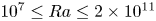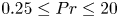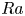Refine listing
Actions for selected content:
1418748 results in Open Access
Bouncing behaviour of a particle settling through a density transition layer
-
- Journal:
- Journal of Fluid Mechanics / Volume 997 / 25 October 2024
- Published online by Cambridge University Press:
- 16 October 2024, A49
-
- Article
- Export citation
Characterization of the end-use Quality of Soft Wheat Cultivars from the Eastern and Western US Germplasm ‘Pools’
-
- Journal:
- Plant Genetic Resources / Volume 2 / Issue 1 / April 2004
- Published online by Cambridge University Press:
- 16 October 2024, pp. 59-69
-
- Article
- Export citation
Pharmacoepidemiology of combination pulmonary vasodilator therapy in critically ill infants
-
- Journal:
- Cardiology in the Young / Volume 35 / Issue 1 / January 2025
- Published online by Cambridge University Press:
- 16 October 2024, pp. 93-101
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Looking to Livestock: Gauging the Evolution of the EU's Agri-Climate Law and Policy
-
- Journal:
- Transnational Environmental Law / Volume 14 / Issue 1 / March 2025
- Published online by Cambridge University Press:
- 16 October 2024, pp. 69-93
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Simone Guesser, Ani Carla Marchesan and Paulo Medeiros Junior (eds.), Wh-exclamatives, imperatives and wh-questions: Issues on Brazilian Portuguese. Berlin: De Gruyter, 2024. Pp. 463.
-
- Journal:
- Journal of Linguistics / Volume 61 / Issue 1 / February 2025
- Published online by Cambridge University Press:
- 16 October 2024, pp. 203-207
- Print publication:
- February 2025
-
- Article
- Export citation
VERTEX-PRIMITIVE s-ARC-TRANSITIVE DIGRAPHS OF ALMOST SIMPLE GROUPS
- Part of
-
- Journal:
- Bulletin of the Australian Mathematical Society / Volume 110 / Issue 3 / December 2024
- Published online by Cambridge University Press:
- 16 October 2024, pp. 566-567
- Print publication:
- December 2024
-
- Article
-
- You have access
- HTML
- Export citation
Early life health adversity and internalizing disorders in the transition from adolescence to adulthood
-
- Journal:
- Development and Psychopathology / Volume 37 / Issue 4 / October 2025
- Published online by Cambridge University Press:
- 16 October 2024, pp. 1846-1858
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Flow state transition induced by emergence of orbiting satellite eddies in two-dimensional turbulent Rayleigh–Bénard convection
-
- Journal:
- Journal of Fluid Mechanics / Volume 997 / 25 October 2024
- Published online by Cambridge University Press:
- 16 October 2024, A54
-
- Article
- Export citation
PGR volume 2 issue 1 Cover and Front matter
-
- Journal:
- Plant Genetic Resources / Volume 2 / Issue 1 / April 2004
- Published online by Cambridge University Press:
- 16 October 2024, pp. f1-f4
-
- Article
-
- You have access
- Export citation
How can you help us, Mr Darwin? Social Darwinism in the history of Chinese international thought
-
- Journal:
- Review of International Studies / Volume 51 / Issue 2 / March 2025
- Published online by Cambridge University Press:
- 16 October 2024, pp. 237-255
- Print publication:
- March 2025
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
A Naturalist's Defence of Meaning in Religious Pursuits
-
- Journal:
- Dialogue: Canadian Philosophical Review / Revue canadienne de philosophie , First View
- Published online by Cambridge University Press:
- 16 October 2024, pp. 1-20
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Global Animal Law from the Margins: International Trade in Animals and Their Bodies, by Iyan Offor Routledge, 2023, 320 pp, £145 hb, £35.99 ebk ISBN 9781032226989 hb, 9781003273783 ebk
-
- Journal:
- Transnational Environmental Law / Volume 13 / Issue 3 / November 2024
- Published online by Cambridge University Press:
- 16 October 2024, pp. 652-656
-
- Article
- Export citation
Momentum, energy and vorticity balances in deep-water surface gravity waves
-
- Journal:
- Journal of Fluid Mechanics / Volume 997 / 25 October 2024
- Published online by Cambridge University Press:
- 16 October 2024, A55
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Relationship between subjective cognitive functioning and fluid and crystallized cognitive abilities in bipolar disorder
-
- Journal:
- Journal of the International Neuropsychological Society / Volume 30 / Issue 8 / October 2024
- Published online by Cambridge University Press:
- 16 October 2024, pp. 719-727
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
Jennie E. Burnet. To Save Heaven and Earth: Rescue in the Rwandan Genocide. Ithaca, NY: Cornell University Press, 2023. xxix + 277 pp. Maps. Illustrations. Notes. Bibliography. Index. $35.95. Paper. ISBN: 9781501767111.
-
- Journal:
- African Studies Review / Volume 68 / Issue 1 / March 2025
- Published online by Cambridge University Press:
- 16 October 2024, pp. 203-204
-
- Article
-
- You have access
- HTML
- Export citation
EFFECT OF EXTERNAL HARMONIC VIBRATION ON THE FORMATION OF SOLITARY WAVES IN FALLING TWO-LAYER LIQUID FILMS
- Part of
-
- Journal:
- The ANZIAM Journal / Volume 67 / 2025
- Published online by Cambridge University Press:
- 16 October 2024, e5
-
- Article
-
- You have access
- Open access
- HTML
- Export citation
VALENTINA LIMINA, POTERI E STRATEGIE FAMILIARI DI VOLTERRA: IL CASO DI UNA COMUNITÀ ETRUSCA NEL MONDO ROMANO. Oxford: BAR Publishing, 2021. Pp. xiii + 194, illus. isbn 9781407357881. £48.00.
-
- Journal:
- The Journal of Roman Studies / Volume 114 / November 2024
- Published online by Cambridge University Press:
- 16 October 2024, pp. 230-231
- Print publication:
- November 2024
-
- Article
- Export citation
New Saccharum hybrids in S. spontaneum cytoplasm developed through a combination of conventional and molecular breeding approaches
-
- Journal:
- Plant Genetic Resources / Volume 2 / Issue 2 / August 2004
- Published online by Cambridge University Press:
- 16 October 2024, pp. 131-139
-
- Article
- Export citation
A modified dumbbell-shaped highly efficient broadband multifunctional polarizer for X and Ku band applications
-
- Journal:
- International Journal of Microwave and Wireless Technologies / Volume 16 / Issue 9 / November 2024
- Published online by Cambridge University Press:
- 16 October 2024, pp. 1552-1560
-
- Article
- Export citation
The (Il)legitimacy of Constitutional Amendments in Africa and Democratic Backsliding
-
- Journal:
- Asian Journal of Comparative Law / Volume 19 / Issue 3 / December 2024
- Published online by Cambridge University Press:
- 16 October 2024, pp. 473-492
-
- Article
- Export citation