Introduction
The transition from the Late Archaic Period (3300–1000 BCE) to the Formative Period (1000 BCE–900 CE) in the Atacama Desert was marked by substantial changes in the lifestyles of human groups, shifting from predominantly hunter-gatherer subsistence to the incorporation of agriculture and more sedentary ways of life (Muñoz et al., Reference Muñoz, Agüero, Valenzuela, Falabella, Uribe, Sanhueza, Aldunate and Hidalgo2016). This transformation was accompanied by the adoption of new productive and technological practices, such as pottery (Agüero et al., Reference Agüero, Ayala, Carrasco, Cases, Uribe and Lechtman2006; Uribe, Reference Uribe, Zarankin and Acuto2008; Uribe et al., Reference Uribe, Angelo, Capriles, Castro, De Porras, García, Gayo, González, Herrera, Izaurieta and Maldonado2020). On the coast of northern Chile, this new technology has been associated with cooking and the introduction of new recipes and culinary practices (Correa et al., Reference Correa, Carrasco, Ballester and Gallardo2018). The archaeological site Aragón-1, located in the Jazpampa ravine in the Pampa del Tamarugal (Tarapacá region, Northern Chile), is 30 km from the Pacific Coast and at an altitude of 1100 masl (Figures 1 and 2). The site’s occupation spans from 3400 BCE to 1400 CE. Despite receiving less than 5 mm of annual rainfall (Huston, Reference Huston2006), the presence of groundwater and sporadic surface water sustains diverse vegetation in the Aragón-1 site (Núñez and Zlatar, Reference Núñez and Zlatar1976; Uribe et al., Reference Uribe, Angelo, Capriles, Castro, De Porras, García, Gayo, González, Herrera, Izaurieta and Maldonado2020; Acosta and Rivera, Reference Acosta and Rivera2022). It has been proposed that human groups at Aragón-1 had a major dependence on coastal resources and practiced a marine-based lifestyle (Núñez and Zlatar, Reference Núñez and Zlatar1976, Reference Núñez and Zlatar1977), as evidenced by the presence of shellfish and fish (fish scales, bones and otoliths), together with associated technologies (i.e., cactus fishhooks) found at the site. Zooarchaeological analyses show the constant presence of marine resources throughout the occupation of Aragón-1. Species such as T. murphyi (Chilean Jack Mackerel) and various species of the Clupeidae family, for example, sardines and anchovies, are found in great numbers throughout most of the occupation, with many fish bones showing exposure to fire (González, unpublished results). Marine resources were probably part of the Aragón-1 people’s diets, implying a high risk of enteroparasitic infection. These findings also suggest frequent coast-inland mobility, and strong interaction between these ecological zones and populations to access these resources (Núñez and Zlatar, Reference Núñez and Zlatar1976, Reference Núñez and Zlatar1977; Núñez and Hall, Reference Núñez and Hall1982).
Map of Northern Chile indicating the location of Aragón-1 and other archaeological sites.

Panoramic view of the Aragón-1 archaeological site and the Jazpampa ravine (looking west).

Palaeoparasitological studies provide valuable information on the presence of parasites in human groups and their relationship with the diverse lifestyles and cultural practices of past populations (Araujo et al., Reference Araujo, Reinhard and Ferreira2008; Fugassa, Reference Fugassa2009; Araújo et al., Reference Araújo, Reinhard and Ferreira2015). Parasitic infections have been reported in several archaeological sites of the Atacama Desert. Insufficient hygiene and overcrowding, resulting from a gradual increase in sedentary lifestyle, the intake of insufficiently cooked or spoiled food, and contact with infected animals (zoonosis), could be the cause or influence the proliferation of parasites (Retamal et al., Reference Retamal, Pacheco and Uribe2006; Rodríguez, Reference Rodríguez2012; Castells et al., Reference Castells, González and Westfall2010). Palaeoparasitological studies of coprolites from northern Chile and digestive tract contents recorded the presence of Paragonimus sp. (Núñez and Hall, Reference Núñez and Hall1982), Enterobius vermicularis (Araújo et al., Reference Araújo, Ferreira, Confalonieri, Nuñez and Ribeiro Filho1985; Correa and Ferreira, Reference Correa and Ferreira2016; Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021), Trichuris trichiura, (Castells et al., Reference Castells, González and Westfall2010; Correa and Ferreira, Reference Correa and Ferreira2016; Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021), Ascaris lumbricoides (Castells et al., Reference Castells, González and Westfall2010), strongylids (Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021) and Dipylidium caninum (Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021). Parasites of the genus Dibothriocephalus/Adenocephalus sp. (fish tapeworm) have also been registered in inland and coastal sites from the Tarapacá and Antofagasta regions in northern Chile, throughout the Archaic Period (10000–1000 BCE), Formative Period (1000 BCE–900 CE) and Late Intermediate Period (900–1450 CE) (Araujo and Remington, Reference Araujo and Remington1980; Reinhard and Urban, Reference Reinhard and Urban2003; Castells et al., Reference Castells, González and Westfall2010; Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021). This parasitic genus was also documented in the Tiliviche-1B and Pisagua 7 sites, 6.5 and 30 km from Aragón-1, respectively. (Ferreira et al., Reference Ferreira, Araújo, Confalonieri and Nuñez1984; Araújo et al., Reference Araújo, Ferreira, Confalonieri, Nuñez and Ribeiro Filho1985; Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021). The presence of Dibothriocephalus/Adenocephalus sp. is linked to the consumption of raw or undercooked fish (Scholz et al., Reference Scholz, Garcia, Kuchta and Wicht2009; Centers for Disease Control and Prevention, n.d.). Parasitic infections require a combination of biological, cultural and ecological conditions that enable specific parasite transmission pathways (Neghme and Silva, Reference Neghme and Silva1971). In this context, subsistence strategies, food preparation techniques and settlement patterns, together with environmental factors, influence the establishment of specific parasitic infections. Together with the palaeoparasitological record, this indicates that the presence of parasites reflects the relationship between cultural practices and the environment, both of which contribute to conditions that favour parasite reproduction (Rodríguez, Reference Rodríguez2012).
Human coprolite samples were recovered throughout the entire stratigraphic sequence of the Aragón-1 occupation. The climatic conditions of the Atacama Desert have favoured the privileged conservation of these archaeological remains. A well-documented and dated stratigraphic sequence conducted at Aragón-1 enabled chronological comparisons within the site. This allowed palaeoparasitological analysis to be carried out considering a chronological context of transition to agriculture, in which important cultural transformations occurred. Following this, the aim of this study was to evaluate human–parasite interactions, through palaeoparasitological analysis of human coprolites, in order to understand the lifestyles in Aragón-1 and their relationship with the environment during the Late Archaic and Formative Periods.
Materials and methods
Paleoparasitological analysis was conducted on 22 different human coprolites (Figure 3). Three excavations were carried out at the Aragón-1 site: the North Sector unit, the South Sector unit and the 2019 unit. A total of 84 human coprolites were obtained from the South Sector and 2019 excavation units. From these, a subset of 22 coprolites, recovered from stratigraphic units with radiocarbon dates, was selected based on their weight and preservation. Furthermore, the aim of the sampling was to achieve a balanced representation of the Archaic and Formative archaeological Periods, thereby enabling meaningful statistical comparisons. Within the Formative Period, we also sought to include specimens from both the early (1000 BCE–200 CE) and late phases (200–900 CE).
Human coprolite sample CP112.

The human origin of the coprolites is inferred based on their morphology and the faunal composition of the site. No other animal species documented at Aragón-1 produces faecal remains with characteristics that could be mistaken for the coprolites analysed in this study. Additionally, humans are the only omnivorous mammals present in the study area during the Periods analysed, further supporting the attribution.
Among the selected samples, 14 were collected from the South Sector unit, and 8 from the 2019 unit. Of the samples from the South Sector, 11 belong to the Late Archaic Period, with radiocarbon dates ranging from 3300 BCE to 1000 CE. The remaining 3 are dated to the Early Formative Period (800–400 BCE). All eight coprolites from the 2019 unit date to the Late Formative Period, spanning from 240 to 530 CE (Table 1).
Summary of palaeoparasitological findings per sample

Sample preparation
The samples were processed at the Laboratory of Archaeology in the Pontificia Universidad Católica de Chile following a 3-step protocol. First, the coprolite samples (Callen and Cameron, Reference Callen and Cameron1960) were rehydrated, followed by spontaneous sedimentation (Lutz, Reference Lutz1919) and optical microscopy observation. During the rehydration stage, 0.32–5.82 g of coprolite (Table 1), depending on the sample size, were placed in 50 mL 0.5% trisodium phosphate (0.5% Na3PO4) (Callen and Cameron, Reference Callen and Cameron1960). The coprolite samples were dissolved in the solution and subsequently incubated at 4°C for 72 h to prevent fungal proliferation, which could damage the sample and hinder observations. The aim of rehydrating the samples is to break down the various elements and particles attached to the parasitic remains and restore their morphological characteristics for identification. The human origin of the samples was also evaluated during the rehydration process, in which a dark-coloured solution is generally considered a good indicator of human origin (Reinhard and Bryant, Reference Reinhard, Bryant and Schiffer1992; Pearsall, Reference Pearsall2016; Reinhard, Reference Reinhard2017). The rehydrated samples were then concentrated through decantation at room temperature for 24 h in a conical measure using a mesh (1 × 1 mm pore size) to filter macroremains that could hamper the visualization of parasites. This step aims to concentrate all microremains and parasitic structures at the bottom of the conical glass, from which a 250 μL aliquot is recovered from each sample for microscopic observation.
Each 250 μL sample was subdivided into ten 25 μL aliquots, mixed with 50 μL glycerine, mounted onto glass slides, and covered with coverslips. Each sample was examined in its entirety using a Leica DM750 optical microscope at 100X (total magnification). The parasitic remains were photographed at 400X using a Leica ICC50 W camera and the LAS-V4.12 software for image acquisition and analysis. The taxonomic identification of parasites was based on their morphological and morphometric characteristics (length and width, measured in micrometres [µm]), using the manual ‘Laboratory identification of parasites of public health concern’ (Centers for Disease Control and Prevention, n.d.) and previous published parasitological and paleoparasitological literature as reference guides. Parasite eggs were measured using the LAZ-V4.12 programme. Only intact and undeformed parasite eggs were measured. Broken, crushed or altered parasite eggs were only recorded and photographed.
Statistical analysis
Considering the egg counts obtained and the mass of each analysed coprolite, eggs per gram (EPG) were calculated for each sample. Based on these values, statistical analyses were conducted using R: The RStudio Team (2023). A binomial probability test (Bitest) was performed to evaluate differences in the number of eggs between the different archaeological periods. Additionally, a Spearman correlation analysis was conducted to assess if the coprolite mass and egg frequency were related. Finally, the mean and standard deviation of egg dimensions (length and width) were calculated for each sample and archaeological period.
Results
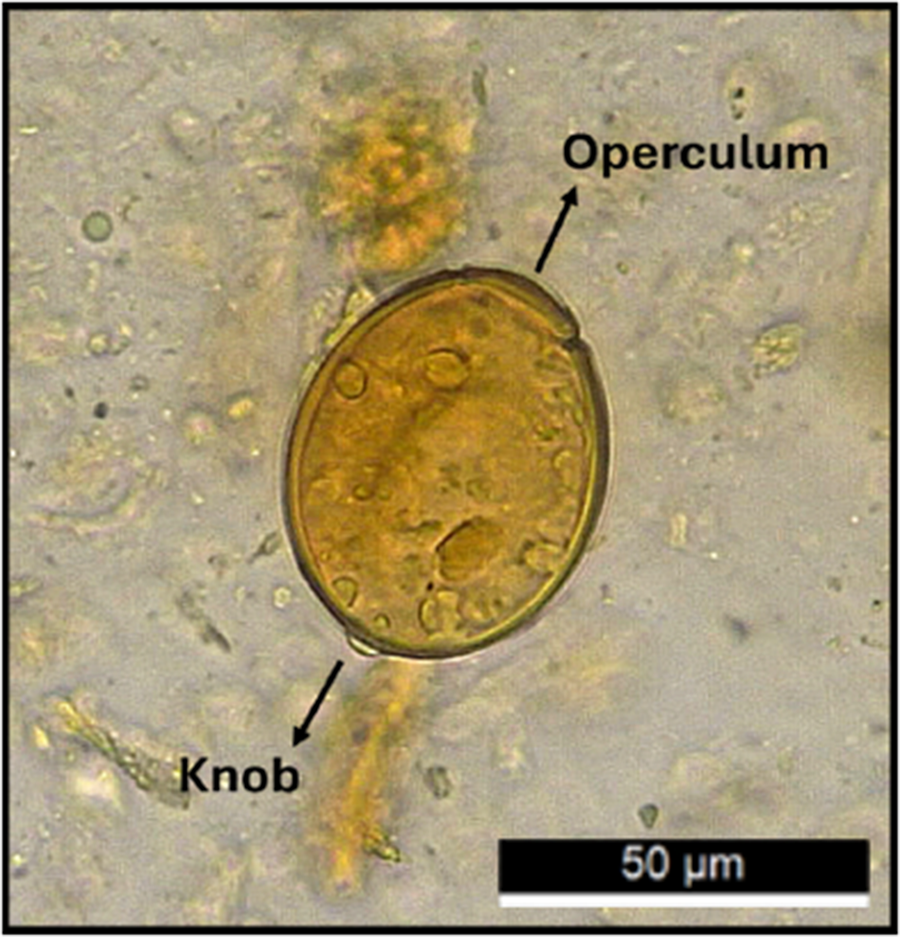
Microscopic examination revealed a high number of well-preserved parasite eggs. Enteroparasitic infections were detected in 12 out of the 22 samples analysed (54.55%), with seven associated with the Late Archaic Period (64%) and five with the Formative Period (45%) (Table 1). Based on morphological and morphometric characteristics, eggs belonging to the fish tapeworm Dibothriocephalus/Adenocephalus sp. were identified (Figure 4).
Light microscope image of Dibothriocephalus/Adenocephalus sp. Egg (dimensions 54.09 μm × 45.35 μm) from CP002 sample, where the operculum, terminal button and eggshell are clearly visible.

The identification of diphyllobothriid eggs was based on key morphological characteristics, including their ovoid shape, thick outer shell, presence of a terminal button and operculum (Baer et al., Reference Baer, Miranda, Fernandez and Medina1967), as shown in Figure 4.
In addition to morphological characteristics, measurements of egg length and width (μm) were carried out to support taxonomic identification. Most diphyllobothriid eggs fell within the expected modern size range of 49–75 μm in length and 36–55 μm in width (Centers for Disease Control and Prevention, n.d.; Hernández-Orts et al., Reference Hernández-Orts, Scholz, Brabec, Kuzmina and Kuchta2015), although smaller eggs were also identified in samples CP105 and CP110. The only egg recovered from sample CP018 did not meet the minimum measurement criteria; therefore, morphometric analysis was not performed for this sample. A large number of incomplete, crushed, and/or broken eggs were also identified in other samples, as can be seen in Figure 5. These are the majority of the eggs identified. Even though the egg’s preservation condition was not optimal, and did not meet the criteria required for length and width measurements (Fugassa, Reference Fugassa2014), taxonomic identification was possible. The Late Archaic eggs were 56.03 µm (±2.85) long and 42.65 µm (±2.38) wide, and the Formative Period eggs were 53.25 µm (±4.63) long and 39.74 µm (±4.53) wide.
Altered Dibothriocephalus/Adenocephalus sp. Eggs from sample CP003. (A) Egg with the operculum opened, (B) broken egg, (C) egg without operculum, (D) crushed egg.

The Late Archaic Period exhibited a higher number of eggs, with a total of 48 023 eggs (98.83%). Egg frequency decreases abruptly in later Periods. During the Early Formative Period, 354 eggs were identified (0.73%), and an even lower frequency was observed in the Late Formative Period, with only 215 eggs identified (0.44%).
To account for differences in sample mass, egg frequency was adjusted for each sample mass by calculating EPG for each sample (Table 1). During the Late Archaic Period, a total of 26 945.40 (≈26 945) EPG (95.74%) was recorded, whereas a total of 1198.62 (≈1199) EPG was attributed to the Formative Period (4.26%). Figure 6 shows the frequency distribution of diphyllobothriids EPG per sample, highlighting the higher frequency recorded in the Late Archaic Period.
Bar graph showing the EPG Frequency of Dibothriocephalus/Adenocephalus sp. Per sample. Samples CP002, CP003, CP004, CP008, CP010, CP013, CP014, CP016, CP018, CP027 and CP097 dates correspond to the Late Archaic Period which are represented in blue. Samples CP005, CP006 and CP001 dates correspond to the Early Formative Period and are represented in red. Samples CP112, CP104, CP105, CP106, CP107, CP108, CP109 and CP110 dates correspond to the Late Formative Period and are represented in green.

All analysed coprolites exhibited excellent preservation. The overall stratigraphic sequence and associated archaeological materials show consistently high levels of preservation across the site. This suggests that taphonomic alterations are likely to have a minimal impact on egg concentrations in the sampled coprolites.
A binomial probability test was conducted based on EPG values to assess differences in egg frequency across archaeological Periods, using a null hypothesis proportion of 50% (0.5) between the Periods. Comparing the Late Archaic and Formative Periods, the Late Archaic showed a significantly higher egg count (p-value < 2.2e−12). The test indicates a statistically significant deviation from the null hypothesis.
These findings demonstrate that the distribution of eggs across the different Periods does not follow a uniform 50% proportion, suggesting significant differences in egg frequencies between the Late Archaic and Formative Periods.
Additionally, a Spearman correlation analysis between coprolite mass and number of eggs from each sample showed no statistically significant relationship (rho = –0.32; P-value = 0.3126), suggesting that coprolite size is not a determining factor in egg frequency.
On the other hand, some samples, specifically CP002, CP004, and CP013 from the Late Archaic Period, contained a high number of eggs, exceeding 2000 in each case. Many of these eggs were very well preserved and suitable for precise morphometric analysis (Figure 7). In some cases, different early embryonic developmental stages can even be observed (Figure 8). In the case of samples CP002 and CP004, measurements were performed on a subset of 200 eggs from each sample.
Twenty Dibothriocephalus/Adenocephalus sp. eggs from sample CP002.

Dibothriocephalus/Adenocephalus sp. eggs from the CP002 sample, showing different stages of early egg development.

Discussion
The present palaeoparasitological study of 22 different coprolite samples revealed the presence of diphyllobothriid eggs in 12 of the analysed human coprolite samples from Aragón-1, spanning the Late Archaic and Formative Periods (Early and Late Formative), in which an exceptional frequency and preservation of parasitic eggs was observed. The extremely arid conditions of the Atacama Desert favour the preservation of parasitic eggs in archaeological sites (Reinhard et al., Reference Reinhard, Arriaza, Avery, Buikstra, Camacho, Goodman, Obafunwa, Owen and Teixeira-Santos2023). Thousands of very well-preserved eggs were observed, in which even different early embryonic developmental stages were recognized. The identification of diphyllobothriid eggs in over half of the coprolites suggests that infections caused by this zoonotic parasite were relatively common in Aragón-1 during these Periods.
Examining the variations of this parasite over time may contribute to the understanding of past human, parasite and environmental dynamics. Below, cultural practices related to food preparation, potential implications for human health, and environmental and climatic changes that may have shaped the presence of diphyllobothriids in Aragón-1 are explored.
Parasite life cycle and fish consumption
The diphyllobothriid tapeworm is one of the largest enteroparasites that infect humans, reaching up to 15 m in length (Centers for Disease Control and Prevention, n.d.). Its life cycle involves two intermediate hosts: copepods (first intermediate host) and fish (second intermediate host). The second intermediate hosts include a wide variety of freshwater and marine fish species. There are diverse animal species that can serve as definitive hosts for the diphyllobothriid cestode. Diphyllobothriasis (infection by diphyllobothriids) is commonly associated with humans, but other species of mammals and birds can contract the disease, including sea lions and dogs, which can be infected by eating raw or undercooked fish (Centers for Disease Control and Prevention, n.d.; Scholz et al., Reference Scholz, Garcia, Kuchta and Wicht2009; Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010).
Plerocercoid larvae are found in the viscera, stomach walls and gonads of fish (Reinhard et al., Reference Reinhard, Arriaza, Avery, Buikstra, Camacho, Goodman, Obafunwa, Owen and Teixeira-Santos2023). Therefore, cleaning and removing these elements drastically reduces the potential for infection (Reinhard et al., Reference Reinhard, Arriaza, Avery, Buikstra, Camacho, Goodman, Obafunwa, Owen and Teixeira-Santos2023). Once the fish is removed from water, the larvae may migrate to, and seek shelter in muscle tissue (Scholz et al., Reference Scholz, Garcia, Kuchta and Wicht2009), increasing the risk of human infection. Cooking fish at a specific and controlled temperature kills the parasitic larvae, preventing infection (Dick et al., Reference Dick, Nelson and Choudhury2001).
The identification of diphyllobothriids, together with the site´s proximity to the coast and the ichthyological identification of intermediate diphyllobothriid hosts (Trachurus murphyi and the family Clupeidae), suggests that these fish species were part of the diet of these human groups. Both taxa represent the most abundant fish remains recorded in the Aragón-1 analysed units (González, unpublished results). In addition, these findings indicate the transport of these fish species from the coast to Aragón-1 by human groups. In this scenario, the Jazpampa ravine likely acted as a natural pathway to the interior, which would be consistent with the existence of a mobility network between the coast and the Pampa del Tamarugal, accompanied by various cultural contacts to access marine resources (Núñez and Zlatar, Reference Núñez and Zlatar1976; Núñez and Hall, Reference Núñez and Hall1982; Grosjean et al., Reference Grosjean, Cartajena, Geyh and Núñez2003).
It has been documented that egg size cannot clearly differentiate between diphyllobothriid species due to overlapping size ranges (Hernández-Orts et al., Reference Hernández-Orts, Scholz, Brabec, Kuzmina and Kuchta2015). However, it is still important to evaluate the smaller eggs found on samples CP105 and CP110. There are multiple factors that could result in smaller eggs. Eggs from Adenocephalus pacificus have been reported to be generally smaller, which could indicate that smaller eggs may be attributed to this specific species. However, they also show a large size variability (49–60 μm in length and 36–45 μm in width) (Hernández-Orts et al., Reference Hernández-Orts, Scholz, Brabec, Kuzmina and Kuchta2015). On the other hand, it has been reported that younger cestode worms produce smaller eggs than senescent individuals (Magath, Reference Magath1929). However, this relationship remains debated, as mean egg size has been shown to vary independently of worm age, and more recent data do not reveal a clear age-dependent pattern in egg size (Andersen and Halvorsen, Reference Andersen and Halvorsen1978). Cestode egg size remains understudied, and the considerable variability in egg sizes constrains interpretations of these variations.
Prevalence of Dibothriocephalus/Adenocephalus sp. and cultural factors
Fish tapeworm infections provide important insights into cultural variations and lifestyle changes in ancient populations (Le Bailly and Bouchet, Reference Le Bailly and Bouchet2013). Diphyllobothriids’ findings suggest the consumption of raw or undercooked fish infected with tapeworm larvae. Exposure to a specific temperature (55°C) achieved by fire during a minimum of 5 min effectively kills parasitic larvae (Dick et al., Reference Dick, Nelson and Choudhury2001). Therefore, the introduction of more efficient cooking practices in later periods could have contributed to the decrease in the frequency of diphyllobothriids. This may suggest a culinary shift (Correa et al., Reference Correa, Carrasco, Ballester and Gallardo2018; Reinhard et al., Reference Reinhard, Arriaza, Avery, Buikstra, Camacho, Goodman, Obafunwa, Owen and Teixeira-Santos2023) from consuming raw or poorly cooked fish to cooking methods that can reach the necessary temperatures and exposure times required to prevent parasitic infections.
Among the coprolites in which parasites were identified, a wide variation in egg frequencies was observed. Some samples contained a notably high number of diphyllobothriid eggs, and in others only 1 or 2 eggs were identified. The coprolites containing high egg frequency reflect a greater parasite load, which could indicate a more severe infection. This suggests a potentially increased transmission risk within the population, as a greater number of eggs increases the likelihood of parasite re-entry into the reproductive life cycle (Mackiewicz, Reference Mackiewicz1988). This finding, combined with the archaeological context of a shift in subsistence and lifestyle, highlights the significant relevance of assessing this marked decrease in egg frequency between periods. On the other hand, coprolite samples with low egg frequency, such as samples CP018, CP005 and CP105, may be the result of false parasitism, where eggs may have been ingested fortuitously from another infected host (mammal or bird) and, without causing infection, are subsequently passed in human feces (Reinhard, Reference Reinhard2017).
The transition from the Archaic to the Formative Periods is marked by substantial changes in human lifestyles, and Aragón-1 material evidence could be the key to understanding the decline of this parasite through time. The presence of ceramics with traces of fire exposure from the Formative Period at Aragón-1 could indicate expertise in ceramic production and the existence of local specialists engaged in pottery making. This technical knowledge appears to be the result of a long-term process of experimentation that likely began during the late Archaic Period (Uribe, Reference Uribe, Zarankin and Acuto2008; Núñez and Santoro, Reference Núñez and Santoro2011; Santana-Sagredo et al., Reference Santana-Sagredo, Uribe, Herrera, Retamal and Flores2015; Uribe et al., Reference Uribe, Angelo, Capriles, Castro, De Porras, García, Gayo, González, Herrera, Izaurieta and Maldonado2020). It may also indicate changes in fire management practices, which could have led to shifts in dietary habits.
There are also other culinary practices that do not require ceramics for cooking. Near the Aragón-1 site, at Tiliviche-1B, there is evidence of fire-altered fish bones and hearths without ceramic presence (Núñez and Hall, Reference Núñez and Hall1982). In the Andes region, there are cooking strategies like the use of hot stones, pits and earth ovens that have been employed to expose food to fire since pre-Hispanic times and some of them continue to be used in the present (Choy, Reference Choy1979; Castillo-Posadas, Reference Castillo-Posadas2014). However, the incorporation of ceramic technology for cooking could have been the method that allowed for more controlled, sustained and homogeneous heating, which is required to destroy parasitic larvae and other pathogens.
The presence of ichthyofauna that persists throughout the occupation of Aragón-1 (González, unpublished results), together with human palaeodiet reconstructions based on stable isotope analysis from surrounding coast and inland sites (Santana et al., Reference Santana, Herrera and Uribe2012; Santana-Sagredo et al., Reference Santana-Sagredo, Lee-Thorp, Schulting and Uribe2019), supports continued reliance on marine resources in the Tarapacá region through time, as it has been consistently documented (Núñez and Zlatar, Reference Núñez and Zlatar1976, Reference Núñez and Zlatar1977; Núñez, Reference Núñez1979; Núñez and Hall, Reference Núñez and Hall1982; Muñoz Ovalle, Reference Muñoz Ovalle2004; Standen et al., Reference Standen, Santoro and Arriaza2004; Núñez and Santoro, Reference Núñez and Santoro2011; Uribe et al., Reference Uribe, Angelo, Capriles, Castro, De Porras, García, Gayo, González, Herrera, Izaurieta and Maldonado2020). Therefore, an alternative explanation for the reduction in parasite frequency could be the ingestion of dried fish or charki (a word originating from Quechua/Aymara and currently used in Chile and other Andean countries), a preparation and preservation method that can reduce the risk of parasitic larval infection, although it does not guarantee complete elimination (Ortiz et al., Reference Ortiz, Uys, Seguino and Thomas2024). Archaeological evidence from sites across the Atacama Desert indicates dried fish as a dietary staple during the Formative Period (Castillo et al., Reference Castillo, Ballester, Calás, Labarca, Gallardo, Gallardo, Ballester and Fuenzalida2017; Ballester et al., Reference Ballester, Calás, Labarca, Pestle, Gallardo, Castillo, Pimentel and Oyarzo2019).
It is essential to recognize that changes in eating habits and cooking methods can sometimes be culinary choices that transcend subsistence needs and may also reflect cultural preferences (Sy, Reference Sy2009; Correa et al., Reference Correa, Carrasco, Ballester and Gallardo2018). This concept is particularly relevant when interpreting potential shifts from raw or poorly cooked fish to cooking practices capable of preventing parasitic infection. Decisions about food preparation do not necessarily change in response to health risks, as dietary practices also carry significant symbolic, social, and traditional meanings. Raw fish consumption persists in many societies despite the known risk of contracting parasitic diseases such as those caused by diphyllobothriids (Darwin and Fried, Reference Darwin and Fried2007). For example, ceviche is a traditional dish in Andean countries, consisting of raw fish. Instead of modifying culinary practices, human groups may address the consequences through the use of medicinal plants or other post-infection treatments. This cultural perspective helps to contextualize why the adoption of ceramic technology, which enables more controlled and sustained heating, may represent not only a technological change but also a significant transformation in daily practices with implications for parasite transmission.
Absence of other parasites in human coprolites from Aragón-1
The absence of other parasites is possibly due to both environmental and cultural factors. Palaeoparasitological analysis in the Tarapacá region indicates that diphyllobothriids’ infection was mainly found among coastal people and groups inhabiting areas near the seaside. Diphyllobothriids have been identified in sites such as Tiliviche-1B, dating to the Archaic Period (Ferreira et al., Reference Ferreira, Araújo, Confalonieri and Nuñez1984; Araújo et al., Reference Araújo, Ferreira, Confalonieri, Nuñez and Ribeiro Filho1985), as well as Pisagua 7 and Caleta Huelen 43, from the Formative Period (Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021). On the other hand, the parasites E. vermicularis and T. trichiura were found mainly in inland sites and associated with later periods such as the Tarapacá-40, Caserones (Formative Period) and Pica 8 (Late Intermediate Period, 900–1450 CE) archaeological sites (Araújo et al., Reference Araújo, Ferreira, Confalonieri, Nuñez and Ribeiro Filho1985; Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021). These parasites are linked to sedentary lifestyles, village way of life, higher population density, and overcrowded living areas (Castells et al., Reference Castells, González and Westfall2010; Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021). However, E. vermicularis was also found at the Archaic site Tiliviche-1B, closer to the coast (Araújo et al., Reference Araújo, Ferreira, Confalonieri, Nuñez and Ribeiro Filho1985).
The presence of diphyllobothriids could affect the possibility of concomitant endoparasitic infections. Dibothriocephalus/Adenocephalus sp., being one of the largest parasites that infect humans, would compete efficiently for nutrients, not only with the definitive host (Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010), but also with other potential intestinal parasites. Competition for nutrients greatly hinders the development of other parasites. This can be evidenced even in the case of diphyllobothriasis, in which, in most cases, only a single tapeworm can inhabit the host, as there are not enough nutrients for another to develop (Jiménez et al., Reference Jimenez, Rodriguez, Gamboa, Rodriguez and Garcia2012).
Health implications and lifeways
Parasitic diphyllobothriid infection can cause several effects on human health. In general, the symptoms are mild, and the vast majority of hosts remain asymptomatic (Semenas, Reference Semenas2013). However, other factors, such as age, overall health and nutritional status, influence the severity of symptoms (Neghme and Silva, Reference Neghme and Silva1971; Sy, Reference Sy2009). Mild symptoms include fatigue, abdominal pain, constipation, vomiting, diarrhoea, weight loss, headache and loss of appetite (Scholz et al., Reference Scholz, Garcia, Kuchta and Wicht2009; Jimenez et al., Reference Jimenez, Rodriguez, Gamboa, Rodriguez and Garcia2012). The presence of proglottids (adult worm segments) in the feces can be used for the diagnosis of diphyllobothriasis today (Jimenez et al., Reference Jimenez, Rodriguez, Gamboa, Rodriguez and Garcia2012) and may have also been used for its diagnosis in the past.
In addition, prolonged or heavy infection may cause symptoms related to the circulatory system as megaloblastic anaemia due to vitamin B12 deficiency (Torres, Reference Torres1998; Scholz et al., Reference Scholz, Garcia, Kuchta and Wicht2009; Jimenez et al., Reference Jimenez, Rodriguez, Gamboa, Rodriguez and Garcia2012). Diphyllobothriids’ absorption of nutrients can decrease vitamin B12 levels (Jimenez et al., Reference Jimenez, Rodriguez, Gamboa, Rodriguez and Garcia2012). Modern clinical studies indicate that approximately 40% of infected individuals exhibit reduced B12 levels, and clinical anaemia is rare, affecting only up to 2% of cases (Scholz et al., Reference Scholz, Garcia, Kuchta and Wicht2009).
Cases of severe anaemia can lead to porotic hyperostosis and cribra orbitalia (porosity on the cranial bones and the orbital roof), which can be identified through the analysis of skulls in the archaeological records. According to the literature, in some cases, these alterations could be related to parasitic infections (Schultz, Reference Schultz2001; Blom et al., Reference Blom, Buikstra, Keng, Tomczak, Shoreman and Stevens-tuttle2005; Walker et al., Reference Walker, Bathurst, Richman, Gjerdrum and Andrushko2009; Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010). Even though severe anaemia is a rare symptom of infection by this specific parasite, it remains relevant to discuss further.
Palaeopathological studies have identified porotic hyperostosis and cribra orbitalia in many archaeological sites spanning various time Periods through the Tarapacá region (Cocilovo et al., Reference Cocilovo, Varela, Valdano and Silvia Quevedo1999, Reference Cocilovo, Varela, Costa-Junqueira and Quevedo2005; Retamal et al., Reference Retamal, Pacheco and Uribe2006; Uribe et al., Reference Uribe, Agüero, Catalán, Herrera and Santana-Sagredo2015; González-Ramírez et al., Reference González-Ramírez, Herrera-Soto and Menanteau2017). Additionally, evidence of parasitic infections has also been reported in or near these sites (Ramírez et al., Reference Ramírez, Herrera-Soto, Santana-Sagredo, Uribe-Rodríguez and Nores2021), suggesting a potential link between parasitic infection and the development of these osteological pathologies. Although it is essential to acknowledge the limitations of this interpretation, porotic hyperostosis and cribra orbitalia are non-specific stress markers, meaning they can result from a wide range of causes and cannot be attributed exclusively to a single specific aetiology. However, high parasite loads during childhood have been considered a more likely cause in past Andean coastal societies (Blom et al., Reference Blom, Buikstra, Keng, Tomczak, Shoreman and Stevens-tuttle2005; Klaus, Reference Klaus2020). In the case of diphyllobothriid infections, individuals with normal B12 intake may not develop symptoms. Nevertheless, when parasitic infection is combined with malabsorption, chronic diarrhoea and low dietary intake of vitamin B12, severe anaemia may occur (Walker et al., Reference Walker, Bathurst, Richman, Gjerdrum and Andrushko2009). Finally, due to the absence of human skeletal remains at Aragón-1, it has not been possible to assess these conditions osteologically in individuals from the site or to establish any link between these pathologies and parasitism.
More severe cases of diphyllobothriasis can cause intestinal obstructions (Jimenez et al., Reference Jimenez, Rodriguez, Gamboa, Rodriguez and Garcia2012; Scholz et al., Reference Scholz, Garcia, Kuchta and Wicht2009). Although it is now easy to diagnose and eliminate parasitic infections with the administration of drugs (Semenas, Reference Semenas2013), in the past, the development of more serious symptoms, such as intestinal obstruction, could have led to the death of the infected person.
Environmental and ecological influences
Environmental aspects that affect the presence of diphyllobothriids must also be taken into consideration. El Niño-Southern Oscillation (ENSO), where El Niño (warm events) and La Niña (cold events) generate variations in the presence of parasitic infections over time (Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010).
Warmer waters and a climate with higher humidity during the El Niño phenomenon favour parasitic life cycle and proliferation (Sagua et al., Reference Sagua, Neira, Araya and González2001; Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010; Reinhard et al., Reference Reinhard, Arriaza, Avery, Buikstra, Camacho, Goodman, Obafunwa, Owen and Teixeira-Santos2023). These climatic conditions also impact Pacific Ocean biomass (Sagua et al., Reference Sagua, Aliaga, Neira, Araya and González2000, Reference Sagua, Neira, Araya and González2001; Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010). The populations of crustaceans (first intermediate hosts of diphyllobothriids), fish (second intermediate hosts), and various species of mammals and birds (definitive hosts) (Sagua et al., Reference Sagua, Neira, Araya and González2001; Reinhard et al., Reference Reinhard, Arriaza, Avery, Buikstra, Camacho, Goodman, Obafunwa, Owen and Teixeira-Santos2023) increase due to migrations towards the coast of northern Chile, potentially contributing to increased parasitic infections caused by fish tapeworm zoonosis (Sagua et al., Reference Sagua, Neira, Araya and González2001; Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010). ENSO-related variability may have influenced parasitic variations in the archaeological record, as it continues to affect parasite proliferation in the present (Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010).
El Niño and La Niña temperature variations of ocean water phenomena last up to 7 years (Sandweiss et al., Reference Sandweiss, Maasch and Anderson1999; McPhaden et al., Reference McPhaden, Zebiak and Glantz2006). Palaeoclimatic data confirm this type of water temperature variation existed in the past (Moy et al., Reference Moy, Seltzer, Rodbell and Anderson2002; Rein et al., Reference Rein, Lückge and Sirocko2004; McPhaden et al., Reference McPhaden, Zebiak and Glantz2006; Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010). Palaeoclimatological reconstructions based on stable oxygen isotope (δ1⁸O) ratios from marine shells can be reliable proxies for water temperature and in identifying ENSO activity (Carré et al., Reference Carré, Bentaleb, Blamart, Ogle, Cardenas, Zevallos, Kalin, Ortlieb and Fontugne2005a, Reference Carré, Bentaleb, Fontugne and Lavallee2005b; Carré et al., Reference Carré, Sachs, Schauer, Rodríguez and Ramos2013). Palaeoceanographic studies from southern Peru employing this technique have produced high-resolution reconstructions that reflect the variability of these phenomena (Carré et al., Reference Carré, Bentaleb, Blamart, Ogle, Cardenas, Zevallos, Kalin, Ortlieb and Fontugne2005a, Reference Carré, Bentaleb, Fontugne and Lavallee2005b; Carré, et al., Reference Carré, Sachs, Schauer, Rodríguez and Ramos2013). Notably, shell remains were recovered from all excavation units at Aragón-1 and will be analysed to assess these ocean variations.
The prevalence of diphyllobothriid infection likely depended on whether El Niño or La Niña episodes were occurring, as these climatic events would have constrained or enhanced the reproductive cycles of both parasites and their hosts (Arriaza et al., Reference Arriaza, Reinhard, Araújo, Orellana and Standen2010). Therefore, climatic effects cannot be ruled out as possible causes of the absence of parasitic structures and low frequency of diphyllobothriid found in Aragón-1 in later periods.
Conclusions
The palaeoparasitological analysis of Aragón-1 coprolites revealed the presence of eggs of the parasitic fish tapeworm Dibothricephalus/Adenocephalus sp. during the Late Archaic and Formative Periods, indicating a close ecological interaction between this parasite and the humans who inhabited the site.
The frequency of diphyllobothriids decreases significantly towards the Formative Period, a phenomenon that could be explained by cultural and/or environmental factors affecting this human-parasite relationship. Among the cultural changes, modifying culinary practices, such as cooking the fish, would eliminate diphyllobothriid larvae from the fish and consequently reduce the presence of the parasite. This would imply not only a dietary shift but also transformations in cultural knowledge systems, culinary practices and traditions. New cooking techniques may reflect a growing awareness of the role of food preparation in mitigating health risks. Additionally, environmental factors, such as the El Niño and La Niña phenomena, can cause variations in the occurrence of parasitic infections.
This type of analysis provides valuable insights into the cultural practices and ways of life of Aragón-1 inhabitants. The findings also reveal dietary habits based on marine resources, as well as mobility between the coast and the site for the procurement of these products, despite the distance, as it has been consistently documented for the Archaic and Formative Periods in the Tarapacá region.
High frequency and exceptional preservation of diphyllobothriid eggs are particularly noteworthy. The environmental conditions of the Atacama Desert contribute to the remarkable preservation of this archaeological record.
Further studies are needed to gain a better understanding of the relationship between parasites and the human groups that inhabited Aragón-1 during the Late Archaic and Formative Periods. Future molecular analyses would complement our investigation and strengthen Diphyllobothriid identification. In addition, lipid residue analysis in ceramics to identify whether they were used for cooking fish (Lucquin et al., Reference Lucquin, Gibbs, Uchiyama, Saul, Ajimoto, Eley, Radini, Heron, Shoda, Nishida, Lundy, Jordan, Isaksson and Craig2016), and oxygen isotope analysis in shells to reconstruct marine temperatures (Carré et al., Reference Carré, Bentaleb, Blamart, Ogle, Cardenas, Zevallos, Kalin, Ortlieb and Fontugne2005a, Reference Carré, Bentaleb, Fontugne and Lavallee2005b; Carré et al., Reference Carré, Sachs, Schauer, Rodríguez and Ramos2013), would greatly contribute to our discussion and interpretations. Finally, the analysis of a larger number of samples, including those from animals, from this site and various archaeological sites in Northern Chile, will contribute to a more comprehensive understanding of human–parasite interactions, as well as the cultural practices and their relationship with the environment.
Acknowledgements
We thank all the members of the FONDECYT 1221166 team, without whom this research would not have been possible. Special thanks to Alicia Nogueras, Claudio Wande, Mauricio Uribe Rodríguez, Josefina González, Marco Minniti, Bruno Minniti, Rebeca Aldunate, Valentina Espinosa, Javiera Navarro, Luis Nacra, Alondra Staforelli, Rodrigo Retamal, Ariel Sperling, Pablo Méndez-Quiroz and Ale Vidal-Elgueta.
Author contributions
SM-N: Conceptualization, writing original draft, analysis, writing/review and editing. FS-S: Funding acquisition, writing/review and editing. DR: Writing-review and editing. MH-S: Writing/review and editing.
Financial support
This work was supported by grant FONDECYT 1221166 (Fondo Nacional de Desarrollo Científico y Tecnológico).
Competing interest
The authors declare there are no conflicts of interest.
Ethical standards
Not applicable.