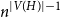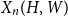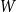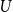Refine search
Actions for selected content:
52651 results in Statistics and Probability
Portfolio management under drawdown constraint in discrete-time financial markets
- Part of
-
- Journal:
- Journal of Applied Probability / Volume 60 / Issue 1 / March 2023
- Published online by Cambridge University Press:
- 09 December 2022, pp. 127-147
- Print publication:
- March 2023
-
- Article
- Export citation
On the splitting and aggregating of Hawkes processes
- Part of
-
- Journal:
- Journal of Applied Probability / Volume 60 / Issue 2 / June 2023
- Published online by Cambridge University Press:
- 09 December 2022, pp. 676-692
- Print publication:
- June 2023
-
- Article
- Export citation
Fluctuations of subgraph counts in graphon based random graphs
- Part of
-
- Journal:
- Combinatorics, Probability and Computing / Volume 32 / Issue 3 / May 2023
- Published online by Cambridge University Press:
- 09 December 2022, pp. 428-464
-
- Article
- Export citation
5 - Sex and the Single Patient
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 97-120
-
- Chapter
- Export citation
Dedication
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp v-v
-
- Chapter
- Export citation
2 - The Diceman Cometh
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 28-53
-
- Chapter
- Export citation
7 - Time’s Tables
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 135-156
-
- Chapter
- Export citation
Index
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 317-322
-
- Chapter
- Export citation
10 - The Law Is a Ass
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 206-233
-
- Chapter
- Export citation
1 - Circling the Square
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 1-27
-
- Chapter
- Export citation
6 - A Hale View of Pills (and Other Matters)
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 121-134
-
- Chapter
- Export citation
Contents
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp vii-viii
-
- Chapter
- Export citation
Epigraph
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp vi-vi
-
- Chapter
- Export citation
4 - Of Dice and Men*
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 74-96
-
- Chapter
- Export citation
Preface to the First Edition
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp xiii-xv
-
- Chapter
- Export citation
Preface to the Second Edition
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp ix-xii
-
- Chapter
- Export citation
Notes
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 297-316
-
- Chapter
- Export citation
Permissions
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp xvi-xvi
-
- Chapter
- Export citation
12 - Going Viral
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 255-296
-
- Chapter
- Export citation
3 - Trials of Life
-
- Book:
- Dicing with Death
- Published online:
- 18 November 2022
- Print publication:
- 08 December 2022, pp 54-73
-
- Chapter
- Export citation