Refine search
Actions for selected content:
106116 results in Materials Science
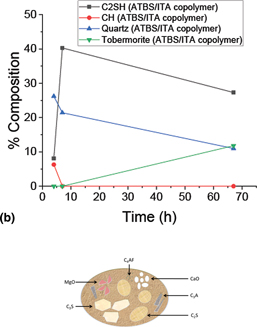
The impact of powerful retarding additive synergies on the kinetic profile of cementitious mineralogical transformations at high temperature
-
- Journal:
- MRS Communications / Volume 9 / Issue 3 / September 2019
- Published online by Cambridge University Press:
- 19 June 2019, pp. 1022-1028
- Print publication:
- September 2019
-
- Article
- Export citation
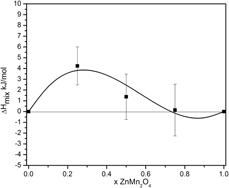
Thermodynamics of ZnxMn3−xO4 and Mg1−zCuzCr2O4 spinel solid solutions
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 19 / 14 October 2019
- Published online by Cambridge University Press:
- 17 June 2019, pp. 3305-3311
- Print publication:
- 14 October 2019
-
- Article
- Export citation

Handbook of Industrial Crystallization
-
- Published online:
- 14 June 2019
- Print publication:
- 20 June 2019
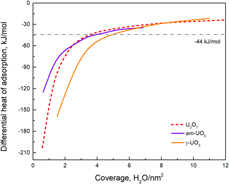
Energetics of hydration on uranium oxide and peroxide surfaces
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 19 / 14 October 2019
- Published online by Cambridge University Press:
- 14 June 2019, pp. 3319-3325
- Print publication:
- 14 October 2019
-
- Article
- Export citation
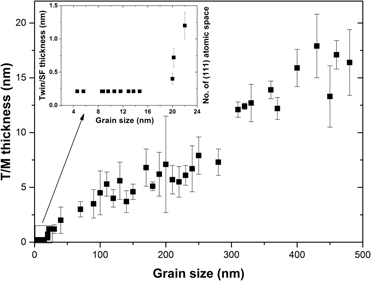
Grain size effect on deformation twin thickness in a nanocrystalline metal with low stacking-fault energy
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 13 / 15 July 2019
- Published online by Cambridge University Press:
- 14 June 2019, pp. 2398-2405
- Print publication:
- 15 July 2019
-
- Article
- Export citation
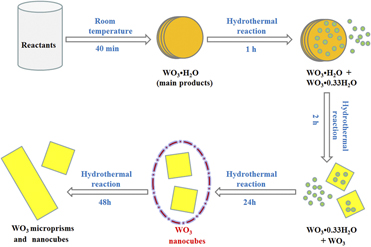
WO3 nanocubes: Hydrothermal synthesis, growth mechanism, and photocatalytic performance
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 17 / 16 September 2019
- Published online by Cambridge University Press:
- 14 June 2019, pp. 2955-2963
- Print publication:
- 16 September 2019
-
- Article
- Export citation
JMR volume 34 issue 11 Cover and Front matter
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 11 / 14 June 2019
- Published online by Cambridge University Press:
- 14 June 2019, pp. f1-f5
- Print publication:
- 14 June 2019
-
- Article
-
- You have access
- Export citation
JMR volume 34 issue 11 Cover and Back matter
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 11 / 14 June 2019
- Published online by Cambridge University Press:
- 14 June 2019, pp. b1-b4
- Print publication:
- 14 June 2019
-
- Article
-
- You have access
- Export citation
Introduction
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 11 / 14 June 2019
- Published online by Cambridge University Press:
- 14 June 2019, p. 1827
- Print publication:
- 14 June 2019
-
- Article
- Export citation
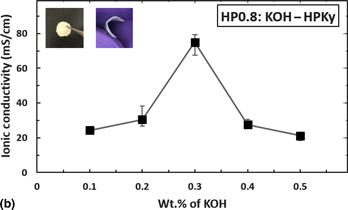
Wood cellulose-based thin gel electrolyte with enhanced ionic conductivity
-
- Journal:
- MRS Communications / Volume 9 / Issue 3 / September 2019
- Published online by Cambridge University Press:
- 13 June 2019, pp. 1015-1021
- Print publication:
- September 2019
-
- Article
- Export citation
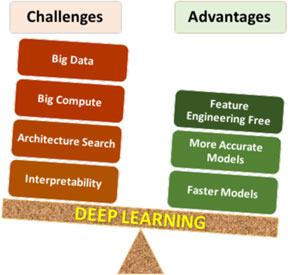
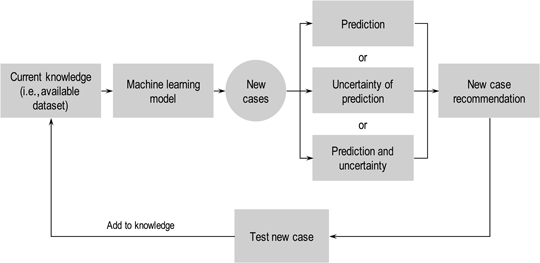
Deep materials informatics: Applications of deep learning in materials science
-
- Journal:
- MRS Communications / Volume 9 / Issue 3 / September 2019
- Published online by Cambridge University Press:
- 13 June 2019, pp. 779-792
- Print publication:
- September 2019
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Active-learning and materials design: the example of high glass transition temperature polymers
-
- Journal:
- MRS Communications / Volume 9 / Issue 3 / September 2019
- Published online by Cambridge University Press:
- 13 June 2019, pp. 860-866
- Print publication:
- September 2019
-
- Article
- Export citation
High-temperature properties of titanium-substituted yttrium niobate
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 19 / 14 October 2019
- Published online by Cambridge University Press:
- 11 June 2019, pp. 3312-3318
- Print publication:
- 14 October 2019
-
- Article
- Export citation
Amphiphilic fluorescent copolymers via one-pot synthesis of RAFT polymerization and multicomponent Biginelli reaction and their cells imaging applications
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 17 / 16 September 2019
- Published online by Cambridge University Press:
- 11 June 2019, pp. 3011-3019
- Print publication:
- 16 September 2019
-
- Article
- Export citation
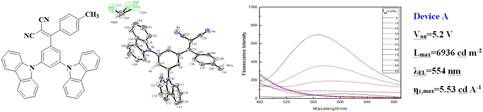
Synthesis, crystal structure, photoluminescence, and electroluminescence properties of a new compound containing diphenylmethylene, carbazole, and malononitrile units
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 17 / 16 September 2019
- Published online by Cambridge University Press:
- 10 June 2019, pp. 3000-3010
- Print publication:
- 16 September 2019
-
- Article
- Export citation
Identifying rhenium substitute candidate multiprincipal-element alloys from electronic structure and thermodynamic criteria
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 19 / 14 October 2019
- Published online by Cambridge University Press:
- 10 June 2019, pp. 3296-3304
- Print publication:
- 14 October 2019
-
- Article
- Export citation
Study of diffraction data sets using factor analysis: a new technique for comparing mineralogical and geochemical data and rapid diagnostics of the mineral composition of large collections of rock samples
-
- Journal:
- Powder Diffraction / Volume 34 / Issue S1 / September 2019
- Published online by Cambridge University Press:
- 07 June 2019, pp. S59-S70
-
- Article
- Export citation
A model revealing grain boundary arrangement-dominated fatigue cracking behavior in nanoscale metallic multilayers
-
- Journal:
- MRS Communications / Volume 9 / Issue 3 / September 2019
- Published online by Cambridge University Press:
- 07 June 2019, pp. 936-940
- Print publication:
- September 2019
-
- Article
- Export citation
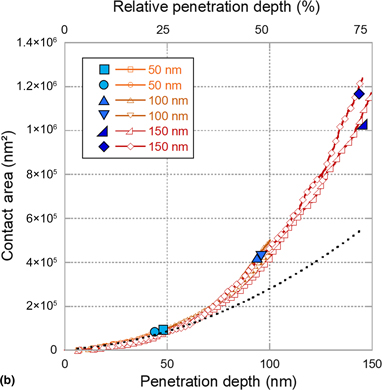
Resistive-nanoindentation: contact area monitoring by real-time electrical contact resistance measurement
-
- Journal:
- MRS Communications / Volume 9 / Issue 3 / September 2019
- Published online by Cambridge University Press:
- 07 June 2019, pp. 1008-1014
- Print publication:
- September 2019
-
- Article
- Export citation
A self-aligning microtensile setup: Application to single-crystal GaAs microscale tension–compression asymmetry
-
- Journal:
- Journal of Materials Research / Volume 34 / Issue 14 / 28 July 2019
- Published online by Cambridge University Press:
- 07 June 2019, pp. 2517-2534
- Print publication:
- 28 July 2019
-
- Article
-
- You have access
- Open access
- HTML
- Export citation