♂♀1.1 Introduction
The urogenital system consists of the urinary system, which purifies the blood and eliminates catabolites through the urine, and the genital system, which enables production of gametes, fertilization, and the generation and development of a new individual. The urinary and genital systems are closely associated, embryologically, anatomically, and functionally.
The urogenital system develops from the intermediate mesoderm, a long cord of cells that originate during gastrulation in the 3rd week, along the dorsal body wall of the embryo. In the 4th week, as a consequence of folding, it is localized ventrally in the embryo, and develops into the renal structures, and parts of the gonads and male genital tract. The urogenital ridge is a longitudinal elevation of the intermediate mesoderm that gives rise to both the nephrogenic cord, from which the urinary system develops, and the genital ridge, from which parts of the genital system (gonads) develop.Reference Avni, Maugey-Laulom, Cassart and Callen1, Reference De Felici and Canipari.2
The genital system is complex, bringing together structures with different embryonic origin. The gonads develop from the genital ridges, the genital tract from the mesonephros, the external genitalia from the region around the cloacal membrane, and the primordial germ cells (future gametes) come from the endoderm of the yolk sac.
The genital system is the only system characterized by sexual dimorphism, so the earliest stages of development are common to both sexes, but proceed separately starting from the 6th–7th weeks in males, and from the 10th week in females.
In the following chapters, the development of the urinary system and sexual differentiation are discussed in detail, emphasizing the fundamental preconditions of male or female differentiation.
♂♀1.2 Embyronic Development
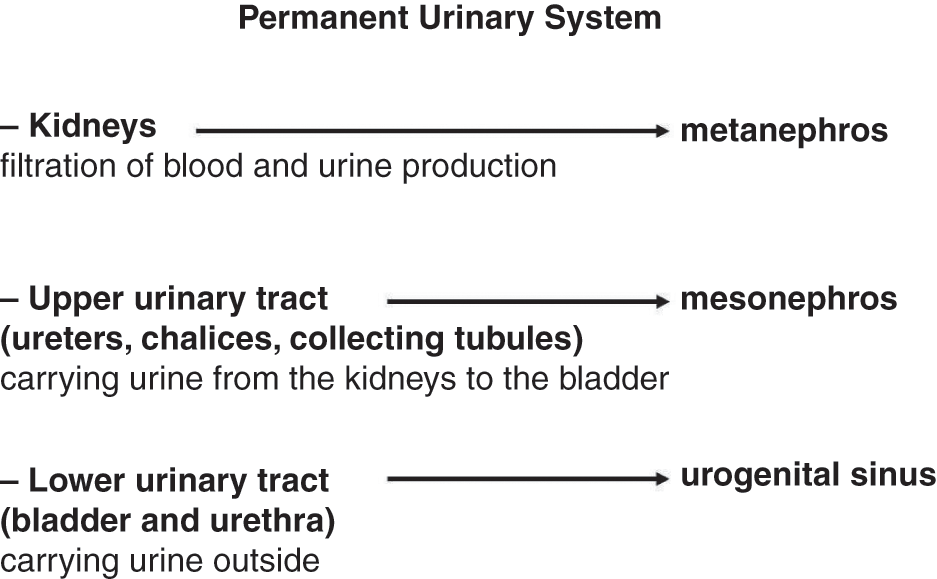
The urinary system (Figure 1.1)consists of:
1. kidneys, responsible for filtration of blood and urine production;
2. upper urinary tract (ureters, chalices, and collecting tubules) that carry urine from the kidneys to the bladder;
3. lower urinary tract (bladder, which collects the urine, and urethra, which carries urine to the outside, allowing its elimination).
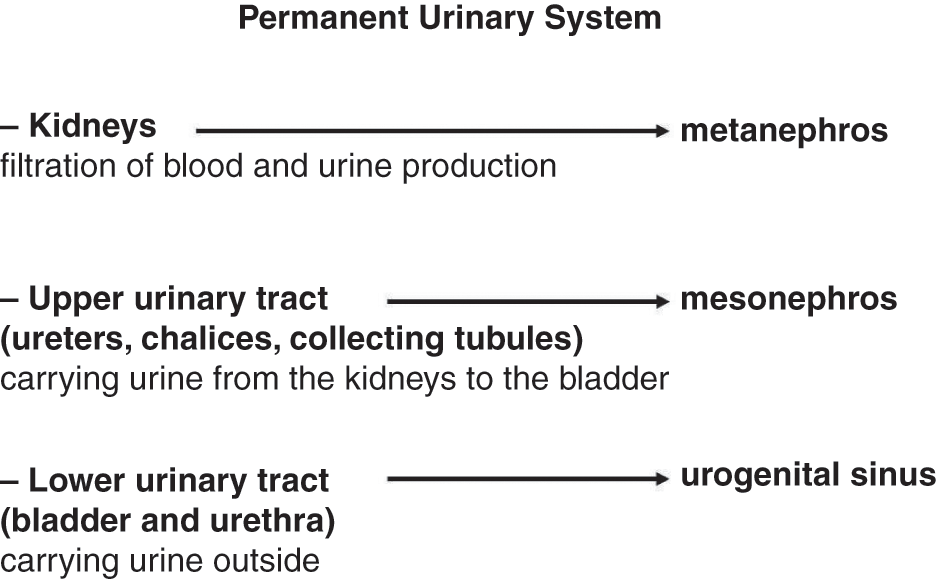
♂♀Figure 1.1 Permanent urinary tract: components, roles, and embryological origin.
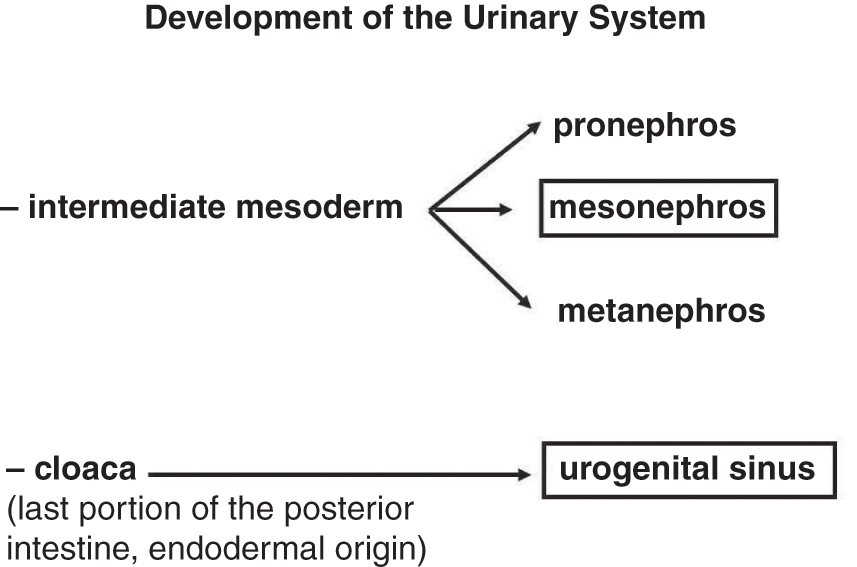
It originates from the nephrogenic cords, two cell cords that develop from the intermediate mesoderm immediately after gastrulation. From the nephrogenic cords, three sets of renal structures arise (Figure 1.2): pronephros (rudimentary), mesonephros (working for a short period), and metanephros (permanent kidney).
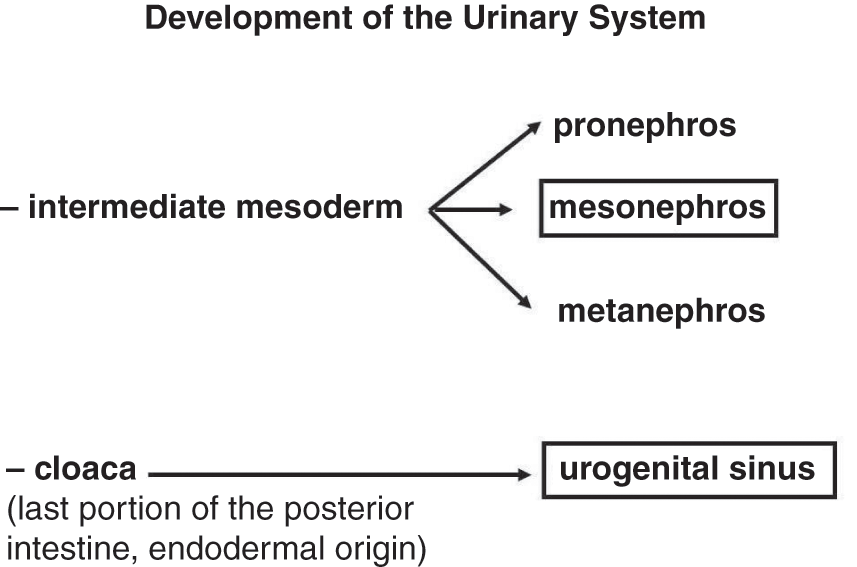
♂♀Figure 1.2 Embryological origin of the urinary system. Boxes indicate the portions also involved in genital system development.
The pronephros develops first, in the 4th week. In the two nephrogenic cords, the cranial region is fragmented into 5–7 cell clusters associated with the pronephric duct, which is a longitudinal duct growing caudally to the cloaca (last portion of the posterior intestine, endodermal origin).Reference Cuckiw, Nyirady and Winyard3
At the end of the 4th week, the pronephros degenerates and the pronephric ducts give rise to the mesonephric (or Wolffian) ducts, which subsequently open into the cloaca. Below the pronephroi and adjacent to the Wolffian ducts, each nephrogenic cord is induced by the pronephroi to develop mesonephric tubules whose distal ends open into the Wolffian ducts, while the opposite ends surround a ball of capillaries arising from the dorsal aorta. The mesonephric tubules and the Wolffian ducts constitute the mesonephros, which has an inductive role in the subsequent development of the definitive kidney and the internal male genital tract. In the male embryo the more cranial mesonephric tubules stop renal function and become efferent tubules and the Wolffian ducts give rise to the epididymis, the vas deferens, the ejaculatory duct, and the seminal vesicles.Reference Short and Smyth4
During the 5th week, the metanephroi (permanent kidneys) begin their differentiation. In the most caudal portion of each Wolffian duct, the ureteric bud develops and penetrates the metanephric mass, mesenchyme of the sacral region of nephrogenic cords, and at this point begins a sequence of bifurcations and coalescences. Within the metanephric mass, the apex of the ureteric bud expands and gives rise to the renal pelvis that later branches, forming the collecting tubules.
The first generations of the collecting tubules merge and give rise to the major calix; their ends branch and merge to form the minor calix that further branches to form numerous collecting tubules (collecting system). The ends of the collecting tubules are surrounded by the metanephric mass cells and are induced to develop the different portions of the nephrons (distal convoluted tubule, loop of Henle, proximal convoluted tubule, Bowman’s capsule), which form the excretory system of the kidneys. The normal development of the permanent kidney depends on reciprocal interactions between the ureteric bud (mesonephric origin) and the metanephric mass.
The ureters develop by lengthening of the ureteric bud. Initially the ureters open into the Wolffian ducts, then open directly into the bladder, losing contact with the Wolffian ducts.
The developing kidneys are originally located in the sacral region, then move towards the posterior wall of the abdominal cavity; consequently, the ureters become longer. At the end of the 2nd month, the kidneys are in their definitive seat (at the level of the first four lumbar vertebrae and below the adrenal glands).
The bladder originates from the upper portion of the urogenital sinus, which is the front portion of the cloaca divided by the urorectal septum into two portions, the urogenital sinus and anorectal canal. The urogenital sinus give rise to the bladder, whose differentiation occurs in similar ways in both sexes, and the urethra and the definitive urogenital sinus, which have different fates in the two sexes. The urethra forms much of the male urethra and the entire female urethra, the definitive urogenital sinus gives rise to the penile urethra in the male and the vestibule of the vagina in the female. As a result of the ascent of the kidneys and then the displacement of the orifices of the ureters more cranially than the Wolffian ducts, the ureters open directly into the bladder, and the Wolffian ducts open more caudally and directly into the urethra. These movements result in incorporation of metanephric tissue (mesoderm) into the endodermic posterior wall of the bladder, defining the bladder trigone. The further development of the urethra and the vaginal vestibule are described in the chapters related to the development of the external genitalia.
♂♀1.3 Indifferent Embryonic Development of the Genital System (Pre-sex Determination)
The genital system consists of the gonads, the genital tract, the external genitalia, and the germ cells. The genital drafts appear in different parts of the embryo at about 4 weeks of development, and then they converge into a unique system that differentiates from the 4th to the 7th weeks. Throughout this period, called the indifferent stage, the genital system is identical in males and females. Only at the end of the 2nd month, do the first signs of differentiation begin to appear and the indifferent genital drafts acquire male or female characteristics. The development of the indifferent genital system is closely related to the urinary system.
At the end of the 3rd week, in a region located between the allantois and the yolk sac, it is possible to recognize a small group of cells, the primordial germ cells (PGCs).Reference De Felici5 The PGCs are the cell line that allows the process of gametogenesis. They proliferate and move in the developing gonads (genital ridges) where they continue to proliferate and then differentiate into oogonia or spermatogonia, depending on the sex of the embryo.
During the 5th week, in splanchopleura (lateral mesoderm) localized ventrally and close to the mesonephros, two longitudinal thickenings (genital ridges), which are the gonadal drafts, arise. Genital ridges are characterized by coelomic epithelium (cortical) that originates from the proliferation of splanchopleura, and mesenchyme (medullary) arising from the intermediate mesoderm underlying the splanchopleura. When the PGCs reach the genital ridges, they are surrounded by coelomic epithelium cells, forming the gonadal cords. If the PGCs are not coated by the gonadal cords, they undergo degeneration.
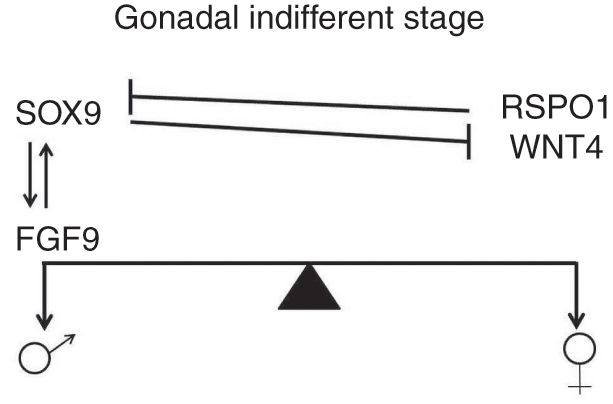
This stage of gonadal development is termed the indifferent gonad (Figure 1.3) until the end of the 6th week, as the male and female gonads are morphologically identical. In the mouse, several essential genes for the development and differentiation of male and female genital systems have been identified, including Sf1 (steroidogenic factor 1) and Wt1 (Wilms tumor 1).Reference Wilhelm and Englert6

♂♀Figure 1.3 Molecular model of the indifferent gonad.
The differentiation of the indifferent gonad in the testicle temporally precedes its alternative differentiation in the ovary.
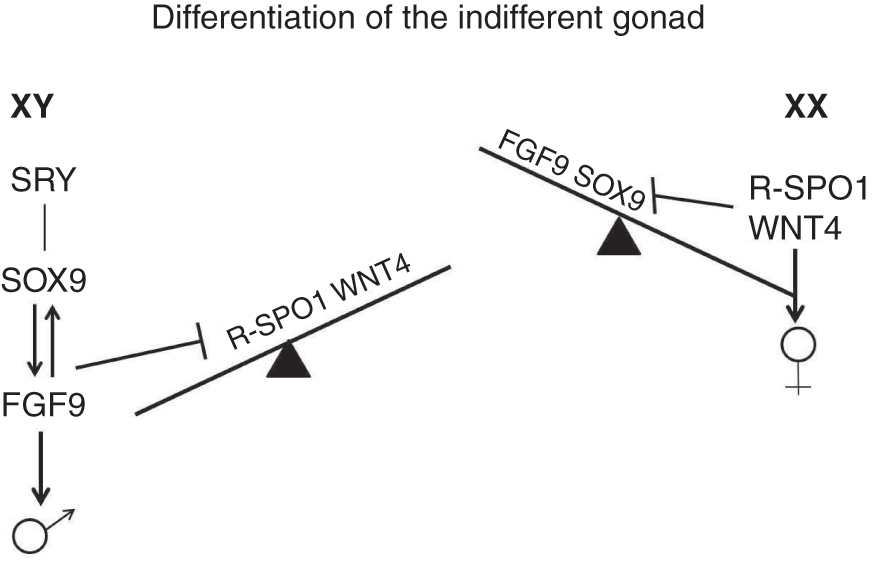
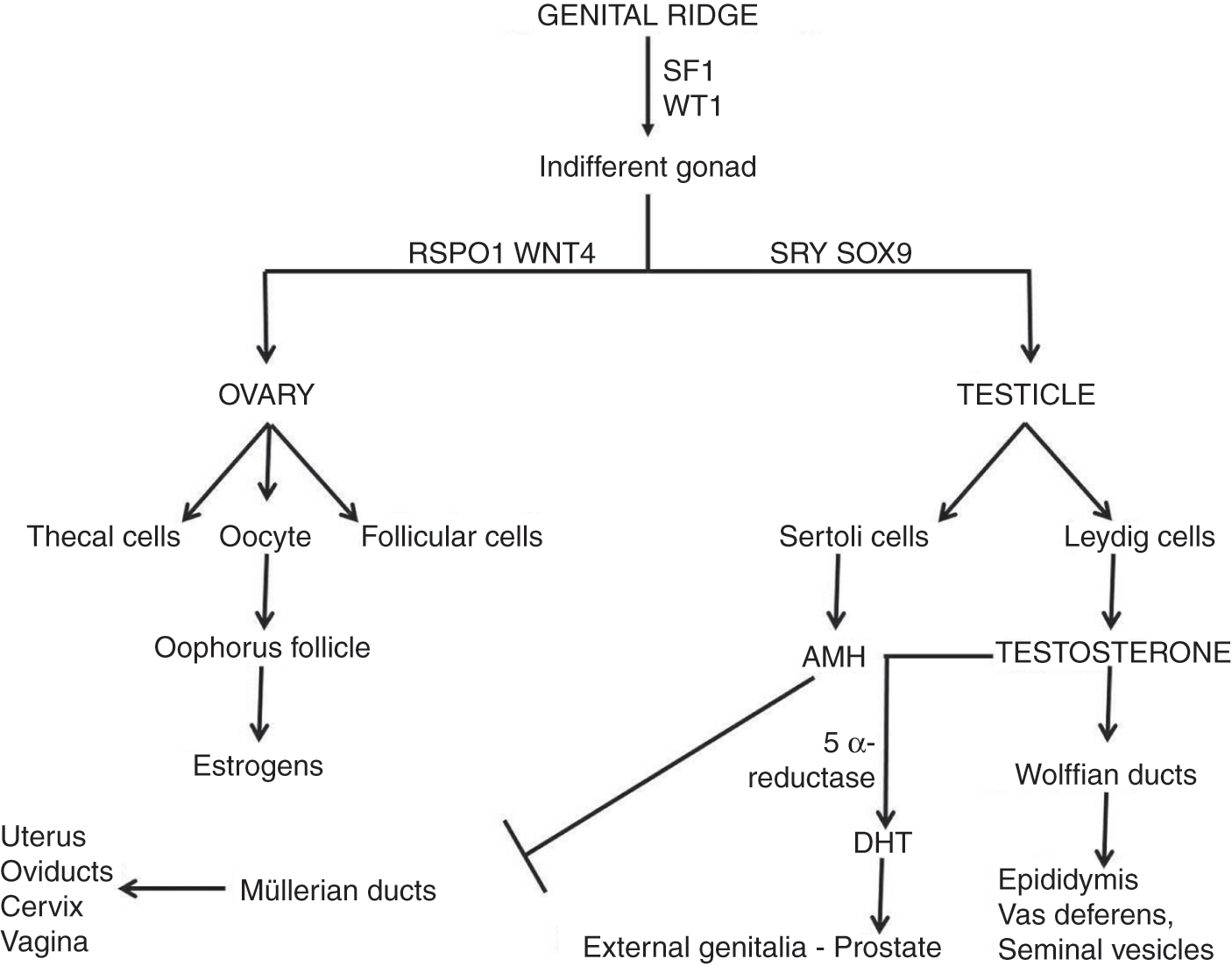
♂It has been well demonstrated that the male gonad differentiation takes place as a result of Sry (sex determining region Y) gene activation producing a testicular-determining factor (TDF), localized in the short arm of the Y chromosome in somatic cells of the indifferent gonad that differentiate into Sertoli cells.Reference Merchant-Larios and Moreno-Mendoza7–Reference Larney, Bailey and Koopman10 The expression of Sry, in synergy with Sf1, is followed by that of the Sox9 gene, which together with the paracrine action of FGF9 (fibroblast growth factor-9) stabilizes the masculinizing environment, allowing development of the Leydig cells and male germ cells (Figure 1.4).Reference Chung, Pask and Yu11, Reference Kanai, Hiramatsu and Matoba12
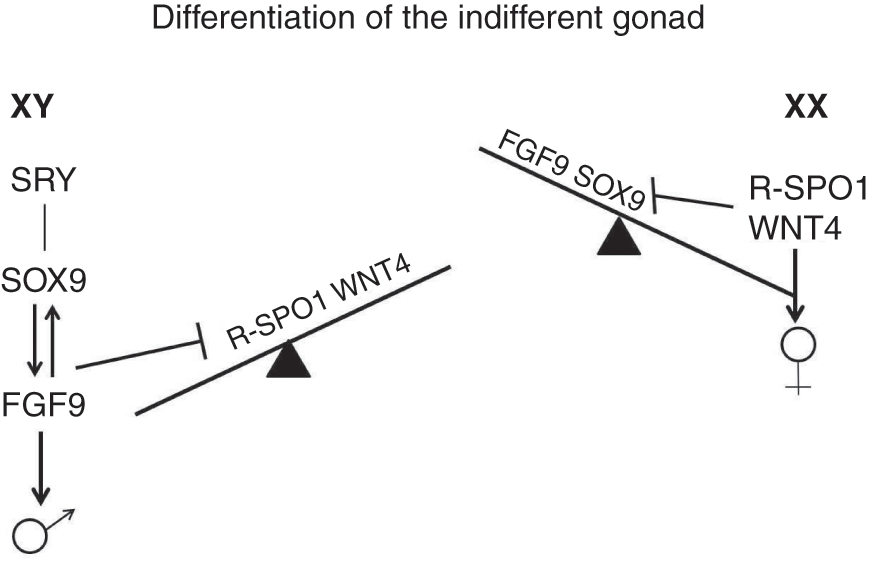
♂♀Figure 1.4 Molecular model of gonadal sexual differentiation, in mammals. The differentiation into male or female gonad is based on the level of expression and/or repression of “masculine” or “feminine” factors.
♀The differentiation of the female gonad seems not to be determined by a single gene, but is under the control of intra- and extracellular signals. Among the latter, the feminizing growth factors WNT4 (wingless-related and integrated 4) and RSPO1 (R-spondin 1) produced by the ovary cells regulate the development of the female gonad (Figure 1.4). Reference Jeays-Ward, Hoyle and Brennan13–Reference Chassot, Gregoire and Magliano15 A model for gonadal sex determination has been proposed in which FGF9 and WNT4/RSPO1 are the extracellular growth factors that maintain the indifferent gonad in balance between the two fates: the prevalence of FGF9 causes testicular differentiation, the prevalence of WNT4/RSPO1 causes ovarian (Figure 1.4).Reference Kim, Kobayashi and Sekido16, Reference Chassot, Gillot and Chaboissier17
The development of the genital tract also begins in the same way in both sexes.
♂In the 4th week, the mesonephros (transitional primitive kidney) develops a cord of cells, which subsequently canalizes: the mesonephric (Wolffian) duct. Each duct develops in the caudal direction and opens into the cloaca.
♀In the 6th week, on the outside of the Wolffian duct, a fold of the splanchnic mesoderm is formed, giving rise to the paramesonephric (Müllerian) duct.
Starting from the 7th week, the development of the Wolffian and/or Müllerian ducts takes different directions depending on the sex of the embryo. The initial development of the genital tract is independent of hormones; later it is controlled by hormones and growth factors.
As well as the gonads and genital tract, during the first period of development (from the 5th to the 8th weeks) the morphology of the external genitalia is similar in embryos of both sexes. At the end of the 5th week, at the sides of the cloacal membrane there are two cloacal folds that merge giving rise to the genital tubercle. During the 7th week, after the formation of the perineum that separates cloacal membrane into urogenital membrane and anal membrane, the cloacal folds are divided into urethral folds and anal folds. At the sides of the urethral folds, two genital swellings are formed. Simultaneously, the urogenital membrane is perforated, opening the cavity of the urogenital sinus to the outside. Male or female differentiation of the different parts of the external genitalia is visible after the 12th week and is a hormone-dependent process.Reference Schlomer, Feretti and Rodriguez18
♂1.4 Male
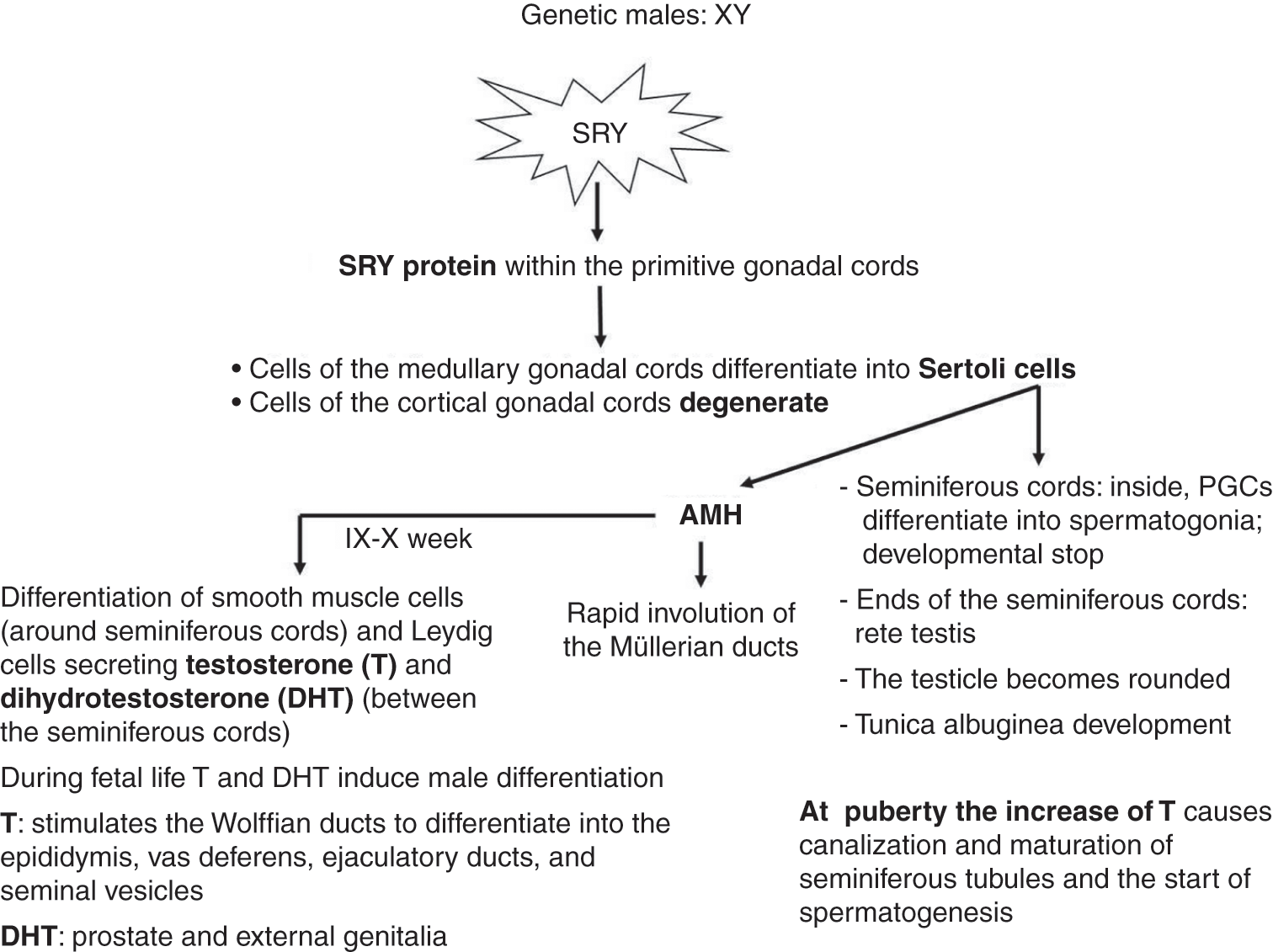
In the 7th week, the differentiation of male gonads (testes) begins (Figure 1.5).

♂Figure 1.5 Diagram of male sexual differentiation.
Thanks to the action of masculinizing factors, produced by the activation of Sry gene (present in the Y chromosome), the gonadal cords extend in the medullary of the genital ridges and lose contact with the coelomic epithelium defining the seminiferous cords characterized by Sertoli cells forming the cord wall. The PGCs that differentiate into spermatogonia are located within the seminiferous cord, although the Sertoli cells inhibit their development before the start of meiosis. The ends of the seminiferous cords ramify and anastomose to form the rete testis. During the 7th week, the testicle begins to round off, reducing the contact surface with the mesonephros. Subsequently, the seminiferous cords lose contact with the coelomic epithelium through connective tissue, which forms the tunica albuginea. When future Sertoli cells begin their morphological differentiation, they begin to produce a protein called antimüllerian factor (AMH). AMH causes regression of the paramesonephric (Müller) ducts in males, via an apoptotic mechanism.
Smooth muscle cells differentiate around the seminiferous cords and the Leydig cells differentiate in the areas between the seminiferous cords. The Leydig cells produce testosterone, a steroid hormone necessary for the subsequent development of the testicle itself, of the male genital tract, and of the external genitalia. In the 20th week, the testicles move from the abdominal wall of the embryo to the scrotum.Reference Teerds and Huhtaniemi19
At puberty, the increase in testosterone causes canalization of the seminiferous cords containing the PGCs, forming seminiferous tubules. Even the seminiferous cords superiorly located (rete testis) develop a lumen and differentiate into thin-walled ducts.
The male genital tract mainly originates from the Wolffian ducts. The permanence of the Wolffian ducts is regulated by androgens, particularly testosterone and dihydrotestosterone (DHT) produced by the Leydig cells. The rete testis, formed from the ends of the anastomosed seminiferous cords, is connected to 15–20 mesonephric tubules (transitional primitive kidney) that become efferent tubules connected to Wolffian ducts that develop into the epididymis in the proximal portion to the testicle and the vas deferens in the distal portion. The vas deferens continues with the ejaculatory ducts that open in the prostatic urethra, which continues with the penile urethra, both arising from the urogenital sinus. The seminal vesicles originate near the opening of the Wolffian ducts in the prostatic urethra. The prostate and bulb-urethral glands originate from the endoderm of the urogenital sinus.
The male external genitalia (penis, penile urethra, and scrotum) originate from the differentiation of the genital tubercle, which elongates to form the penis, of the urethral folds, which merge to close the orifice of the urogenital sinus and form the penile urethra, and of the genital swellings, which merge down the midline to form the scrotum to house the testicles after their descent in the lumbar region (Figure 1.6, Table 1.1)

♂♀Figure 1.6 Molecular regulation of sexual differentiation.
| Initial Draft | Male Structures | Female Structures |
|---|---|---|
| Primordial germ cells (PGCs) | Spermatogonia | Oogonia |
| Epithelium of the genital ridge | Sertoli cells | Follicular cells |
| Mesenchyme of the genital ridge | Smooth muscle cellsLeydig cells | Thecal cells |
| Wolffian ducts | EpididymisVas deferensEjaculatory ductSeminal vesiclesAppendix of the epididymis | Degenerate, only residues remain: Gartner’s duct Gartner’s cyst |
| Müllerian ducts | Degenerate, only residues remain: Appendix of the testicle Prostatic uricle | OviductsUterusCervixThe upper third of the vagina |
| Mesonephric tubules | Vas deferens | Degenerate, only residues remain: Epoophoron Paraoforon |
| Genital tubercle | Glans and penis shaft | Clitoris |
| Definitive urogenital sinus | Penile urethra | Vestibule of the vagina |
| Urethral folds | Portion of the penis surrounding the penile urethra | Labia minora |
| Genital swellings | Scrotum | Labia majora |
♀1.5 Female
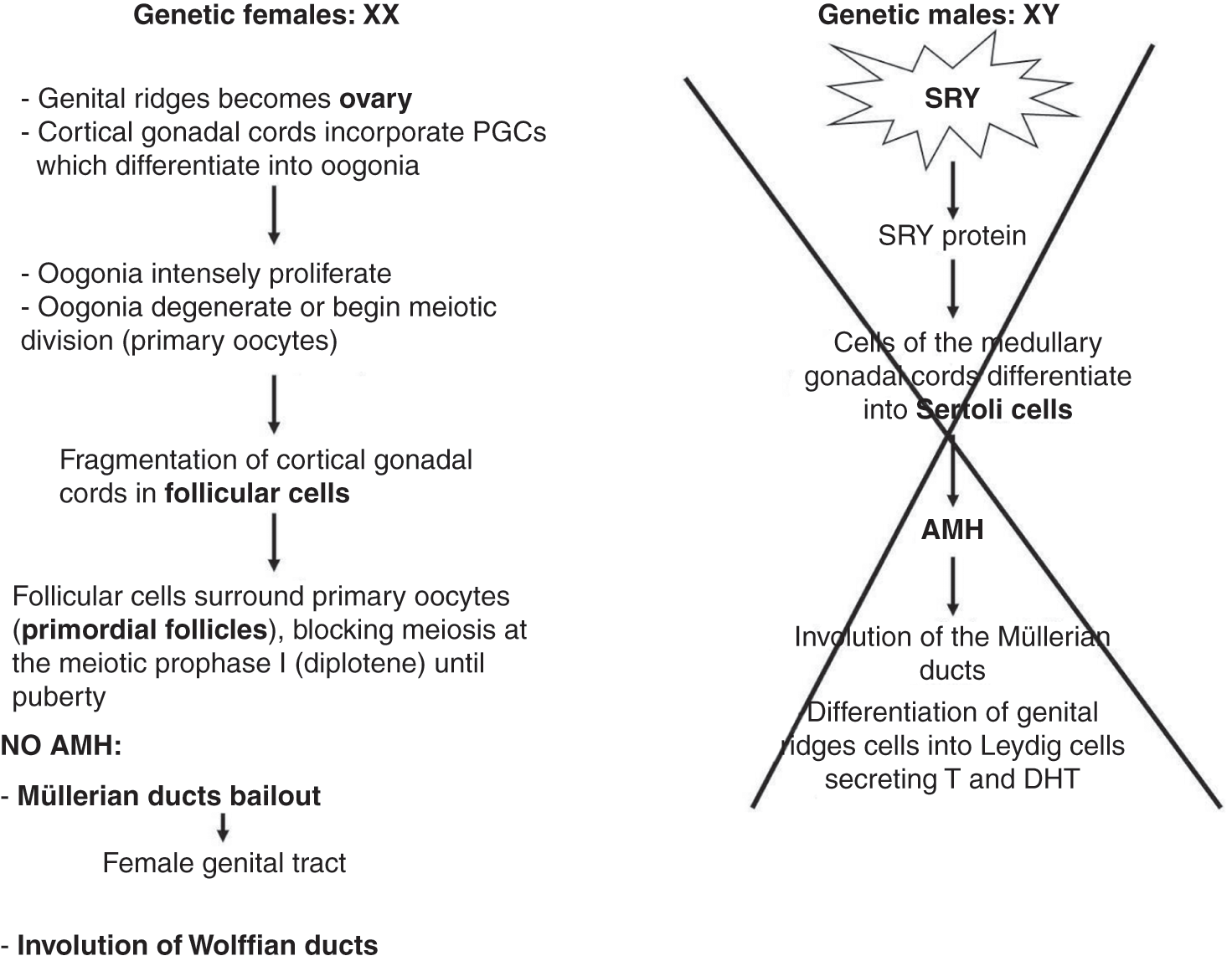
In the female embryo, the somatic cells of the genital cords do not have a Y chromosome, and therefore do not activate the TDF and differentiate into Sertoli cells. As a result, AMH, Leydig cells, and testosterone are not produced, thus there is no stimulus for male sexual differentiation. In these conditions, sexual differentiation is female. In female embryos, somatic cells of the genital cords differentiate into follicular cells, and genital ridge becomes the ovary (Figures 1.6 and 1.7).

♀Figure 1.7 Diagram of female sexual differentiation.
In the female embryo, the ovary is not morphologically identifiable until the 10th week.
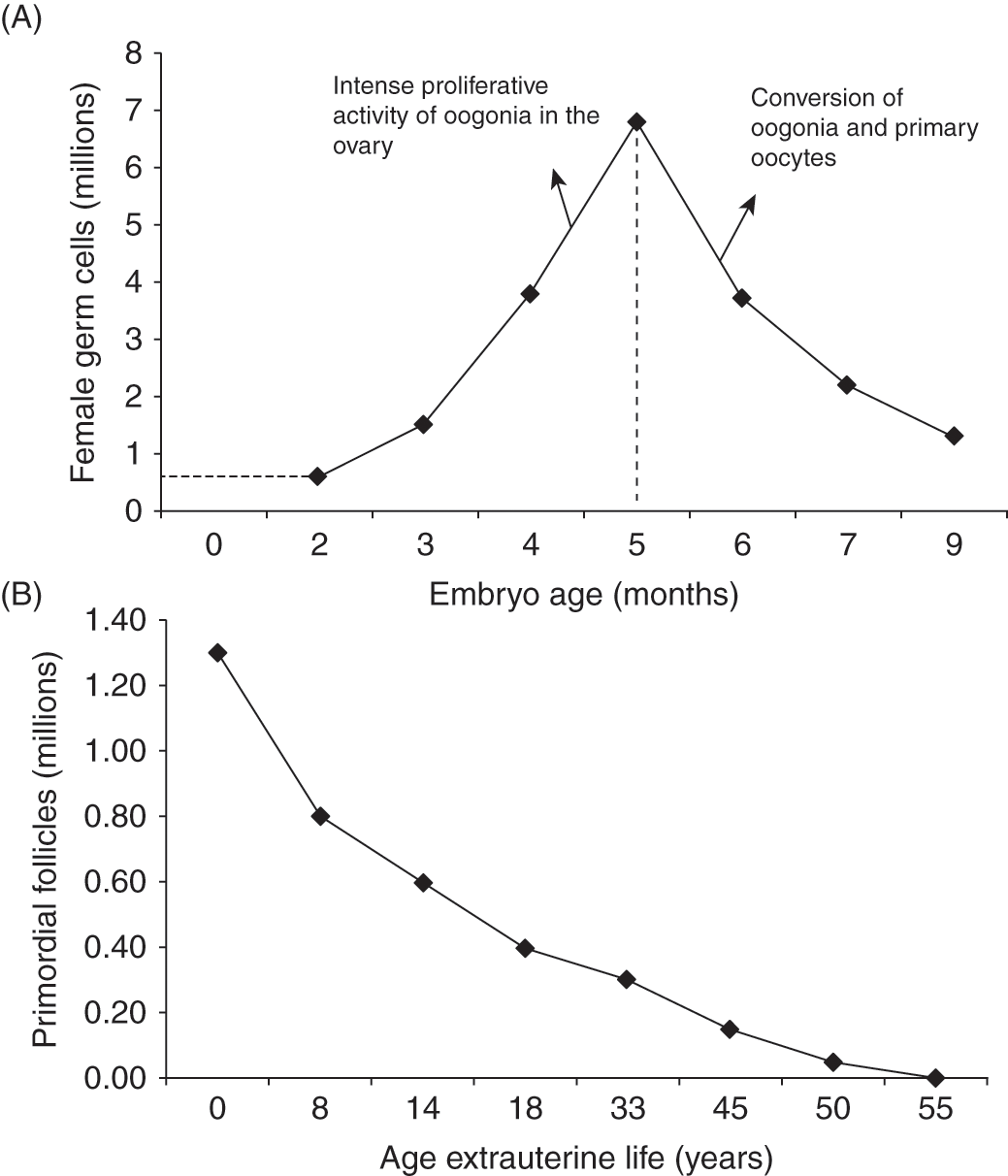
The cortical genital cords extend in the medullary of the genital ridge forming a rudimentary rete ovari, a network of tubules that subsequently degenerates and disappears. The cortical genital cords incorporate PGCs. These latter differentiate into oogonia, which intensively proliferate (Figure 1.8) after the proliferative phase; the oogonium degenerates or undertakes meiotic division. Female PGCs that begin the first meiotic division are called primary oocytes. Within the 7th month all oogonia become primary oocytes and conclude prophase I of meiosis (diplotene).

♀Figure 1.8 Graphs of the behavior of female germ cells: (A) between the 2nd and the 9th month of intrauterine life; (B) during puberty and childbearing years.
At about 16 weeks, the cortical genital cords begin to break up into groups of cells, forming the follicular cells. Between the 5th and the 9th months, the primordial follicles are generated; they are composed of four or five flattened follicular cells with a primary oocyte in the diplotene stage. Follicular cells cause the blockage of meiosis until puberty, when the individual oocytes again begin gametogenesis in response to the periodic rise of gonadotropin levels during the ovarian cycle. Differently from the male, at birth there are no PGCs in the ovary, only primary oocytes in the diplotene stage.
The tissue located among the primordial follicles is called interstitial tissue, rich in vessels and nerves. The ovary is coated with an epithelium derived from the coelomic epithelium called the ovarian surface epithelium (OSE). Before birth, the ovaries move from the posterior abdominal wall to the pelvis. At birth, due to degeneration and differentiation processes, the two ovaries contain a total of about 1 000 000 primordial follicles, each containing a primary oocyte at the diplotene stage.
From birth until puberty, some primordial follicles begin but do not complete their development and degenerate through a process called atresia. There is also considerable degeneration of the follicles within the fertile period. Normally in adult women, only one follicle completes development each month, releasing a mature oocyte for fertilization (ovulation). It has been calculated that only about 400 000 oocytes are present at puberty (1 000 000 at birth), of which only 300–400 restart meiosis and are ovulated during the fertile years. The exhaustion of the oocytes occurs at approximately 50 years of age and determines the end of the fertile period and the start of menopause. The absence of PGCs or oogonia in the ovary is of considerable clinical importance, in fact, harmful agents such as ionizing radiation and chemotherapeutic compounds cause irreversible damage to the oocytes and early infertility in the woman.
The female genital tract (oviducts, uterus, and vagina) originate from the Müllerian ducts thanks to the estrogen produced by the ovaries, while the Wolffian ducts regress due to the absence of testosterone and AMH, necessary to be trophic. The cranial portions of the Müller ducts do not merge with each other and give rise to the oviducts that open in the coelom near the ovaries; the most caudal portions merge along the midline and give rise to the uterus and the upper part of the vagina. The lower portion of the vagina originates from urogenital sinus derived from the cloaca, allowing the opening of the vagina in the vestibule of the vagina that originates from the definitive urogenital sinus.
The female external genitalia (labia minora, labia majora, clitoris, and vestibule of the vagina) originate from the differentiation of the genital tubercle to form the clitoris, of the urethral folds to form the labia minora, of the genital swellings to form the labia majora, and of the definitive urogenital sinus to form the vestibule of the vagina. The differentiation of the external genitalia appears after the 12th week and is a hormone-dependent process (Tables 1.1 and 1.2).Reference Rodriguez, Weiss and Ferretti20
| Weeks | Events | |
|---|---|---|
| Embryonic period | 3 | Differentiation of PGCs |
| 4–5 | PGCs migration to the gonadal drafts | |
| Development of mesonephric ducts | ||
| 5–6 | Development of genital ridges | |
| SRY expression | ||
| 6 | Beginning of the external genitalia development | |
| 7 | Sexual differentiation of gonads | |
| Development of paramesonephric ducts | ||
| 8 | Sertoli cells begin to produce AMH | |
| Differentiation of Leydig cells | ||
| Fetal Period | 9 | The Leydig cells begin to produce testosterone |
| Beginning of the masculinization of both urogenital sinus and external genitalia | ||
| 10–13 | Differentiation of the genital tract | |
| Beginning of meiosis in the ovaries | ||
| Male and female external genitalia identifiable | ||
| 14 | Organogenesis of the male urethra is completed | |
| 20 | Formation of the first primordial follicles in the ovaries | |
| 27–30 | Desensus testis in inguinal-scrotal location |