Refine listing
Actions for selected content:
142352 results in Open Access
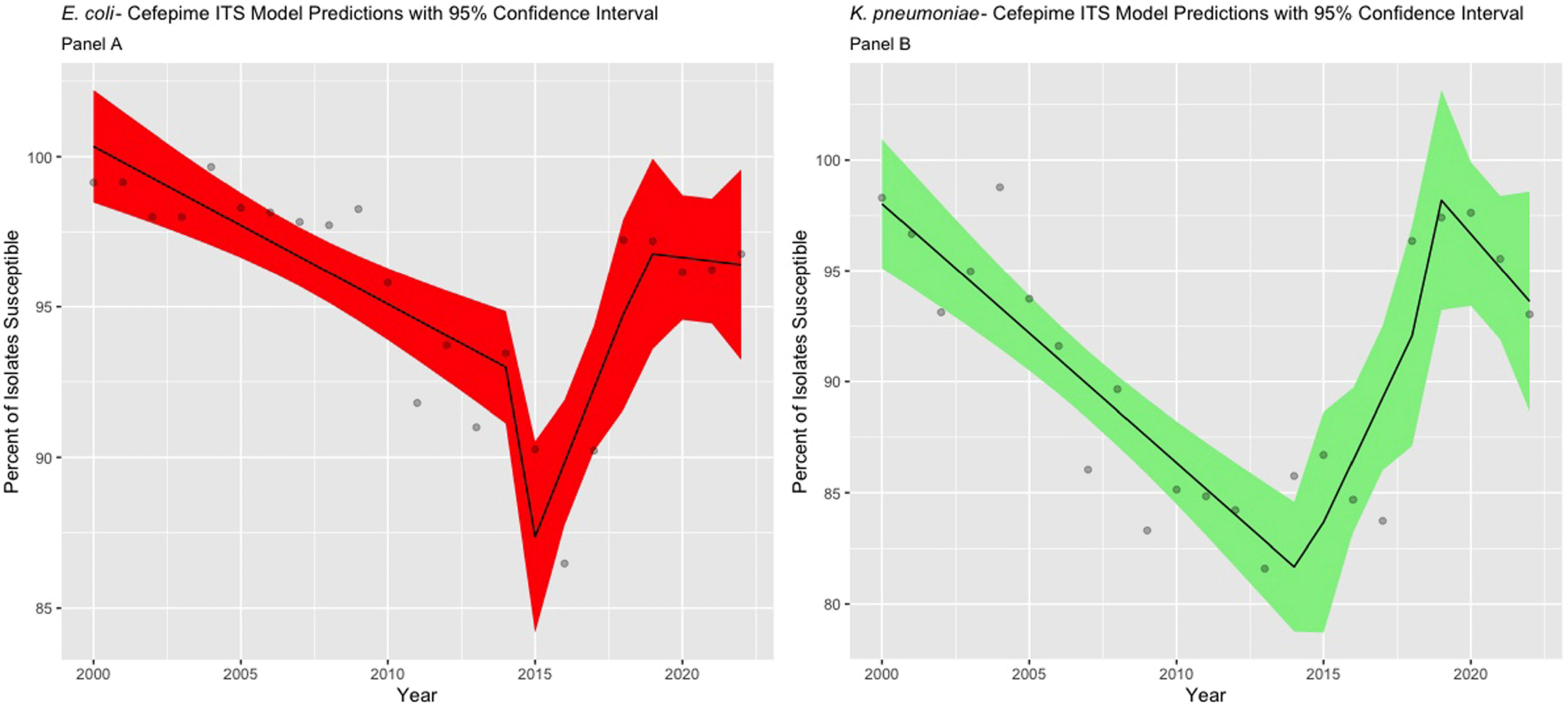
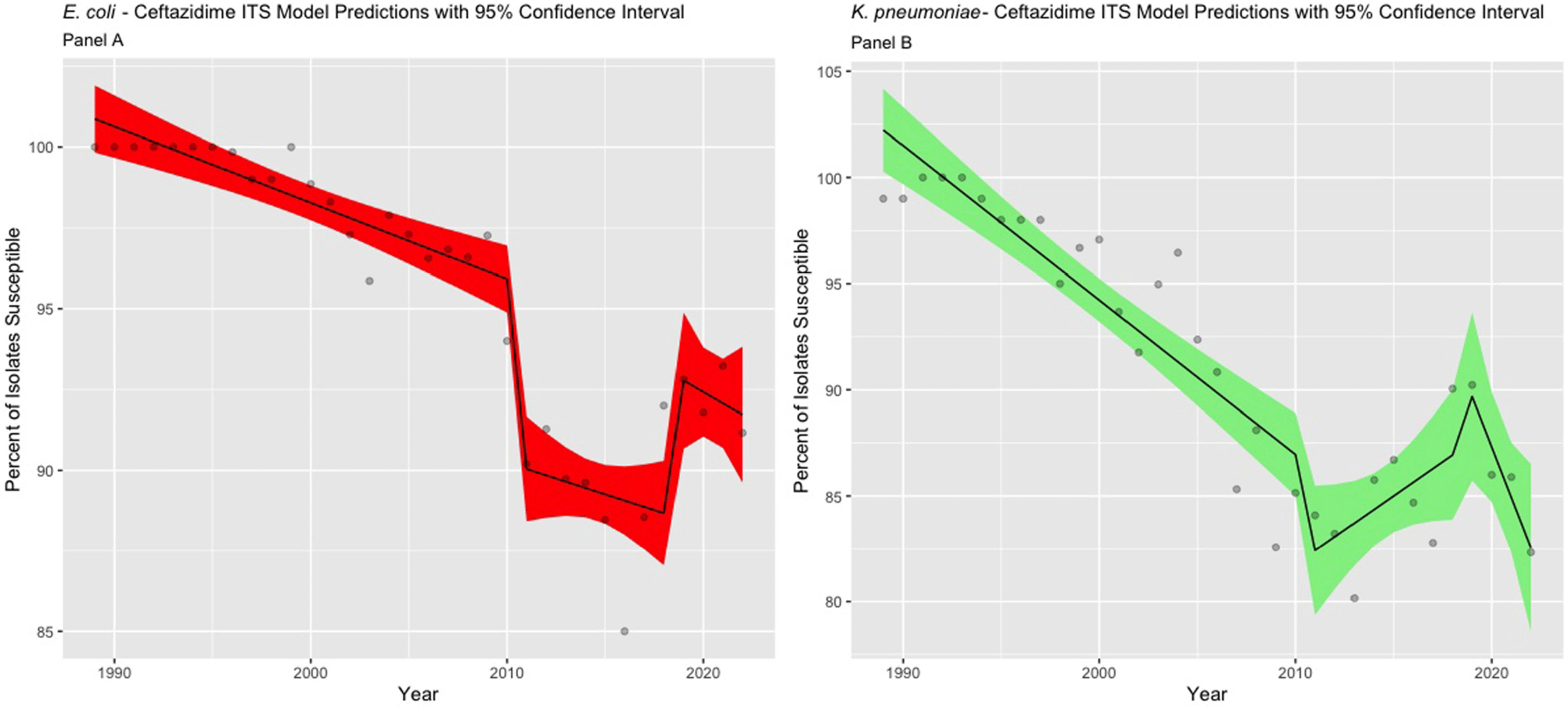
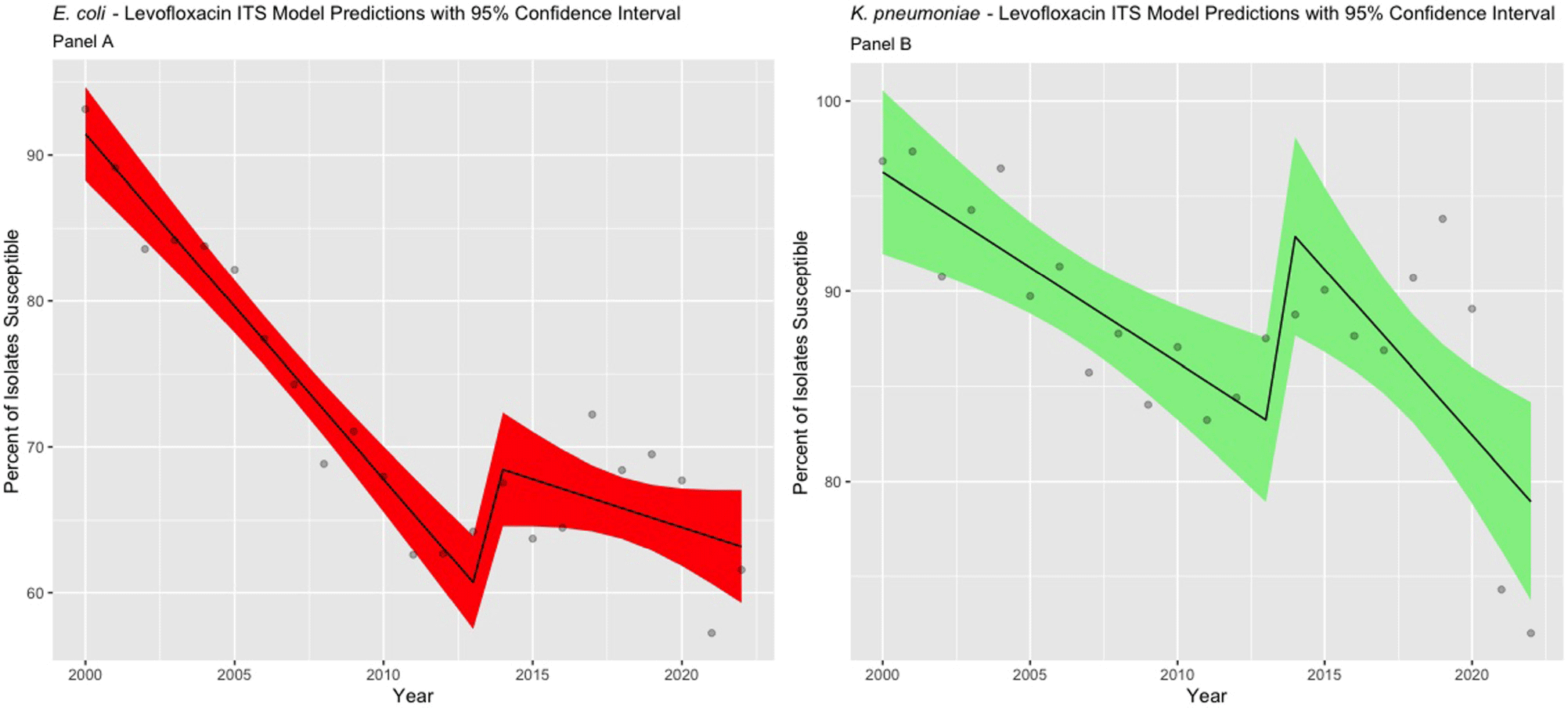
Impact of MIC Breakpoint Changes for Enterobacterales on Trends of Antibiotic Susceptibilities in An Academic Medical Center
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s54-s56
-
- Article
-
- You have access
- Open access
- Export citation
A little rain
-
- Journal:
- Palliative & Supportive Care / Volume 22 / Issue 5 / October 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. 1537
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Nosocomial Transmission of Mycobacterium tuberculosis in an Oncological Setting
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s107
-
- Article
-
- You have access
- Open access
- Export citation
Optimal Weight-Based Dosing of Vancomycin to Achieve an Area Under the Curve of 400 to 600 Stratified by Body Mass Index
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s131
-
- Article
-
- You have access
- Open access
- Export citation
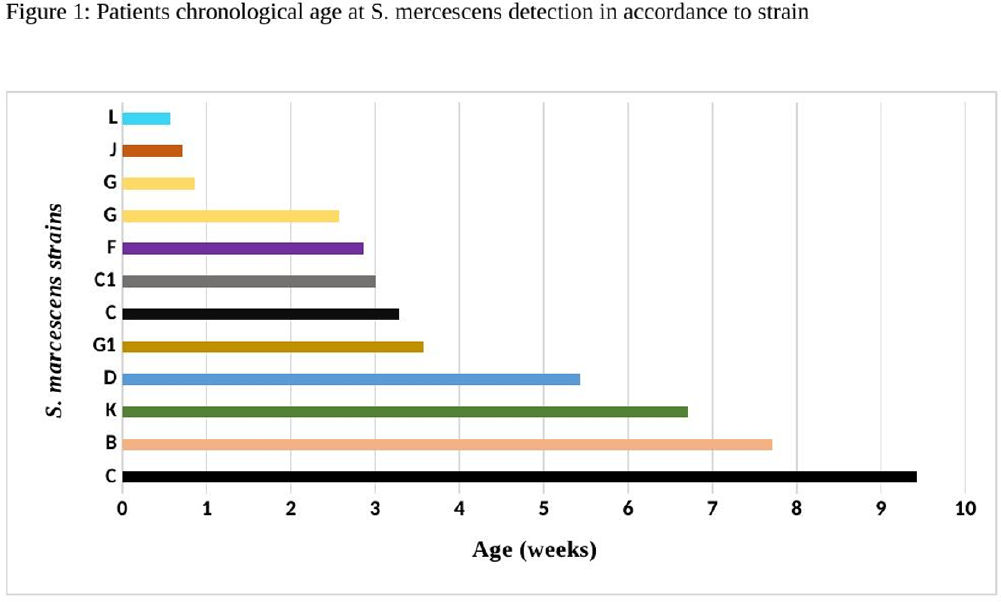
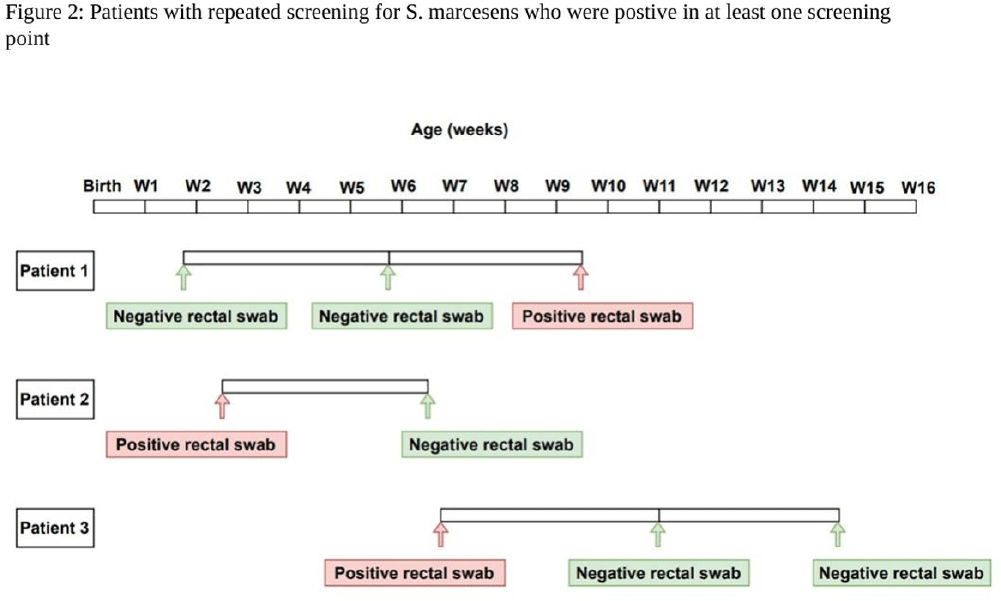
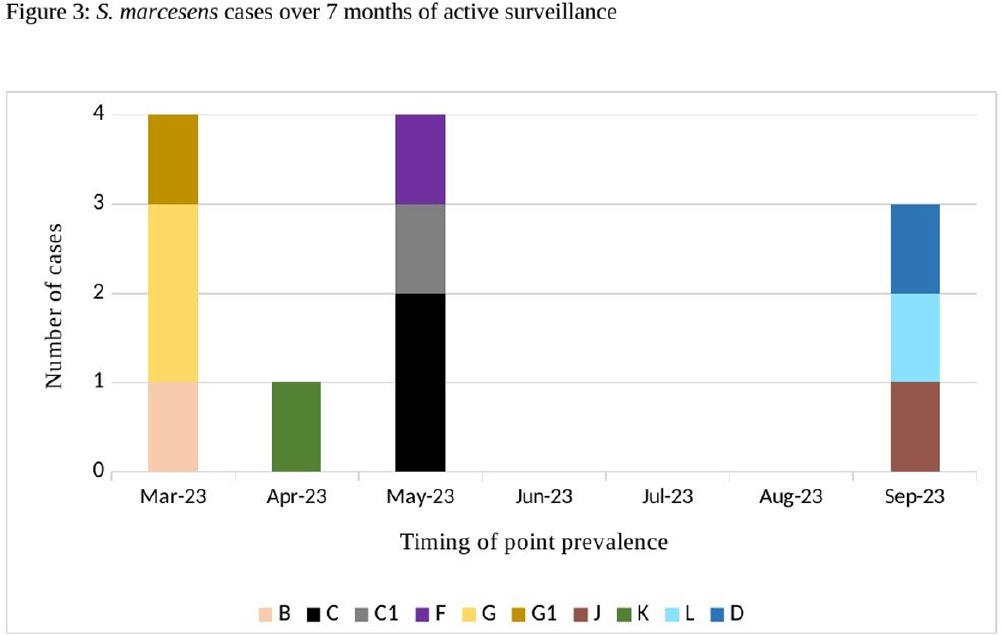
Serratia marcescens Burden in a Neonatal Intensive Care Unit: Colonization Rate, Clinical Infections and Strain Relatedness
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s153-s154
-
- Article
-
- You have access
- Open access
- Export citation
Microbiologic Evaluation of Colorectal Surgical Site Infections to Guide Surgical Prophylaxis Recommendations
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s143
-
- Article
-
- You have access
- Open access
- Export citation
User-Centered Education for Patients/Caregivers about Urinary Tract Infections, Asymptomatic Bacteriuria, and Antibiotics
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s5-s6
-
- Article
-
- You have access
- Open access
- Export citation
Practical, effective and safer: Placing traps above ground is an improved capture method for the critically endangered ngwayir (western ringtail possum; Pseudocheirus occidentalis)
- Part of
-
- Journal:
- Animal Welfare / Volume 33 / 2024
- Published online by Cambridge University Press:
- 16 September 2024, e29
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
A Stepwise Diagnostic Stewardship Approach to Reduce Unnecessary Urine Cultures, Asymptomatic Bacteriuria, and CAUTI Rate
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s79-s80
-
- Article
-
- You have access
- Open access
- Export citation
Comparison of COVID-19 Sentinel Surveillance and COVID-19 School Absentee Surveillance in Japan
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s148-s149
-
- Article
-
- You have access
- Open access
- Export citation
Sustainability of Surgical Site Infection (SSI) Prevention Bundle for Pediatric Cardiothoracic Surgery Patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s145-s146
-
- Article
-
- You have access
- Open access
- Export citation
Clearing the Air, Breathe Easy: Intensive Care Unit Remodeling Unveils Insights into Aspergillosis Prevention
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s126
-
- Article
-
- You have access
- Open access
- Export citation
An “Epic” Journey to Improve Antimicrobial Stewardship
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s157-s159
-
- Article
-
- You have access
- Open access
- Export citation
The Mechanics, Art, and Value of Central Line Stewardship
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s71-s72
-
- Article
-
- You have access
- Open access
- Export citation
Improving Consistency and Accuracy: A Novel C. auris Colonization Screening Strategy Using a Nares + Hands Composite Swab
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s112-s113
-
- Article
-
- You have access
- Open access
- Export citation
Assessing Mupirocin Resistance in MRSA Isolates in Hospitals in Cleveland, OH and Detroit, MI
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s117
-
- Article
-
- You have access
- Open access
- Export citation
Viral Kinetics of SARS-CoV-2 in Nursing Home Residents and Staff
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s73-s74
-
- Article
-
- You have access
- Open access
- Export citation
Antimicrobial Use in Veterans Affairs Community Living Centers, 2015 - 2019
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s6-s7
-
- Article
-
- You have access
- Open access
- Export citation
Epidemiology and Duration of Therapy in Patients with Gram-negative Bloodstream Infections: Retrospective Analysis
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, p. s43
-
- Article
-
- You have access
- Open access
- Export citation
Hospital-Onset Clostridioides difficile infection in chronic kidney disease patients
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 4 / Issue S1 / July 2024
- Published online by Cambridge University Press:
- 16 September 2024, pp. s68-s69
-
- Article
-
- You have access
- Open access
- Export citation