungan (Reference Tubangui and Masilungan1936) based their description of S. eugari. Nevertheless, in the novel material identifies as S. indicum, the vitelline follicles reached to the level of the ventral sucker or just beyond anteriorly in all specimens. Furthermore, S. indicum attains greater overall size (length 5070–6020 in the original material and 5385–6349 in the novel material vs 1900–4000 in S. eugari) and appears to have larger eggs (length 109–148 and 101–124 vs 108–115).
Sagaratrema indicum reliably differs from S. laticaudae by having a relatively longer post-testicular zone, smaller testes, a smaller cirrus-sack and fewer vitelline follicles predominantly confined to the intercaecal space. Sagaratrema indicum can be differentiated from S. linguiforme, which attains much larger size (8000–8800 long), has a laterally more extensive vitelline distribution and a subspherical to triangular posterior testis (vs elongate ellipsoidal).
Sagaratrema rajapaksei, De SilvaPathirana & Martin n. sp.
Type-host: Hydrophis curtus (Shaw).
Type-locality: Bay of Bengal, landed at Nayaru, Northern Province, Sri Lanka.
Other hosts and localities: Hydrophis spiralis (Shaw) from the Bay of Bengal landed at Nayaru, and Hydrophis schistosus (Daudin) and Hydrophis spiralis from the Gulf of Mannar including Portugal Bay and landed at Kalpitiya, Baththalangunduwa and Negombo, Sri Lanka.
Site of infection: Intestine.
Material examined: 17 specimens, comprising 14 from Hy. schistosus, 2 from Hy. spiralis and 1 from Hy. curtus.
Material deposited: 11 specimens deposited: holotype and 2 paratypes in NMSL, 8 paratypes in WAM (13568–75) including 4 hologenophores (WAM 13572–75).
Genetic sequences: COI (partial): 5 sequences differing at 0–1 base-positions with 2 deposited, PX979756 and PX979757 representative of 3 and 2 replicates, respectively; ITS2 (with flanking 5.8S and 28S): 5 identical sequences with 1 deposited, PX981824; 28S (partial): 4 identical replicates with 1 deposited, PX981822. All sequences from Hy. schistosus.
Zoobank LSID: https://zoobank.org/urn:lsid:zoobank.org:act:0B18AFE0-A16F-4170-A333-F44808FA6B9A
Etymology: In honour of the esteemed parasitologist, R.P.V.J Rajapakse of the University of Peradeniya, Sri Lanka, in recognition of his significant contributions to the field of parasitology.
Description: (Figures 4 & E) based on 10 gravid specimens. Body dorsoventrally flattened, elongate linguiform, broadest in region between ventral sucker and anterior testis, gently tapered to bluntly pointed ends, 5350–8430 (7114 ± 1088) × 565–1162 (815 ± 226) or 6–15 (9 ± 4) times width. Forebody 886–1383 (1094 ± 159.8) or 13–17 (15 ± 1.3)% of body length. Tegument seemingly unarmed. Oral sucker small, transverse ellipsoidal, terminal or subterminal 63–115 (89 ± 19) × 47–129 (97 ± 31). Ventral sucker small, feeble, flattened disc with flimsy muscular walls, 117–222 (164 ± 31) × 110–224 (155 ± 38). Pharynx muscular, slightly smaller than oral sucker, 71–108 (91 ± 13) × 79–134 (103 ± 17.2). Prepharynx indistinct. Oesophagus short. Intestine bifurcates below oesophagus; caeca narrow, mostly straight, terminate near posterior end of body.
Testes 2, tandem, medial, smooth, longer than wide, span intercaecal space, well separated; anterior testis subspherical, 289–418 (365 ± 49) × 233–416 (301 ± 63.7), separated from cirrus-sac by short distance 145–783 (432 ± 195) or 2–9 (6 ± 2.78)% of body length; posterior testis elongate ellipsoidal, often with transverse notch so as to appear weakly bilobed, 322–541 (426 ± 76.2) × 182–378 (285 ± 60), separated from anterior testis by 311–953 (655 ± 189.4) or 10–13 (11 ± 0.9)% of body length; post-testicular zone 1421–3090 (2377 ± 563.5) or 26–41 (33 ± 6.4)% of body length. Cirrus-sac large, slightly oblique, 347–574 (497 ± 69.8) × 171–315 (244 ± 54.6), occupies 5–9 (6 ± 1.4)% of body length, situated 1753–3258 (2320 ± 475) or 27–39 (32 ± 4.6)% of body length from anterior body extremity, situated 647–1864 (1102 ± 350) or 10–22 (15 ± 4.2)% of body length from ventral sucker. Seminal vesicle bipartite, enclosed with cirrus. Anterior part of seminal vesicle small, overlapping with large posterior vesicle. Pars prostatica short, simple; prostatic cells fill space within cirrus-sac around seminal vesicle and cirrus. Cirrus spined and club shaped. Common genital atrium indistinct. Genital pore sinistro-submedial, intercaecal.
Ovary spherical, dextro-submedial, between testes, closer to posterior testis, 118–246 (169 ± 37.8) × 101–223 (159 ± 46.3). Vitelline follicles small, irregularly rounded, co-distributed throughout hindbody along intestinal caeca dorsally and ventrally in 2 lateral, confluent fields, extend anteriorly to level of ventral sucker or sometimes just anterior, laterally more extensive dorsally than ventrally, extend laterally beyond intestinal caeca near to body margins, mostly excluded from intercaecal space and in 3–5 asymmetrical patches about caeca in zone between cirrus-sac and posterior testis. Uterus short, intercaecal, constrained between posterior testis and genital pore, ventral to anterior testis; uterine seminal receptacle prominent in proximal part of uterus; metraterm muscular, well developed. Eggs oval, 23–47 (n = 17) in number, 104–123 (115 ± 5.8) × 63–79 (69 ± 4.7). Excretory pore subterminal. Excretory vessel lateral loops span length of caeca.
Remarks: Sagaratrema rajapaksei is most similar to S. indicum, and is similarly distinguished from S. laticaudae by a relatively longer post-testicular zone, smaller testes and smaller cirrus-sac, from S. eugari by a larger body and eggs, and relatively larger testes, and from S. linguiforme by the shape of the posterior testis, larger eggs and a smaller body. Sagaratrema rajapaksei is reliably distinguished from S. indicum by the more extensive lateral distribution of the vitelline follicles. In S. indicum, the dorsal and ventral distributions of the vitelline follicles are mostly restricted to the intercaecal space in the post-testicular zone, though extend further laterally to the extracaecal excretory vessels in the zone between the ventral sucker and cirrus-sac. In S. rajapaksei, the dorsal follicles extend laterally more substantially beyond the caeca and extracaecal excretory vessels to almost fill the available space. This distinction also applies to the vitelline follicles in the region of the gonads and cirrus-sac, but less obviously so because, in both species, and indeed in all species of the genus, the vitelline follicles are mostly excluded from the intercaecal space in this mid-body region where their distribution is typically and variously disrupted.
Sagaratrema rajakarunae De Silva, Pathiran & Martin n. sp.
Type-host: Hydrophis cyanocinctus Daudin.
Type-locality: Bay of Bengal, landed at Nayaru, Northern Province, Sri Lanka.
Other hosts and localities: none.
Site of infection: Intestine.
Material examined: 17 novel specimens
Material deposited: 10 specimens deposited: holotype and 2 paratypes in NMSL, 7 paratypes in WAM (V13576–13582) including 2 hologenophores (WAM 13581– 82).
Genetic sequences: COI (partial): 3 identical sequences with 1 deposited, PX979758; ITS2 (with flanking 5.8S and 28S): 2 identical sequences with 1 deposited, PX981825; 28S (partial): 2 identical replicates with 1 deposited, PX981821.
Zoobank LSID: https://zoobank.org/urn:lsid:zoobank.org:act:150F0D94-6A51-4622-86FF-BF01F7323DC8
Etymology: In honour of the esteemed parasitologist, R.S. Rajakaruna of the University of Peradeniya, Sri Lanka, in recognition of her significant contributions to the field of parasitology.
Description: (Figure 4C): Based on 10 gravid specimens. Body dorsoventrally flattened, elongate linguiform, gently tapered to bluntly pointed ends, 4194–7611 (6202 ± 1373) × 511–824 (686 ± 100) or 7–11 (9 ± 1.21) times width. Forebody 748–1263 (1039 ± 182) or 14–21 (16 ± 2.4)% of body length. Tegument seemingly unarmed. Oral sucker small, circular, terminal to subterminal 31–111 (71 ± 32) × 39–88 (67 ± 18). Ventral sucker small, feeble, flattened disc with flimsy muscular walls, 103–183 (147 ± 32) × 97–175 (134 ± 23). Pharynx muscular, slightly smaller than oral sucker, 52–102 (72 ± 16.3) × 50–101 (78 ± 15.3). Prepharynx indistinct. Oesophagus short. Intestine bifurcates level of oesophagus; caeca narrow, mostly straight, terminate near posterior end of body.
Testes 2, tandem, medial, smooth, longer than wide, span intercaecal space, well separated; anterior testis subspherical, 194–357 (295 ± 33.5) × 194–301 (244 ± 33.5), separated from cirrus-sac by short distance 172–558 (321 ± 117.4) or 3–7 (5 ± 1.4)% of body length; posterior testis elongate ellipsoidal, often with transverse notch so as to appear weakly bilobed, 205–405 (326 ± 60) × 179–307 (235 ± 46.5), separated from anterior testis by 381–766 (598 ± 132) or 7–12 (9 ± 1.7)% of body length; post-testicular zone 1314–2836 (2209 ± 568) or 31–39 (35 ± 2.7)% of body length. Cirrus-sac large, slightly oblique, 305–526 (432 ± 62.4) × 187–301 (252 ± 30.9), occupies 6–8 (7 ± 0.8)% of body length, situated 1613–2539 (2149 ± 362.1) or 28–38 (33 ± 3.7)% of body length from anterior body extremity and 629–1272 (962 ± 192.2) or 11–17 (15 ± 2.5)% of body length from ventral sucker. Seminal vesicle internal, bipartite, enclosed with cirrus with anterior part slightly smaller. Pars prostatica short, simple. Cirrus spined. Common genital atrium indistinct. Genital pore sinistro-submedial, intercaecal.
Ovary spherical, dextro-submedial, between testis, closer to posterior testis, 110–194 (152 ± 30.7) × 91–198 (147 ± 34). Vitelline reservoir postero-dorsal to ovary. Vitelline follicles small, irregularly rounded, co-distributed throughout hindbody along intestinal caeca in 2 lateral, confluent fields, extend anteriorly near to but separated from posterior margin of ventral sucker, laterally more extensive dorsally than ventrally, excluded from intercaecal space and in asymmetrical patches about caeca in zone between cirrus-sac and posterior testis. Uterus short, intercaecal, constrained between posterior testis and genital pore, ventral to anterior testis; uterine seminal receptacle prominent in proximal part of uterus; metraterm muscular, well developed and occupy posterosinistral region of cirrus sac. Eggs oval, very few (5–17) (n = 20), 103–130 (118 ± 9.8) × 59–76 (68 ± 4.8). Excretory pore subterminal. Excretory vessel lateral loops span length of caeca.
Remarks: Sagaratrema rajakarunae is morphologically similar to S. rajapaksei, yet is much closer to S. indicum in COI mtDNA (Table 2 and Figure 2). Like in S. rajapaksei, in S. rajakarunae the distribution of the vitelline follicles is laterally extensive, differentiating it from S. eugari and S. indicum. However, in S. rajakarunae, the anterior extent of the vitelline follicles falls short of reaching the level of the ventral sucker by a small but consistent margin, whereas in S. rajapaksei the vitelline follicles always reach at least to the level of the ventral sucker and sometimes a little beyond anteriorly. Additionally, S. rajakarunae consistently has fewer eggs, 5–17 vs 23–47 in S. rajapaksei. A comparable gap between the vitelline follicles and ventral sucker is also characteristic of S. eugari and has been depicted for some material identified as S. laticaudae, specifically by Choe et al. (Reference Choe, Kim, Kim, Lee, Kim and Eom2020). Like S. rajapaksei, S. rajakarunae is distinguished from S. laticaudae by a relatively longer post-testicular zone, smaller testes and a smaller cirrus-sack.
Sagaratrema rajakarunae can be distinguished from S. linguiforme by shape of the posterior testis, larger eggs and smaller overall size.
Phylogenetic results
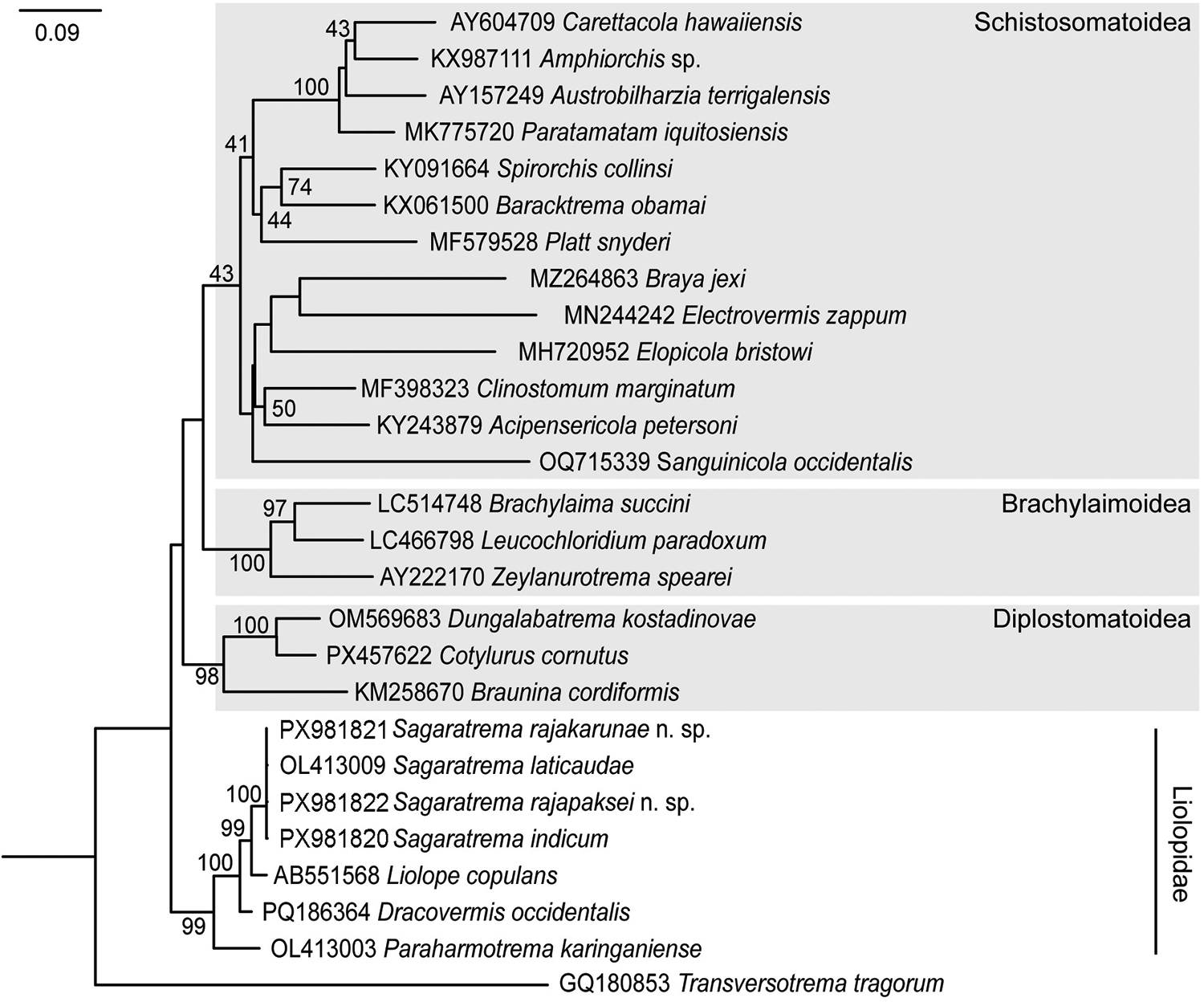
The phylogenetic hypothesis reconstructed for the Diplostomida via maximum likelihood analysis based on partial 28S recovered each of the Brachylaimoidea, Diplostomoidea and Schistosomatoidea as monophyletic (Figure 5). The Liolopidae was recovered as monophyletic and basal within the Diplostomida. Relationships between these major clades were poorly supported, but the topology is consistent with some recent previous analyses (e.g. Cutmore et al., Reference Cutmore, Littlewood, Arellano-Martínez, Louvard and Cribb2023). Within the Liolopidae, species of Sagaratrema formed a strongly supported and derived clade, sister to the sequence for Liolope copulans.

Figure 5. Hypothetical phylogenetic reconstruction for the Diplostomida based on maximum likelihood analysis of partial 28S rDNA. Bootstrap support less than 40 omitted. The scale bar indicates the expected number of substitutions per site.
Discussion
The marine liolopids: a return to the sea
Central to the proposal of Sagaratrema is the hypothesized ecological, host and biogeographic distinctions separating those species, parasites of mostly marine elapid snakes in the Indo-West Pacific, from the now monotypic Harmotrema infecundum, known only from a colubrid associated with freshwater in western Africa. These distinctions are interpreted here to reflect substantial separation in evolutionary history, and this is consistent with the morphological distinctions recognized here as the basis for the new genus concept.
We hypothesize that at least some species of Sagaratrema have entirely marine life cycles, that is, that they use a marine snail first intermediate host and marine fishes as second intermediate hosts, in addition to marine snakes as definitive hosts. Furthermore, we hypothesize that the origins of the Liolopidae were in freshwater, and that Sagaratrema is the only liolopid lineage that has subsequently invaded marine environments. Finally, this hypothesized narrative is framed as a return to the sea, because, although we suspect that the last common ancestral liolopid was a freshwater species, the origins of the Digenea are presumably marine (Cable, Reference Cable and Vernberg1974; Cribb et al., Reference Cribb, Bray and Littlewood2001; Cribb et al., Reference Cribb, Bray, Olson and Littlewood2003).
The evidence supporting each of the components of the above evolutionary narrative is variously compelling. There are no records of first intermediate or second intermediate hosts for any species of Sagaratrema, and so the species examined here from Sri Lanka are hypothesized to be entirely marine based on the habits of the definitive hosts. Of the more than 70 extant species of substantially marine snakes (Rasmussen et al., Reference Rasmussen, Murphy, Ompi, Gibbons and Uetz2011), the viviparous sea snakes (Elapidae: Hydrophinae: Hydrophinii) are the richest and most specialized for the marine environment. Although hydrophins can sometimes be found significant distances upstream in rivers (Rasmussen, Reference Rasmussen, Carpenter and Volker2001), most species typically have restricted activity ranges (Burns and Heatwole, Reference Burns and Heatwole1998; Udyawer et al., Reference Udyawer, Simpfendorfer and Heupel2015), and the snakes sampled here were procured from fishermen operating in inshore marine waters vs estuarine habitats. Therefore, we think the first intermediate hosts are marine snails found in proximity to the sampled snakes; the possibility of amphidromous or estuarine fishes transmitting infections from freshwater or estuarine snails to marine snakes is unlikely, because the parasites still require transport from the snakes back to the snail hosts.
We predict a freshwater origin for the Liolopidae given that the earliest-branching lineages within the family have known or presumed freshwater life cycles (Paraharmotrema and then Dracovermis, see Figure 5 and Dutton et al., Reference Dutton, Jacobs, Beytell, Netherlands, DuPreez and Bullard2026), and that no liolopids other than Sagaratrema spp. are suspected to have marine life cycles. Additionally, the last common ancestor of the Liolopidae presumably predates the recent origins of marine elapids; the viviparous sea snakes and amphibious sea kraits transitioned independently from a terrestrial existence approximately 6–20 million years ago (Sanders et al., Reference Sanders, Lee, Bertozzi and Rasmussen2013; Lee et al., Reference Lee, Sanders, King and Palci2016; Heatwole et al., Reference Heatwole, Grech and Marsh2017; Kim et al., Reference Kim, Park, Suk, Bae, Min, Tsai and Park2018; Sherratt et al., Reference Sherratt, Rasmussen and Sanders2018). Although, time calibration of digenean phylogeny is extremely limited (see De Baets et al., Reference De Baets, Dentzien-Dias, Upeniece, Verneau and Donoghue2015), the basal position and long branch separation of the Liolopidae within the Diplostomida, together with the broad geographic and definitive host range for the family (see Figure 6), suggests an origin comfortably older than for marine snakes. Indeed, Brooks and Overstreet (Reference Brooks and Overstreet1978) hypothesized a Gondwanan last common ancestor for the Liolopidae no later than the Cretaceous.

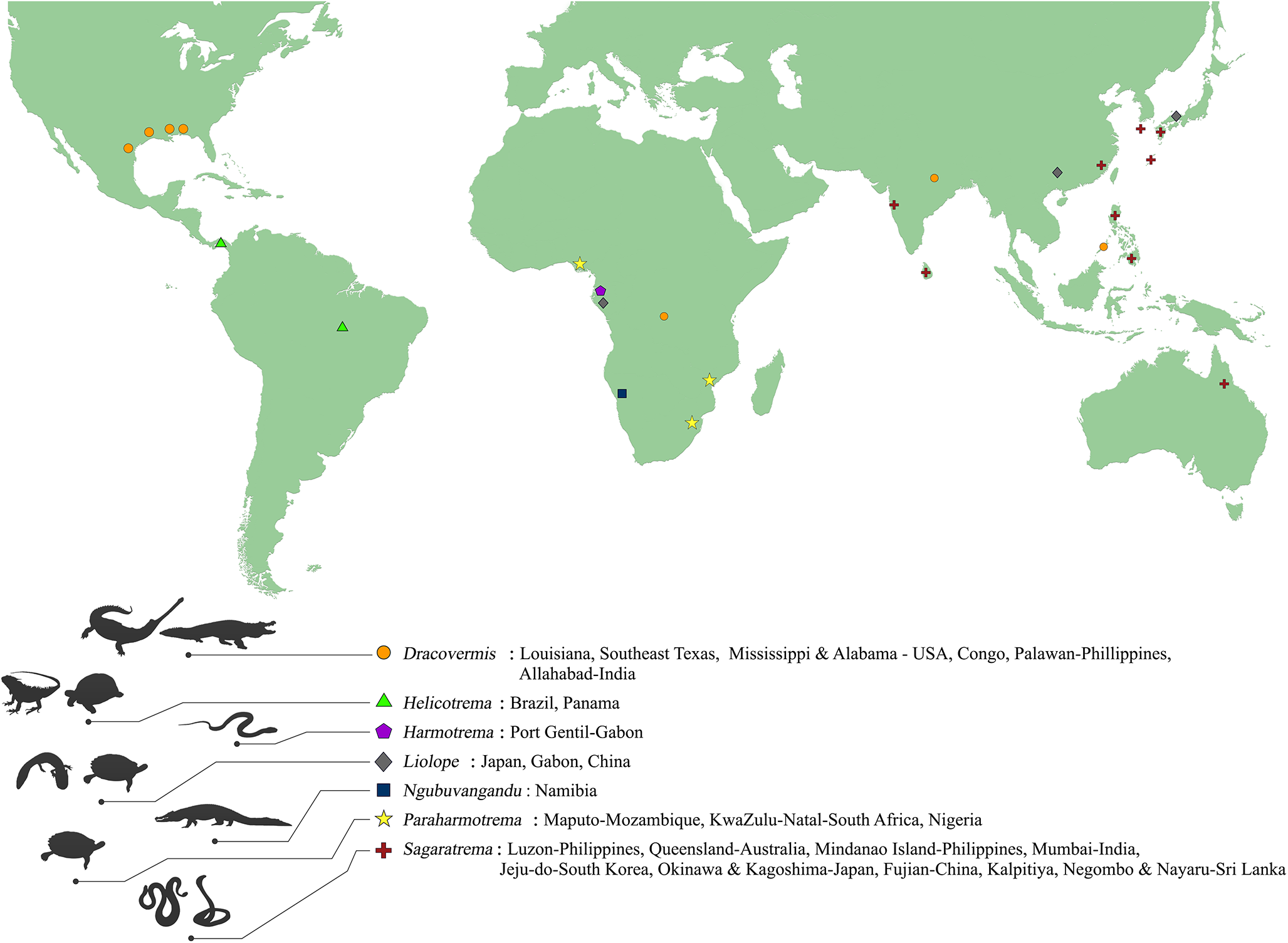
Figure 6. Global distribution of the 7 known genera within the family Liolopidae Dollfus, 1934.
Accepting a freshwater life cycle for the last common ancestral liolopid and that at least some species of Sagaratrema have marine life cycles, it is then either the case that ancestral species of Sagaratrema co-invaded the marine environment with their snake hosts or switched into marine snakes more recently. Both explanations are plausible. Regardless, our hypothesized evolutionary narrative requires that a host switch from freshwater to marine snails occurred, possibly with interim switches through estuarine snails. These first intermediate host switches could have been facilitated by the transitional period in the evolution of marine snakes from terrestrial ancestors (see Heatwole et al., Reference Heatwole, Grech and Marsh2017). However, it is alternatively possible that the parasites host-switched into a transitional snake species (whether ancestral or contemporary) and then subsequently into marine snails, such that it is not necessarily the case that the parasites co-invaded the marine environment with their snake hosts.
The missing link?
The potentially important and enigmatic exception to the ecological and evolutionary narrative proposed here is S. eugari, the only species included in the new genus reported from snakes other than hydrophine elapids. Instead, it is known from the Philippine cobra, a terrestrial species, and the Southeast Asian bockadam, a semi-aquatic, mostly piscivorous and substantially marine species which also ranges through brackish, estuarine and freshwater habitats (Bernstein et al., Reference Bernstein, Bautista, Clores, Brown, Ruane, Sanguila, Alis-Besenio, Pejo and Cuesta2024). Regardless, S. eugari is entirely consistent with Sagaratrema morphologically and shares none of the characters distinguishing H. infecundum from that concept, except, perhaps, that S. eugari is the smallest species of the new genus, comparable in size to H. infecundum.
Presuming that S. eugari indeed forms a natural group with the remaining species of Sagaratrema, the implications for our hypothesized evolutionary narrative depend on 2 pieces of information which are currently unknown: the phylogenetic position of S. eugari, basal or derived, relative to other species of Sagaratrema, and the specific ecology of the life cycle, that is, whether it exploits freshwater, estuarine or marine snails. Obtaining genetic and life cycle data for S. eugari and confirming its typical definitive hosts is therefore of particular interest and consequence.
Richness, host-specificity and difficulty in species delineation
This study increases the number of liolopid species known from Indo-West Pacific marine snakes from 4 to 6 and is the first study to interpret a collection of such material as comprising more than a single species. The morphological and molecular distinctions recognized between species of Sagaratrema are slight. In particular, ITS2 rDNA, the most commonly used genetic marker for delineation of digenean species (Blasco-Costa, Reference Blasco-Costa2025), exhibited no variation among the 3 species from Sri Lanka. Critical to the delineation of these 3 species was that the 28S rDNA genotypes are similarly distinct in sympatry as each is relative to S. laticaudae from Japan, and that these genotypes were consistently corroborated by both COI mtDNA haplotypes and morphotypes.
Furthermore, our modest sampling of snakes found overlapping host and geographic ranges for the 3 species of Sagaratrema detected from Sri Lanka, including coinfections of S. indicum with both S. rajapaksei and S. rajakarunae. Thus, host-specificity is seemingly relaxed among hydrophins, and collections from even a single snake require scrutiny. Reliance on hologenophore specimens was therefore critical and any future investigation of novel Sagaratrema collections should use this approach to prospect for possible genetic diversity among morphologically conserved material.
The known richness of Sagaratrema is much lesser than for marine snakes (some 70 species, see Rasmussen et al., Reference Rasmussen, Murphy, Ompi, Gibbons and Uetz2011). We think it likely that several more species of Sagaratrema are yet to be discovered, as marine snakes have been scarcely examined for helminths and the subtle morphological distinctions detected here allows the possibility that previous efforts unaided by genetic inference might have underestimated richness. However, we also think it unlikely that the richness of Sagaratrema will prove to match that of marine snakes, because many of the hydrophine elapids have specialized diets and so are seemingly unavailable hosts (Voris et al., Reference Voris, Voris and Liat1978; Voris and Voris, Reference Voris and Voris1983; Shine et al., Reference Shine, Bonnet, Elphick and Barrott2004; Li et al., Reference Li, Fry and Kini2005; Brischoux and Lillywhite, Reference Brischoux and Lillywhite2011; De Silva et al., Reference De Silva, Sivaruban, Ukuwela, Rasmussen and Sanders2011).
Contribution to the fauna of Sri Lanka
Together with Tubulovesicula laticaudae Parukhin, 1969 (Digenea: Hemiuridae) reported from this same investigation of marine snakes (Martin et al., Reference Martin, De Silva, Pathirana and Rajapakse2023), the liolopids reported herein are the first parasites to be reported from marine snakes in Sri Lanka. Furthermore, S. rajapaksei and S. rajakarunae are the first new parasitic helminth species from marine wildlife in Sri Lanka to be proposed on the basis of an integrated taxonomic approach including inference from genetic data.
Data availability statement
Raw morphometric data are publicly and freely available at https://data.mendeley.com/datasets/33zjgbd3pc/1
Acknowledgements
We thank the Department of Wildlife Conservation, Sri Lanka for granting the research permit (WL/3/7/11/2022), members of the local fisher communities, and our colleagues YMNP Abeyrathne and R Rajapakse and for assistance with data collection, DPGSP Jayasinghe, A Pallekumbura, A Jayaweera, AMU Abeykoon, A Ranaraja, OC Pathirana and WR Jayaweera for assistance in postmortem examinations, SA Bullard and HR Dutton (Auburn University) for preliminary assistance and guidance in taxonomy of liolopids, M Urabe (University of Shiga Prefecture) for providing photographs and details of the specimens represented by GenBank OL413009 (Harmotrema laticaudae), F Brigg (Western Australian State Agricultural Biotechnology Centre, Murdoch University) for Sanger sequencing and 2 anonymous reviewers for their insightful and constructive remarks.
Author contributions
MLIDS and EP conceived and designed the study. EP enabled field and laboratory work in Sri Lanka. MLIDS collected specimens, generated molecular and morphological data, and prepared species descriptions. SBM provided supervision and training, and directed the manuscript. MLIDS prepared the draft and all authors contributed to the final manuscript.
Financial support
This research was supported by the Australian Government Research Training Program Scholarship, an Australian Biological Resources Study (ABRS) National Taxonomy Research Grant (NTRG) G046WN7, an Australia and Pacific Science Foundation research grant APSF21048, the Nippon Foundation: Nekton Ocean Census programme, and funds from the Centre for Sustainable Aquatic Ecosystems, Harry Butler Institute, Murdoch University.
Competing interests
The authors declare there are no conflicts of interest.
Ethical standards
This study was conducted in compliance with all institutional, national and international guidelines on the care and use of animals.