Refine search
Actions for selected content:
106116 results in Materials Science
Investigation into boron nitride nanoparticle effects on thermal properties of calcium chloride hexahydrate (CaCl2·6H2O) as a phase change material
-
- Journal:
- MRS Communications / Volume 8 / Issue 4 / December 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 1439-1444
- Print publication:
- December 2018
-
- Article
- Export citation
A call to action: High entropy alloy manufacturing
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 19 / 14 October 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 2855-2856
- Print publication:
- 14 October 2018
-
- Article
- Export citation
JMR volume 33 issue 19 Cover and Front matter
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 19 / 14 October 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. f1-f6
- Print publication:
- 14 October 2018
-
- Article
-
- You have access
- Export citation
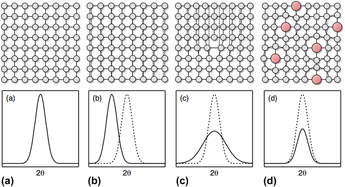
Lattice distortions in high-entropy alloys
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 19 / 14 October 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 2954-2969
- Print publication:
- 14 October 2018
-
- Article
- Export citation
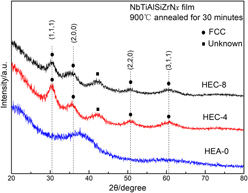
Mechanical properties and thermal stability of (NbTiAlSiZr)Nx high-entropy ceramic films at high temperatures
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 19 / 14 October 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 3347-3354
- Print publication:
- 14 October 2018
-
- Article
- Export citation
Polydiacetylene ribbons formed using the controlled evaporative self-assembly (CESA) method
-
- Journal:
- MRS Communications / Volume 9 / Issue 1 / March 2019
- Published online by Cambridge University Press:
- 12 October 2018, pp. 229-235
- Print publication:
- March 2019
-
- Article
- Export citation
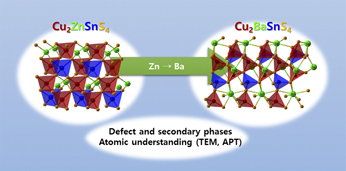
Atomistic consideration of earth-abundant chalcogenide materials for photovoltaics: Kesterite and beyond
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 23 / 14 December 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 3986-3998
- Print publication:
- 14 December 2018
-
- Article
- Export citation
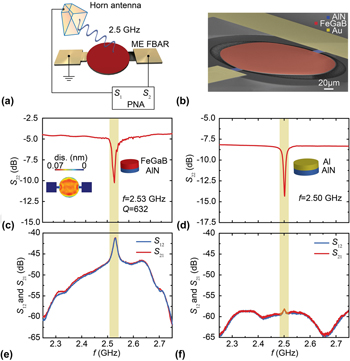
Integrated ferroics for sensing, power, RF, and µ-wave electronics
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 23 / 14 December 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 4007-4017
- Print publication:
- 14 December 2018
-
- Article
- Export citation
In situ synthesis of CsTi2NbO7@g-C3N4 core–shell heterojunction with excellent electrocatalytic performance for the detection of nitrite
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 23 / 14 December 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 3936-3945
- Print publication:
- 14 December 2018
-
- Article
- Export citation
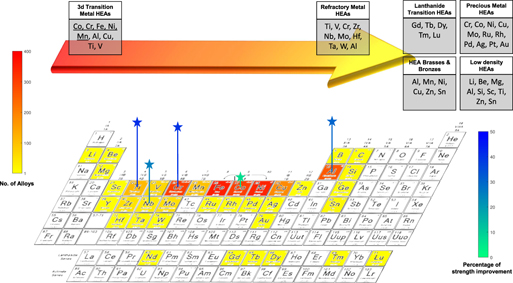
Strengthening mechanisms in high-entropy alloys: Perspectives for alloy design
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 19 / 14 October 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 2970-2982
- Print publication:
- 14 October 2018
-
- Article
- Export citation
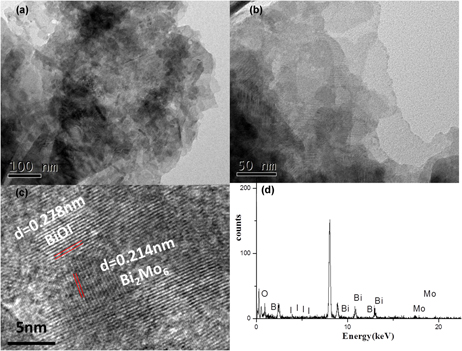
Fabrication of Bi2MoO6/BiOI heterojunction photocatalysts for enhanced photodegradation of RhB
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 23 / 14 December 2018
- Published online by Cambridge University Press:
- 12 October 2018, pp. 3928-3935
- Print publication:
- 14 December 2018
-
- Article
- Export citation
Poisson errors and adaptive rebinning in X-ray powder diffraction data
-
- Journal:
- Powder Diffraction / Volume 33 / Issue 4 / December 2018
- Published online by Cambridge University Press:
- 10 October 2018, pp. 266-269
-
- Article
- Export citation
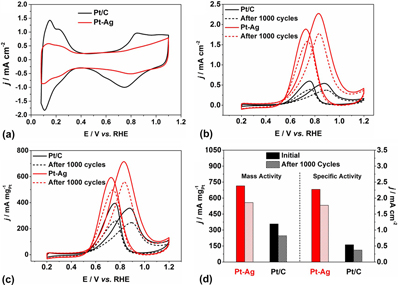
Facile synthesis of Pt–Ag octahedral and tetrahedral nanocrystals with enhanced activity and durability toward methanol oxidation
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 23 / 14 December 2018
- Published online by Cambridge University Press:
- 08 October 2018, pp. 3891-3897
- Print publication:
- 14 December 2018
-
- Article
- Export citation
A high-quality mechanofusion coating for enhancing lithium-ion battery cathode material performance
-
- Journal:
- MRS Communications / Volume 9 / Issue 1 / March 2019
- Published online by Cambridge University Press:
- 05 October 2018, pp. 245-250
- Print publication:
- March 2019
-
- Article
- Export citation
Hydrothermal synthesis of gold nanoplates and their structure-dependent LSPR properties – CORRIGENDUM
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 20 / 29 October 2018
- Published online by Cambridge University Press:
- 05 October 2018, p. 3537
- Print publication:
- 29 October 2018
-
- Article
-
- You have access
- HTML
- Export citation
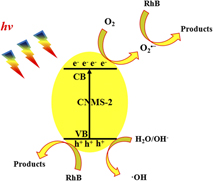
Simultaneous morphology, band structure, and defect optimization of graphitic carbon nitride microsphere by the precursor concentration to boost photocatalytic activity
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 23 / 14 December 2018
- Published online by Cambridge University Press:
- 04 October 2018, pp. 3917-3927
- Print publication:
- 14 December 2018
-
- Article
- Export citation
Crystal structure of vardenafil hydrochloride trihydrate, C23H33N6O4SCl (H2O)3
-
- Journal:
- Powder Diffraction / Volume 33 / Issue 4 / December 2018
- Published online by Cambridge University Press:
- 04 October 2018, pp. 319-326
-
- Article
- Export citation
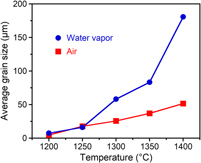
Grain growth of titania to submillimeter sizes using water vapor-assisted sintering
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 3 / 14 February 2019
- Published online by Cambridge University Press:
- 02 October 2018, pp. 474-480
- Print publication:
- 14 February 2019
-
- Article
- Export citation
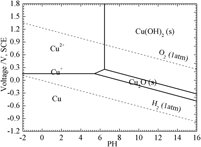
Effect of Cu on the corrosion resistance and electrochemical response of a Ni–Co–Cr–Mo alloy in acidic chloride solution
-
- Journal:
- Journal of Materials Research / Volume 33 / Issue 22 / 28 November 2018
- Published online by Cambridge University Press:
- 02 October 2018, pp. 3801-3808
- Print publication:
- 28 November 2018
-
- Article
- Export citation
Vortex glass-liquid transition and activated flux motion in an epitaxial, superconducting NdFeAs(O,F) thin film
-
- Journal:
- MRS Communications / Volume 8 / Issue 4 / December 2018
- Published online by Cambridge University Press:
- 02 October 2018, pp. 1433-1438
- Print publication:
- December 2018
-
- Article
- Export citation